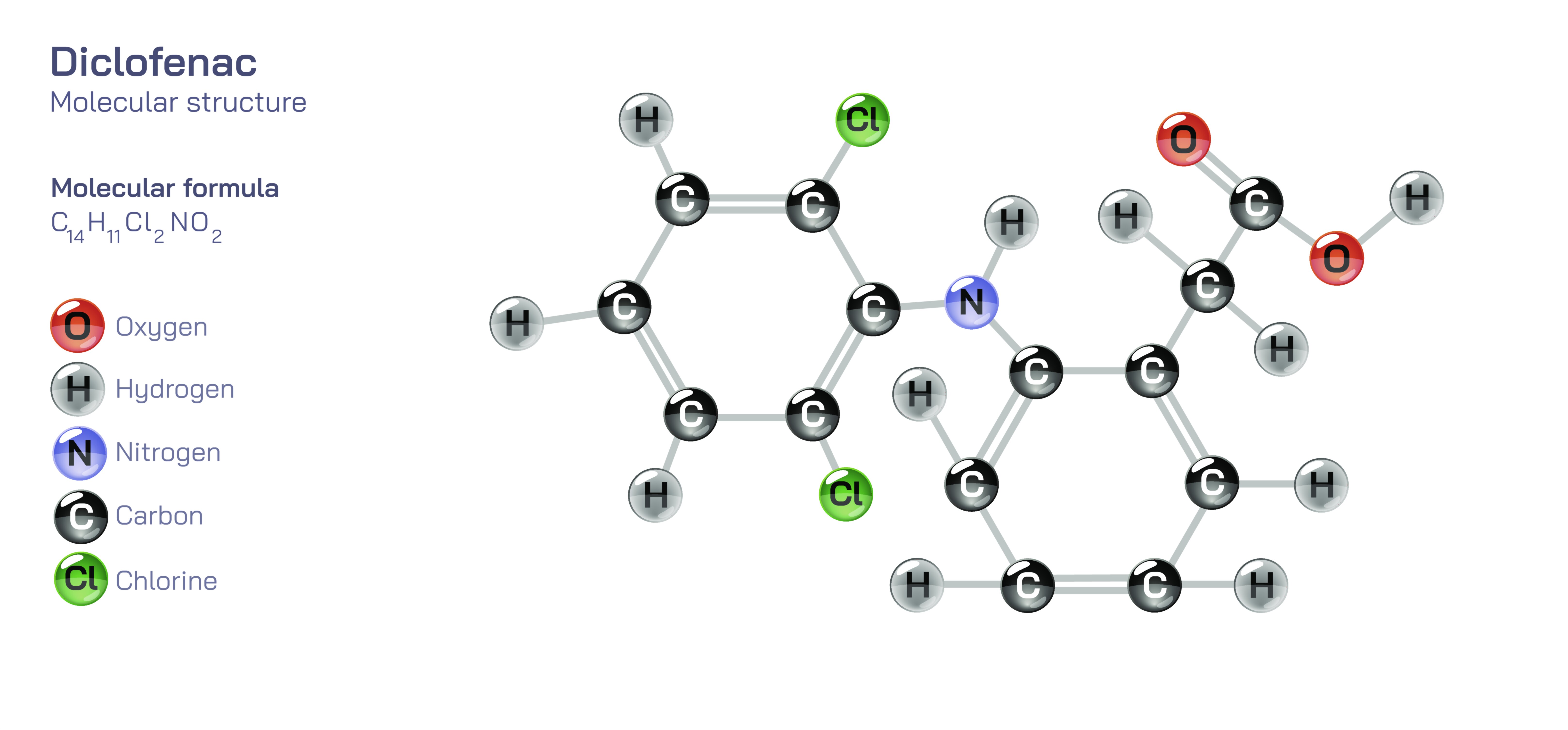
Diclofenac — Anti-Inflammatory Drug Molecular Structure
Diclofenac is one of the most widely used and scientifically studied non-steroidal anti-inflammatory drugs, commonly abbreviated as an NSAID, and its molecular structure, pharmacological behavior, and clinical relevance reveal the intricate ways in which chemistry, physiology, and medicine intersect. Although people often recognize diclofenac in the form of tablets, injections, topical gels, or medicated patches, its therapeutic power originates from the specific arrangement of atoms within the diclofenac molecule and the way that structure interacts with biological systems. Diclofenac’s molecular identity is defined by a combination of aromatic rings, chlorine substituents, and an amino-phenylacetic acid backbone. This configuration gives the molecule the ability to pass through lipid membranes, bind selectively to enzymes involved in inflammation, and modulate biochemical pathways responsible for pain, swelling, and tissue responses. Understanding diclofenac at the structural level offers deeper appreciation of how its chemical framework leads to therapeutic action, how the drug behaves in the body, how it produces both benefits and side effects, and why it has become essential in modern medical practice.
