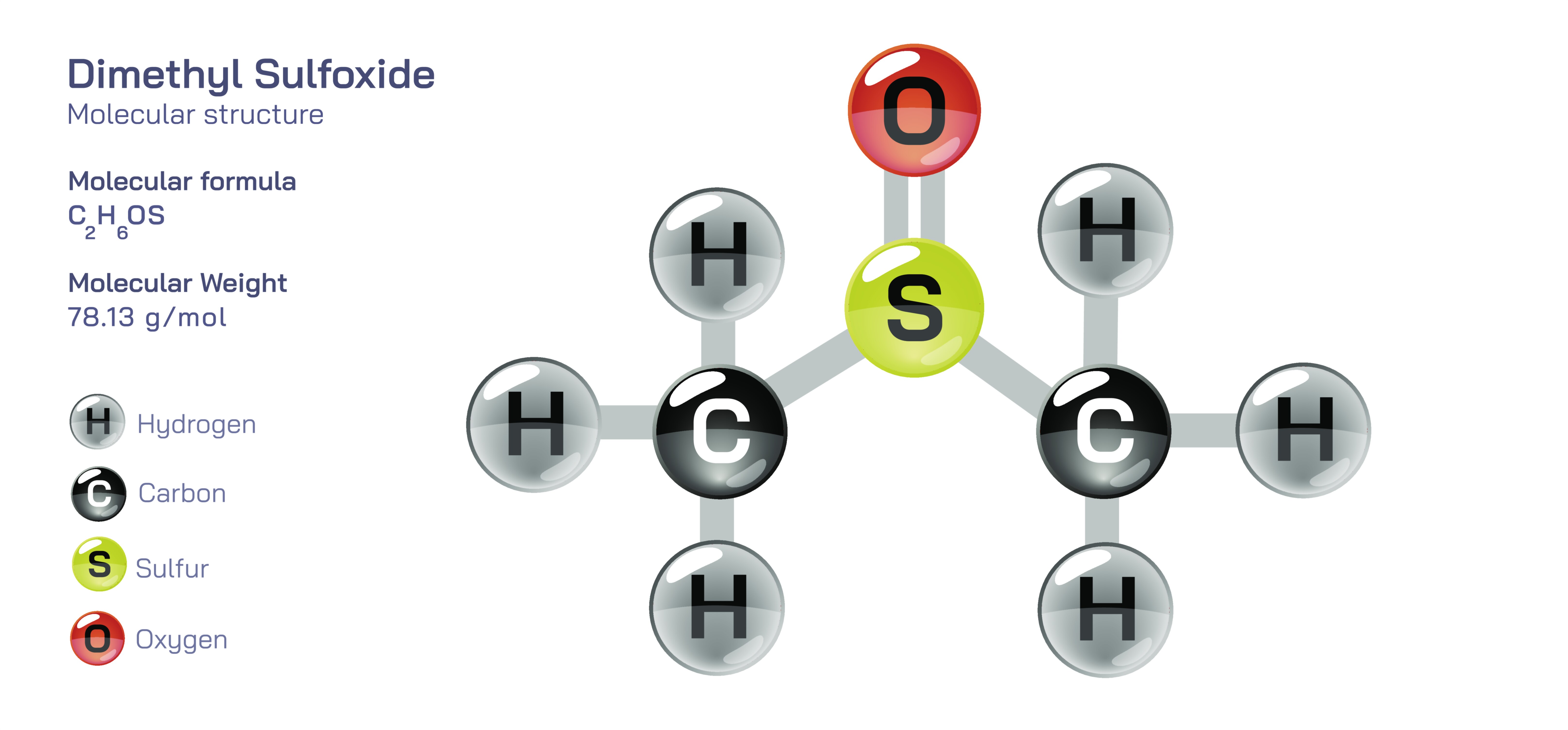
Dimethyl Sulfoxide — Organic Sulfur Compound
Dimethyl sulfoxide, widely known as DMSO, is a uniquely versatile organic sulfur compound whose molecular structure, physical behavior, and biological interactions make it one of the most remarkable and multifaceted substances in modern chemistry, medicine, and materials science. Although its structure appears simple at first glance—consisting of a sulfur atom bonded to an oxygen atom and two methyl groups—this arrangement generates an unusual combination of polarity, electron distribution, and structural stability that allows DMSO to interact with an extraordinary range of materials. It dissolves polar and nonpolar substances, penetrates biological membranes with ease, influences molecular reactivity, and exhibits properties that seem contradictory until one understands how its molecular structure operates. Because of this performance, DMSO has earned extensive use in pharmaceuticals, cryopreservation, analytical chemistry, polymer science, and even alternative medicine, though many of its biological effects require caution, regulation, or deeper scientific scrutiny. The dual nature of its behavior—safe in some contexts, risky in others—makes DMSO a compelling example of how molecular architecture governs function in both beneficial and potentially hazardous ways.
