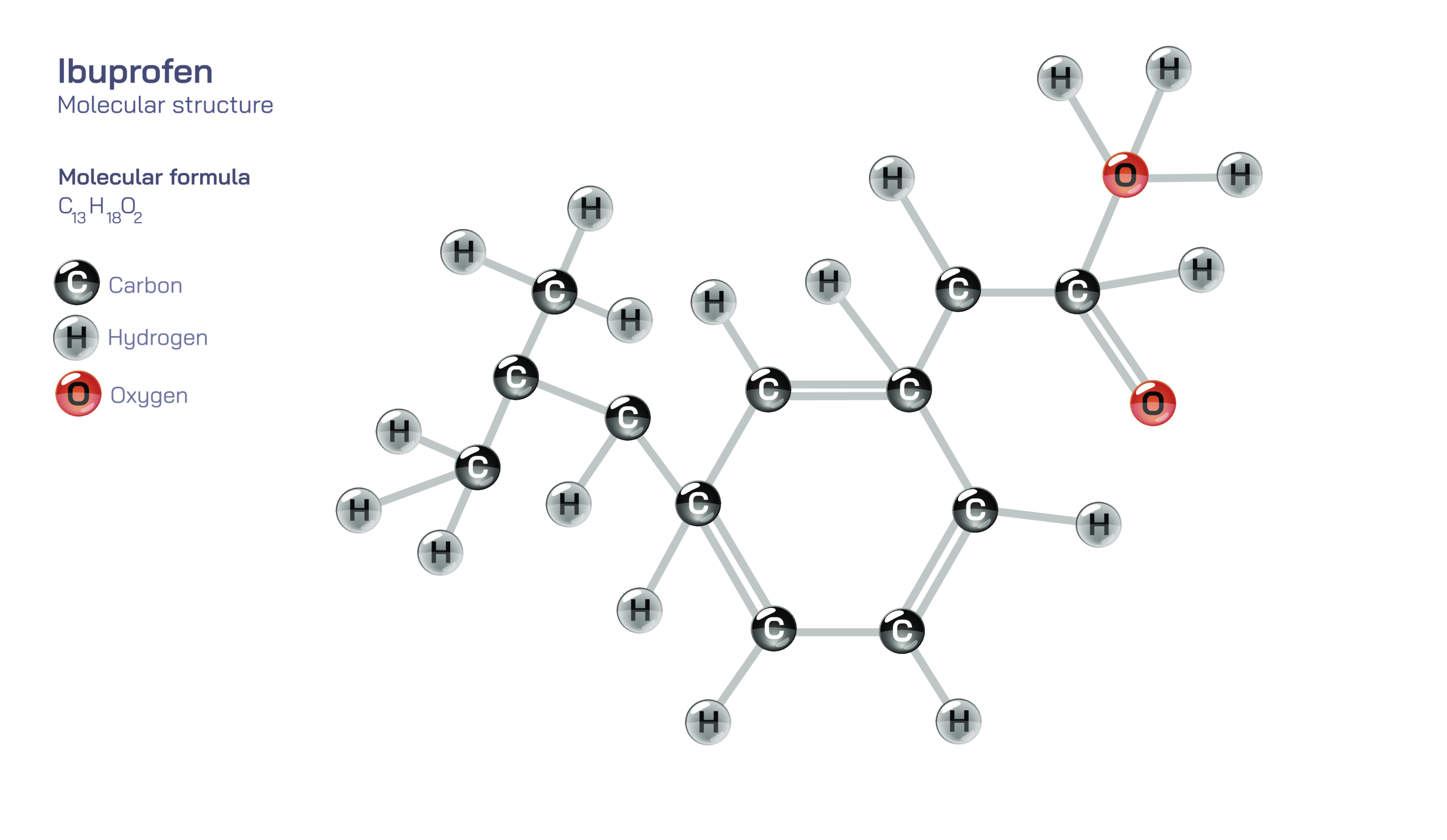
Ibuprofen Chemical Structure — C₁₃H₁₈O₂ Molecule Diagram and Uses Explained
Ibuprofen is one of the most widely used medicines in the world, and although many people recognize it simply as a pain-relieving tablet, its real significance can only be understood by examining the chemistry of the molecule itself — how its atoms are arranged, how that structure interacts with biological systems, and how that interaction ultimately eases pain, fever, and inflammation. With the molecular formula C₁₃H₁₈O₂, ibuprofen belongs to the class of non-steroidal anti-inflammatory drugs (NSAIDs), but the deeper story lies in the specific geometry of its carbon skeleton and the functional groups attached to it. Ibuprofen is built around a hydrophobic aromatic ring fused to a branched hydrocarbon chain ending in a carboxylic acid group. This structural combination allows the molecule to cross biological membranes easily due to its hydrophobic region, and then exert pharmacological effects due to the reactive carboxyl group that anchors ibuprofen to its biological targets. In this way, every aspect of ibuprofen’s behavior — from absorption into the bloodstream to binding inside enzymes — emerges from its chemical architecture.
