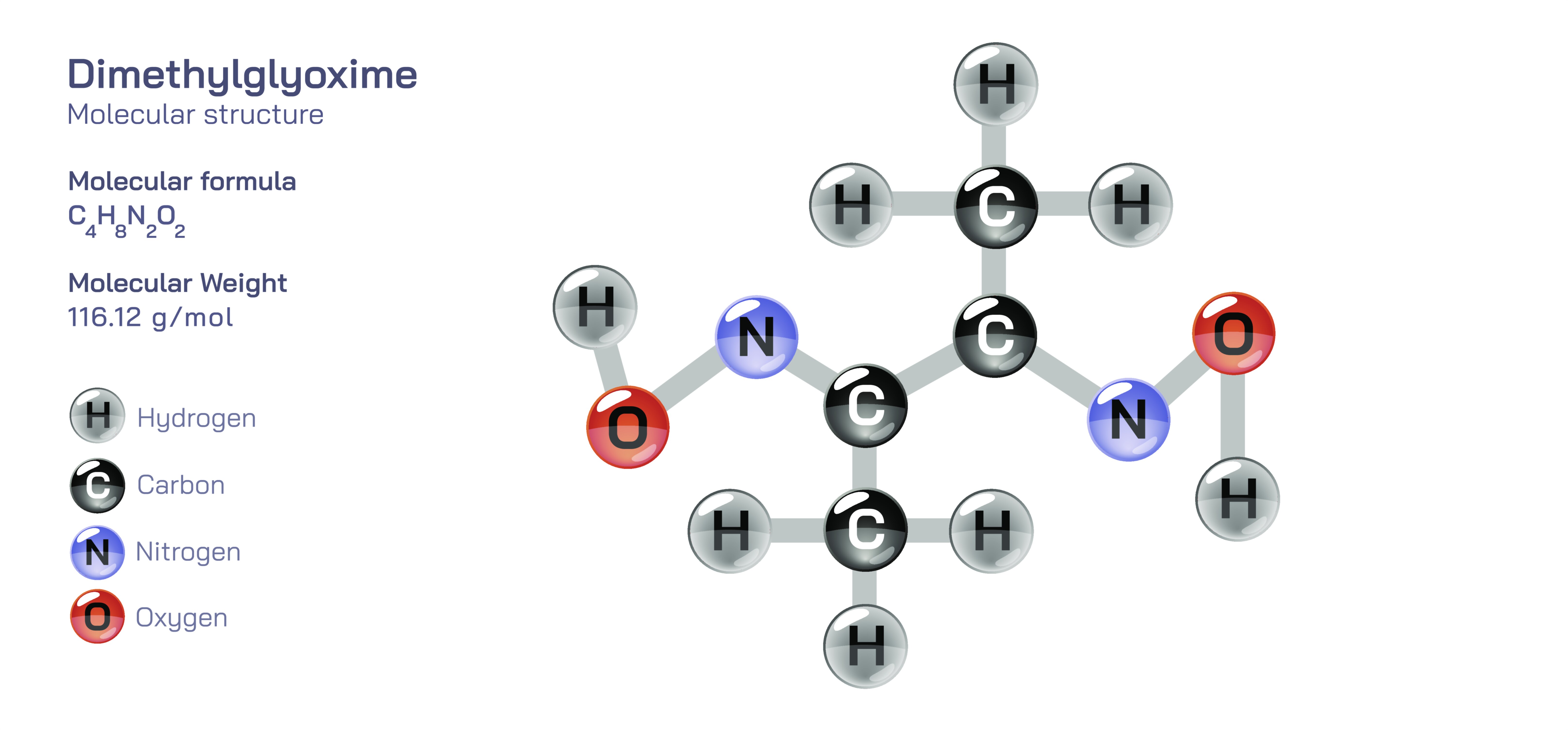
Dimethylglyoxime — Organic Analytical Reagent
Dimethylglyoxime is one of the most iconic and historically significant organic analytical reagents in classical and modern chemistry, renowned for its precision, selectivity, and the visually striking reactions it produces when interacting with specific metal ions. Although its name may appear technical, dimethylglyoxime embodies the central idea of qualitative analysis: using the structural characteristics of a carefully designed molecule to reveal the presence of particular substances through distinct physical changes. For more than a century, this bright red, crystalline compound has played a major role in analytical laboratories across the world, especially because of its unmatched selectivity toward nickel ions. When dimethylglyoxime encounters nickel in the right conditions, it forms an intensely colored, stable complex whose appearance immediately confirms the presence of the metal. Yet the importance of dimethylglyoxime extends far beyond this reaction alone. Its chemical structure, solubility behavior, reactivity, coordination tendencies, and role in various industries demonstrate how a carefully crafted organic molecule can become an indispensable tool for detecting, separating, and understanding the metals that shape modern materials, technologies, and environmental systems.