At the heart of diclofenac’s molecular structure is a distinctive pair of connected aromatic rings—one containing two chlorine atoms and the other linked through an amino bridge to an acetic acid side chain. The presence of the chlorine atoms is far more than a cosmetic chemical feature; these atoms modify the electronic distribution in the molecule, shifting its polarity and increasing its ability to bind tightly to specific biological targets. The molecule’s two-ring system creates a planar arrangement that allows diclofenac to slip into the active sites of cyclooxygenase enzymes, often abbreviated as COX enzymes, which play a central role in producing prostaglandins. Prostaglandins are signaling molecules that mediate pain, fever, and inflammatory responses. By fitting into the enzyme’s active site in place of the enzyme’s natural fatty acid substrates, diclofenac blocks the formation of these prostaglandins. This blockage reduces inflammation and pain at the source, rather than merely masking symptoms, giving diclofenac a powerful therapeutic effect in conditions such as arthritis, musculoskeletal injuries, menstrual cramps, and postoperative inflammation.
Diclofenac’s structural arrangement also contributes to its pharmacokinetics—how it is absorbed, distributed, metabolized, and eliminated in the body. The presence of both hydrophobic aromatic rings and a polar carboxylic acid group creates a balanced molecular personality that allows it to move through both lipid-rich environments, such as cell membranes, and aqueous environments, such as the bloodstream. When taken orally, diclofenac is rapidly absorbed, but it undergoes substantial first-pass metabolism in the liver, meaning that a portion of the drug is transformed before reaching systemic circulation. This transformation involves hydroxylation and conjugation reactions that convert diclofenac into more water-soluble forms for elimination. Its metabolism underscores the fact that even small changes in molecular structure can influence the drug’s potency, duration of action, and safety profile. For example, formulations such as extended-release tablets or topical preparations modify how quickly diclofenac enters the bloodstream or penetrates tissues, tailoring the molecule’s behavior to therapeutic needs.
2. This slight selectivity contributes to its strong anti-inflammatory action while offering a somewhat reduced—but not eliminated—risk of gastrointestinal problems compared to older NSAIDs. However, because diclofenac still inhibits COX-1 to a meaningful degree, the molecular structure that gives it therapeutic power also contributes to potential side effects such as stomach irritation, ulcer formation, and bleeding risk, particularly with long-term use. This dual action reflects the complexity of drug design, where the same structural features enabling therapeutic benefits may also introduce risks.
Beyond inflammation control, diclofenac’s structure interacts with other physiological pathways in ways that influence its therapeutic versatility. Its biochemical effects can help reduce symptoms in conditions such as gout, tendonitis, ankylosing spondylitis, and migraines. The molecule’s ability to reduce prostaglandin production can also contribute to decreased fever, although diclofenac is more renowned for its pain-relieving and anti-inflammatory actions than for its antipyretic effects. In postoperative care, the rapid onset of action associated with diclofenac’s molecular mobility makes it useful in managing acute pain without relying exclusively on opioid medications. In topical formulations, the molecule’s size, polarity, and aromatic character enable it to penetrate skin layers while maintaining sufficient localization to reduce swelling and pain in joints or muscles with minimal systemic absorption. These topical preparations demonstrate how manipulation of the same molecular structure—through gels, patches, or emulsions—can produce a range of therapeutic outcomes tailored to patient needs.
Diclofenac’s structure also relates to its environmental and ecological impact, a topic of increasing concern in recent decades. Certain wildlife species, particularly vultures, suffered catastrophic population declines in regions where livestock treated with diclofenac left behind residues in carcasses. The drug’s specific structural and metabolic properties caused fatal kidney damage in these birds when they consumed treated animals. This consequence, unforeseen when the drug was originally developed, highlights how the stability and biochemical reactivity of a molecular structure extend their influence beyond human medicine into ecological systems. Regulatory agencies responded with restrictions and alternative veterinary medications, but the case remains a powerful reminder that molecular design carries far-reaching implications.