The most defining part of the ibuprofen molecule is the presence of a benzene ring, a six-carbon aromatic structure that makes the molecule fat-soluble and able to travel through the lipid-rich environment of cell membranes. Attached to this ring is a branched alkyl chain that includes a chiral carbon atom, meaning that ibuprofen exists in mirror-image forms called enantiomers. Commercial ibuprofen consists of a racemic mixture containing both the S- and R- forms, but only the S-enantiomer is biologically active in reducing inflammation and pain, highlighting how not only the atoms themselves but their three-dimensional positioning affects the drug’s behavior. At the end of the carbon chain sits a carboxylic acid group (–COOH), and this acidic site is the functional core of ibuprofen’s pharmacological activity. It is the part of the molecule that binds to enzymes in the cyclooxygenase (COX) family, blocking them from converting arachidonic acid into prostaglandins — signaling molecules that promote pain sensation, fever, and inflammation. When this binding occurs, prostaglandin production decreases, and with lower prostaglandin levels the body responds with less pain, less swelling, and lowered temperature. Thus, ibuprofen does not numb the body or suppress the brain’s perception of pain; instead, it interrupts biological chemistry upstream where pain-promoting molecules are synthesized.
The structure of ibuprofen also determines how the body absorbs, distributes, breaks down, and eliminates it. Because of its hydrophobic aromatic portion, the drug is efficiently absorbed through the stomach and small intestine after oral administration. Once in the bloodstream, ibuprofen binds strongly to plasma proteins, especially albumin, which allows it to circulate effectively across tissues where inflammation is present. The carboxyl group is responsible not only for binding to enzymes but also for determining how the body metabolizes ibuprofen. Liver enzymes modify the carboxylic group, producing more water-soluble metabolites that can be excreted through the kidneys. This balance between fat solubility and metabolic convertibility — provided by the pairing of the benzene ring and the acidic group — is why ibuprofen acts quickly yet does not stay in the body for excessively long periods. The chemical design makes the drug effective, temporary, and biologically manageable.
Because ibuprofen targets pathways involved in pain and inflammation, its uses extend across many common conditions. It is used to relieve headaches, migraines, muscle pain, menstrual cramps, toothaches, joint pain, post-exercise soreness, and fever associated with infections. In inflammatory diseases such as arthritis, ibuprofen helps reduce swelling in joints, improving mobility and lowering discomfort. Its ability to reduce fever makes it an essential component of self-care during illnesses that elevate body temperature. What makes ibuprofen especially valuable is its balance of potency and tolerability: it provides strong anti-inflammatory relief while generally allowing individuals to remain alert and functional, unlike sedative or narcotic painkillers. The molecule’s design is therefore not only a triumph of organic chemistry but also a model of medicinal engineering in which a single structure interacts selectively and usefully with biological systems.
However, the same molecular features that make ibuprofen so effective also require careful use. The carboxylic acid group that binds to COX enzymes can irritate the lining of the stomach because it temporarily reduces the production of prostaglandins that normally protect the stomach wall. When prostaglandin levels fall, stomach acid becomes harsher on tissues, and long-term or high-dose use can contribute to gastric irritation, heartburn, or in severe cases ulcer development. The mechanism is therefore not a side effect unrelated to its function — it is the other side of the same biological action that relieves pain. Similarly, prostaglandins support kidney blood flow, and prolonged suppression in susceptible individuals can stress kidney function. Because ibuprofen temporarily alters blood chemistry related to clotting, individuals with cardiovascular disease must also use it thoughtfully. These considerations do not make ibuprofen unsafe; rather, they highlight that its powerful chemical action must be balanced with reasonable and informed dosing. The key is understanding that the same structure that makes ibuprofen effective is also responsible for side effects when used without moderation.
The impact of ibuprofen extends beyond individual healing and into medical history. Before drugs like ibuprofen were developed, managing chronic inflammation or moderate pain often meant relying on opioid medications or steroids, both of which have serious long-term consequences. Ibuprofen and similar NSAIDs provided a safer and non-addictive alternative that could be used daily when needed, dramatically improving quality of life for people with ongoing inflammatory pain. Its discovery and refinement demonstrated how designing a molecule around selective enzyme inhibition could relieve pain not by masking symptoms but by altering biochemical pathways in a measured and reversible manner. The structural insights behind ibuprofen paved the way for many more targeted therapeutic compounds across modern medicine.
Ultimately, the identity of ibuprofen is inseparable from its molecular structure. Every characteristic — fast absorption, pain relief, reduction in swelling, fever control, temporary biological presence, and occasional gastrointestinal irritation — can be traced directly to the arrangement of the thirteen carbons, eighteen hydrogens, and two oxygens in the C₁₃H₁₈O₂ framework. The benzene ring provides lipid solubility and membrane penetration; the branched chiral side chain determines biological specificity; and the carboxyl group governs enzyme binding, physiological action, and metabolic breakdown. Ibuprofen’s story shows that the power of a medicine does not come from strength or complexity alone, but from precise structural design that harmonizes with the chemistry of life. It is a clear illustration of how the architecture of atoms in a single small molecule can shape medical practice, relieve suffering, and improve daily life for millions — not through chance, but through the profound connection between molecular structure and biological effect.