At its molecular core, dimethylglyoxime consists of a symmetrical organic framework containing two oxime functional groups positioned in such a way that they can act cooperatively as a chelating agent. Chelation refers to the ability of a molecule to bind a metal ion using multiple points of attachment, effectively gripping the metal in a stable organic “claw.” In the case of dimethylglyoxime, the two oxime groups form a bidentate system that is ideally positioned to wrap around a metal center. This precise spatial arrangement gives dimethylglyoxime a strong affinity for metals that match its structural requirements, especially nickel. The reagent’s geometry allows it to form a square-planar complex with nickel ions, locking the metal into a rigid structure that remains stable even when subjected to washing or isolation procedures. This stability is one of the main reasons dimethylglyoxime became a standard reagent in gravimetric analysis, where chemists isolate and weigh a precipitate to determine the concentration of a metal ion. The formation of a red nickel–dimethylglyoxime complex is so definitive and reproducible that it became a staple procedure for analytical chemists, metallurgists, and industrial technicians.
One of the most compelling conceptual aspects of dimethylglyoxime is its ability to exhibit such extraordinary selectivity. Many organic reagents react with a variety of ions, producing mixtures of products or vague color changes that complicate interpretation. Dimethylglyoxime, in contrast, binds strongly and distinctively to nickel, producing a deep, rose-red precipitate that is unmistakable. This selectivity arises from the complementary shape and electronic environment between the reagent and the metal ion. Nickel’s preferred geometry and electron configuration match the binding arrangement offered by dimethylglyoxime, allowing a stable coordination complex to form. The reagent’s organic framework is carefully positioned so that only metals with specific structural compatibility can fit into this coordination pattern. While dimethylglyoxime can react with certain other metals under special conditions, none form complexes as stable, insoluble, or dramatically colored as the nickel complex. This creates a clear, binary analytical signal: if the red precipitate appears, nickel is present. This simplicity and reliability made dimethylglyoxime a transformative tool at a time when analytical chemistry relied heavily on observational techniques rather than modern instrumentation.
The use of dimethylglyoxime spread rapidly in the early development of coordination chemistry. As scientists sought to understand how metal ions interact with organic ligands, dimethylglyoxime provided a perfect lens through which to study these behaviors. Its predictable reactions helped shape early theories of complex formation, geometry, and stability. Because the nickel complex formed by dimethylglyoxime exhibited consistent properties across laboratories and generations, it became a reference point for exploring how other ligands behave. The reagent also revealed deeper insights into why certain metals prefer particular coordination environments and how ligand architecture influences selectivity. In this way, dimethylglyoxime not only served as a practical analytical reagent but also as an instructional model, helping chemists visualize and conceptualize the nature of metal–ligand interactions.
Dimethylglyoxime’s role in industry expanded significantly because of the widespread use of nickel in metallurgical processes, electroplating technology, stainless steel manufacturing, battery production, and catalyst development. Ensuring high purity in nickel-containing materials requires accurate and reliable methods for detecting trace amounts of the metal in ores, alloys, industrial waters, and waste streams. Dimethylglyoxime became indispensable in these contexts. Industrial chemists use the reagent to verify nickel content during quality control checks, monitor environmental contamination, and analyze metal mixtures in electroplating baths. The unmistakable nature of the nickel–dimethylglyoxime complex allows rapid and clear identification, providing both qualitative confirmation and, in gravimetric procedures, quantitative precision. Even in an age dominated by advanced instruments such as ICP spectrometers and atomic absorption analyzers, dimethylglyoxime remains a trusted and cost-effective method for routine checks where simplicity, robustness, and low cost are priorities.
Environmental science also benefits from the unique properties of dimethylglyoxime. Nickel contamination in soil and water can arise from mining operations, industrial discharges, improper disposal of electronic waste, and leaching from metal alloys. Detecting nickel reliably in environmental samples is essential for assessing pollution, determining ecological impact, and enforcing safety regulations. Dimethylglyoxime facilitates such detection by providing a clear signal even when nickel concentrations are low or when water samples contain competing ions. Environmental monitoring laboratories often use dimethylglyoxime-based methods for rapid screening before proceeding to more complex analyses. The reagent's strong selectivity reduces the risk of false positives, helping ensure that environmental assessments remain accurate and trustworthy.