Pharmaceutically, the molecular structure of diclofenac has inspired innovations in drug formulation aimed at reducing side effects and improving therapeutic performance. For example, diclofenac salts—such as diclofenac sodium and diclofenac potassium—alter the molecule’s solubility and speed of absorption, making the drug more adaptable for different medical scenarios. Enteric-coated formulations protect the drug from dissolving in the stomach, reducing irritation by delaying absorption until it reaches the intestines. Extended-release preparations adjust the rate at which the drug enters the bloodstream so that its effects last longer while reducing peaks that might cause adverse reactions. Patch-based and topical forms target localized areas, minimizing gastrointestinal or cardiovascular strain. Each of these innovations reflects how a deep understanding of molecular structure enables pharmaceutical scientists to shape drug behavior without altering the core chemical identity of the molecule.
Diclofenac’s molecular interactions also extend into cellular and biochemical pathways beyond COX inhibition. Studies suggest that the drug can modulate ion channels, influence oxidative stress pathways, and interact with components of the inflammatory cascade in ways not fully understood when the compound was first developed. These additional effects do not overshadow its primary mechanism but provide insight into why diclofenac may work particularly well for certain conditions or why it may cause unexpected side effects in some individuals. The structural basis for these interactions underscores the fact that a molecule’s biological activity can reach far beyond its originally intended target, shaped by the complex interplay between structure, reactivity, and biological environment.
A key conceptual insight arising from diclofenac’s molecular structure is the broader principle that small structural variations in a molecule can lead to profound differences in therapeutic action. Diclofenac belongs to a class of NSAIDs that share structural similarities, yet many related compounds exhibit distinct potency, selectivity, and side-effect profiles. This variation reflects how sensitive biological systems are to molecular geometry and electronic distribution. The chlorine-containing aromatic rings, amino linkage, and carboxylate group in diclofenac represent a carefully tuned combination that optimizes binding strength and therapeutic effect while balancing solubility and reactivity. This balance illustrates why medicinal chemistry is both an art and a science—each functional group, bond angle, and atom substitution shapes how a molecule behaves inside the body.
Diclofenac also provides a framework for discussing the principles of drug safety and monitoring. Because the molecule affects physiological pathways linked to gastrointestinal protection, blood flow, and kidney function, healthcare providers must consider patient-specific factors such as age, medical history, existing drug regimens, and underlying health conditions. The drug’s molecular ability to reduce prostaglandins can, in some cases, constrict blood vessels supplying the kidneys or influence the cardiovascular system in ways that require careful dosage control. Patients with hypertension, heart disease, or compromised kidney function may experience increased risk when using diclofenac, and these risks arise directly from how the molecule interacts with enzymes and tissues at the structural level.
Another perspective on diclofenac’s molecular nature comes from the field of pharmaceutical development and regulatory science. Before a drug like diclofenac reaches the market, its structure undergoes rigorous testing for stability, efficacy, pharmacological activity, and safety. Researchers analyze how the molecule behaves under different temperatures, pH levels, and metabolic conditions. They test how it binds to proteins, how it distributes through tissues, and how it is broken down in the body. They must also determine whether structural metabolites of the drug retain activity, become toxic, or interact with other biological molecules. The molecular structure shapes every aspect of this testing process, guiding decisions about effective dosing, safe exposure levels, and regulatory classification.
From a conceptual viewpoint, diclofenac represents the powerful connection between molecular design and human experience. The relief felt by someone suffering from arthritis pain, the reduction in inflammation after an injury, or the improved mobility gained through chronic pain management all originate from the precise spatial arrangement of atoms in the diclofenac molecule. This illustrates the broader lesson that medicines are not abstract remedies but physical structures that interact with the human body at the molecular level. The effectiveness of a drug depends on how well its structure fits a biological target, how predictably it moves through tissues, and how safely it can be metabolized.
Ultimately, diclofenac stands as a compelling example of the intricate relationship between chemistry and medicine. Its molecular structure dictates how it works, how it behaves in the body, how it must be formulated, and how it must be regulated. It serves as a reminder that every therapeutic effect has a molecular origin, and every medical benefit arises from the detailed interplay between structure and biology. Through diclofenac, one gains insight into the broader principles of medicinal chemistry, the careful craftsmanship behind drug design, and the profound impact that a well-designed molecule can have on health, quality of life, and the treatment of human disease.