At the molecular level, the defining feature of DMSO is the sulfoxide functional group, in which sulfur is double-bonded to oxygen while simultaneously carrying two organic substituents. This sulfur–oxygen arrangement creates a strongly polarized bond because oxygen, being more electronegative, draws electrons toward itself, leaving the sulfur end relatively electron-rich. As a result, DMSO becomes a highly polar aprotic solvent. This classification explains much of its behavior: it has significant dipole strength, lacks hydrogen atoms capable of hydrogen bonding donation, and maintains structural rigidity with substantial electron density around the sulfur. These characteristics allow DMSO to dissolve polar salts, coordinate with metals, and break apart strong intermolecular interactions in solutes. At the same time, its two methyl groups render it compatible with nonpolar hydrocarbons, enabling DMSO to blend seamlessly with oils, polymers, organic compounds, and membranes. This hybrid personality makes DMSO one of the few solvents that can dissolve substances spanning nearly the entire polarity spectrum, from organic dyes and plastics to inorganic salts and metal complexes.
Its solvation power arises from this polarity and from the ability of DMSO molecules to arrange themselves in highly ordered but flexible patterns in the liquid phase. These arrangements create a fluid environment that stabilizes ions and electron-rich species without donating hydrogen bonds. Because of this, DMSO has become a crucial solvent in reactions involving strong bases, organometallic compounds, nucleophiles, and other reactive species. Organic chemists rely on DMSO in reactions requiring high-energy intermediates or controlled reactivity. Furthermore, the high boiling point of DMSO allows reactions to be run at elevated temperatures without rapid solvent loss. This thermal stability, directly tied to its molecular structure, supports synthetic chemistry across laboratories and industrial settings.
DMSO’s ability to penetrate biological membranes is one of its most scientifically fascinating and practically important properties. Biological membranes are composed primarily of lipid bilayers that resist the entry of many chemicals. DMSO, because of its amphiphilic nature—a molecule with both polar and nonpolar character—interacts with both the hydrophilic heads and the hydrophobic tails of membrane lipids. This interaction permits DMSO to infiltrate membranes, carrying dissolved substances with it. This penetration power is both beneficial and dangerous. In medical research, it allows DMSO to act as a carrier or enhancer for drugs that struggle to enter cells. In cryopreservation, its membrane permeability protects cells from the formation of sharp ice crystals that would otherwise rupture cell walls during freezing. By interacting with water molecules and modifying ice growth patterns, DMSO prevents intracellular crystallization, giving it a central role in preserving sperm cells, embryos, stem cells, blood cells, and tissue samples. This cryoprotective function has transformed biotechnology and medicine by enabling long-term storage of biological materials that would be impossible to preserve using other solvents.
However, this membrane-interacting ability also demands caution because DMSO can transport harmful contaminants, toxins, or pathogens into living tissues if used irresponsibly. The same structural features that allow it to protect cells during freezing can also unintentionally carry unwanted chemicals deep into the body. This dual behavior underscores the essential principle that the function of a chemical arises from its structure, and the very properties that provide enormous benefits can also create risks if not properly controlled.
DMSO's interactions with biological tissues also extend to its anti-inflammatory and analgesic effects. Although originally explored as a possible pharmaceutical agent, DMSO’s medicinal pathway became controversial due to mixed research findings, regulatory concerns, and the potential for misuse. Nevertheless, it exhibits clear biological activity: DMSO reduces inflammation by modulating oxidative stress pathways, influencing free radical behavior, and affecting local cellular responses. Its analgesic effects are thought to result from changes in nerve conduction and membrane permeability. These features led to its acceptance in certain medical treatments, particularly in the management of interstitial cystitis, a chronic bladder condition. In veterinary medicine, DMSO has seen wider acceptance for its anti-inflammatory effects. Yet many of its marketed uses, especially in alternative medicine, remain scientifically unverified or insufficiently regulated. A full understanding of DMSO’s biological behavior—beneficial and risky alike—comes back to appreciating how its molecular structure interacts with tissues, membranes, and biochemical pathways.