In analytical teaching laboratories, dimethylglyoxime continues to play a central role in introducing students to classical qualitative analysis. Its dramatic color change and visually striking precipitate make it ideal for demonstrating the fundamental concepts of complexation, precipitation, ligand coordination, and analytical selectivity. Students can easily observe the formation of the reddish nickel complex, gaining a concrete understanding of how molecules interact at the chemical level. This hands-on experience serves as a bridge to more advanced analytical methods, reinforcing foundational principles that remain relevant even as technology evolves. Dimethylglyoxime experiments also help students learn about purification, filtration, weighing, and sample preparation—skills still essential in modern laboratory practice.
The physical properties of dimethylglyoxime also contribute to its utility. It appears as a crystalline, pale powder that dissolves in certain organic solvents and occasionally in alkaline aqueous solutions. This solubility pattern allows chemists to tailor reaction conditions based on the specific analytical need. When added to solutions containing nickel ions, the reagent transitions from a dissolved state to an insoluble complex, producing an immediate visible reaction. This dramatic insolubility is advantageous for gravimetric analysis, where the solid precipitate can be filtered, dried, and weighed. In classical chemistry, this straightforward mass-based method provided some of the most accurate determinations of metal concentrations long before digital instruments existed. Dimethylglyoxime is therefore deeply connected to the roots of quantitative analytical science.
Although best known for its affinity for nickel, dimethylglyoxime also plays a role in detecting other metals under modified conditions. It can form complexes with palladium, cobalt, and other transition metals, although these reactions are usually less selective or require more stringent environmental controls. In some cases, chemists adjust pH levels, solvent systems, or temperature conditions to encourage or prevent certain complexation paths. This adaptability highlights how the structure of dimethylglyoxime interacts sensitively with chemical environments, influencing its selectivity and reactivity. These nuances offer fertile ground for research into coordination chemistry, ligand behavior, and the fine-tuning of analytical reagents.
Dimethylglyoxime’s story also provides an important lesson about the intersection of structure and function in chemistry. The molecule’s symmetrical framework, dual oxime groups, and spatial configuration create a perfect “lock” for the “key” of nickel ions. This structural complementarity demonstrates how molecular design can achieve precise analytical goals. Chemists who understand this relationship can design new reagents with targeted affinities for other metals, pollutants, or biomolecules. In this sense, dimethylglyoxime represents not just a reagent but a model for the development of specialized ligands in analytical, medicinal, and materials chemistry. Its success underscores how careful molecular engineering leads to robust and predictable chemical behavior.
In practical laboratory usage, the behavior of dimethylglyoxime also teaches chemists about solution chemistry, pH control, and the effects of competing ions. The reagent typically requires slightly alkaline conditions to react effectively with nickel ions. Under too acidic conditions, the oxime groups do not deprotonate sufficiently, reducing their ability to bind metals. Conversely, excessively alkaline conditions may encourage side reactions. This sensitivity to pH illustrates the broader principle that molecular reactivity is governed by environmental conditions. Instructors often emphasize these conditions when teaching students how to prepare analytical mixtures correctly, reinforcing key ideas about the influence of solution chemistry on reaction outcomes.
From a conceptual standpoint, the importance of dimethylglyoxime lies not only in its practical applications but also in its ability to show how molecular structure governs selectivity, stability, and reactivity. The reagent captures the essence of analytical chemistry: designing molecules that reveal information about other molecules. It forms visible structures that embody abstract chemical processes, making the intangible world of ions and ligands suddenly accessible through color and form. For generations of scientists, the red nickel–dimethylglyoxime complex has provided a vivid demonstration of how chemistry transforms the invisible into the observable.
Ultimately, dimethylglyoxime stands as a testament to the power of molecular design in analytical science. Its distinctive structure creates exceptional selectivity for nickel, enabling accurate identification and quantification across diverse fields. The reagent’s influence spans classical chemistry, metallurgy, environmental monitoring, educational laboratories, and the foundational study of coordination chemistry. Dimethylglyoxime’s enduring value highlights how a single molecule, crafted with structural precision, can shape entire scientific disciplines and maintain relevance across decades of evolving technologies. Through its selective binding, vivid reactions, and conceptual clarity, dimethylglyoxime continues to demonstrate the profound connection between chemical structure and analytical insight, anchoring its place as one of the most celebrated reagents in the world of chemistry.