The unique odor associated with DMSO—often described as garlicky or reminiscent of oysters—provides an interesting illustration of how molecular behavior becomes perceptible at the human sensory level. This odor originates not from DMSO itself but from its metabolic conversion into dimethyl sulfide, a volatile sulfur-containing compound. When DMSO passes through the body, enzymatic pathways reduce it, forming dimethyl sulfide, which is then released through breath and skin. This sensory side effect, though harmless, is a reminder that a molecule’s metabolism is often as significant as its initial structure in determining how it affects living organisms.
Beyond biological and pharmaceutical uses, DMSO has an important role in materials science. Its ability to dissolve a vast range of polymers makes it vital in polymer synthesis, fiber production, and processing technologies. DMSO acts as a medium for shaping high-performance materials, forming films, dissolving polymeric precursors, and enabling the production of flexible, durable, or chemically resistant materials. It supports the creation of synthetic fibers, cellulosic materials, and specialty plastics. DMSO’s compatibility with both organic and inorganic substances also makes it valuable in battery research, electronic materials development, and nanotechnology. It dissolves certain transition metal complexes and supports electrochemical reactions needed for developing advanced material systems.
In environmental chemistry, DMSO displays interesting behavior as well. While it is biodegradable and less persistent in the environment compared to many other organic solvents, its degradation products—such as dimethyl sulfide—can influence atmospheric chemistry. Dimethyl sulfide plays a role in cloud formation and climate processes. Therefore, the environmental pathway of DMSO establishes a connection between molecular behavior in solution and broader ecological or atmospheric effects. This illustrates how even laboratory or industrial chemicals, once released into wider systems, can influence natural cycles in unexpected ways.
In spectroscopy and analytical chemistry, DMSO is widely used as a solvent for nuclear magnetic resonance studies because it dissolves many organic molecules without reacting with them. Its spectral purity and ability to stabilize dissolved compounds make it one of the preferred solvents in experimental studies. The molecule also has a relatively simple infrared and NMR signature that allows clear identification of solute peaks. These analytical roles highlight how DMSO serves as a scientific facilitator, enabling the examination of substances that might otherwise be difficult to study.
Central to all these roles is the distinctive sulfoxide group, whose structure dictates how DMSO binds, dissolves, or interacts with other molecules. This group creates an electron-rich sulfur center that can coordinate with metals or polar molecules, while the attached methyl groups ensure compatibility with organic substances. The balance between the polarity of the S=O bond and the nonpolar characteristics of the methyl groups produces a solvent with highly unusual miscibility, stability, and reactivity patterns. These structural qualities also determine DMSO’s relatively high boiling point, its high dielectric property, and its thermal stability. Every feature—physical, chemical, biological—traces back to this arrangement of atoms.
The importance of DMSO extends into discussions of chemical safety and responsible usage. Because the molecule can carry dissolved substances into the body and influence cellular functions, improper handling can pose health risks. Wearing protective gloves, using proper ventilation, and avoiding contamination are essential practices when working with DMSO in laboratories and industrial settings. This necessity reinforces a major theme in chemical science: the properties that make a molecule useful also demand careful respect and understanding, particularly when those properties involve biological systems.
Ultimately, dimethyl sulfoxide stands as a prime example of how a simple molecular structure can produce an extraordinary range of effects across scientific fields. Its ability to dissolve almost anything, penetrate biological membranes, protect cells from ice, facilitate chemical reactions, stabilize polymers, and influence physiological processes demonstrates the profound impact that atomic arrangement can exert on the material world. Through DMSO, one can appreciate the fundamental truth that chemistry is not only the study of substances but the study of structure-driven behavior. Every interaction, from laboratory experiments to cellular processes, reflects how molecular architecture shapes function, reactivity, and application.
In this way, dimethyl sulfoxide becomes not merely a solvent or a chemical reagent but a lens through which the relationship between molecular structure and scientific utility becomes vividly clear. Its story bridges chemistry, biology, medicine, materials science, and environmental awareness, revealing how a single compound can transform countless processes through the subtle yet powerful influence of its underlying structural identity.