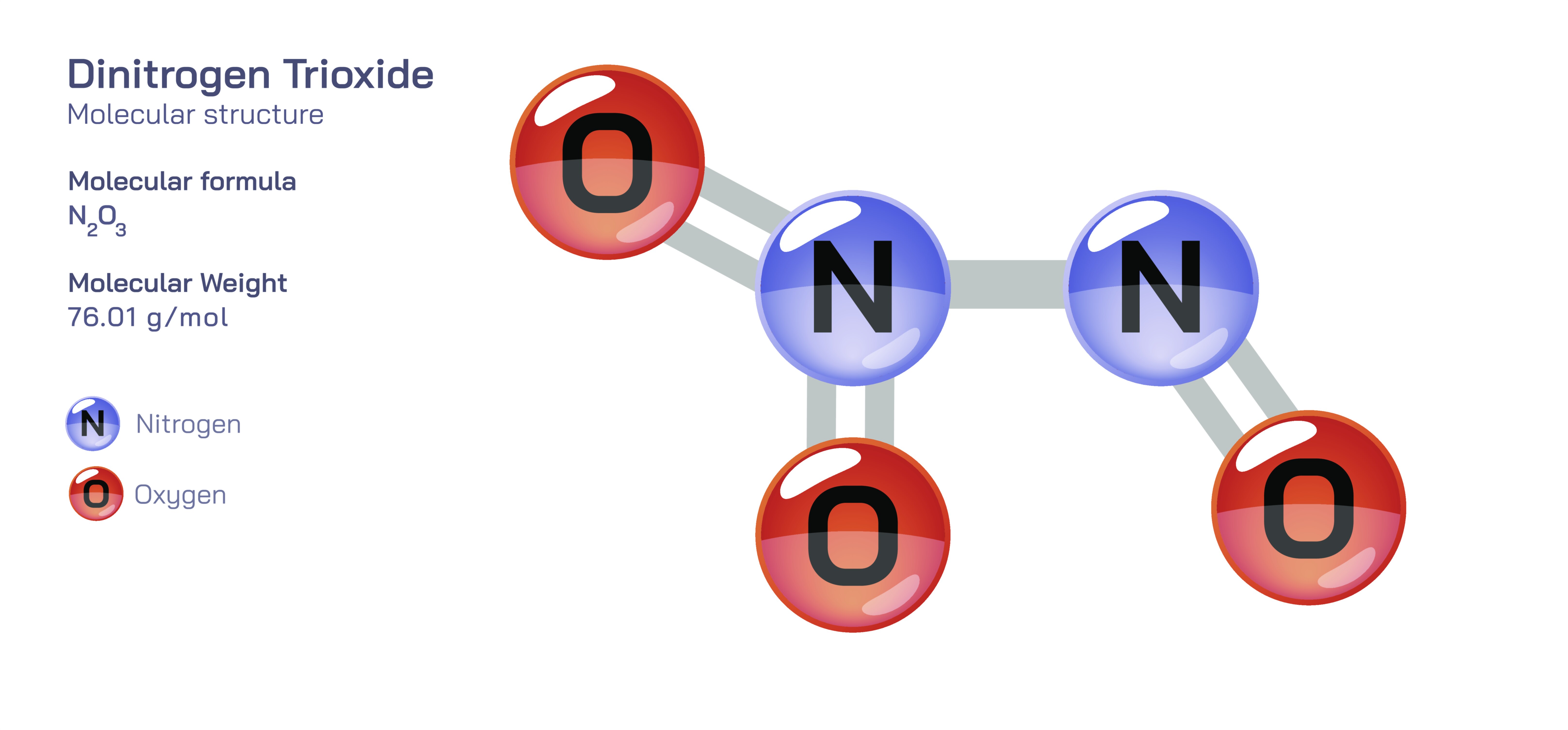
Dinitrogen Trioxide — Nitrogen Oxide Compound
Dinitrogen trioxide is one of the more elusive and unstable members of the nitrogen oxide family, a collection of compounds whose interrelated chemical behaviors shape atmospheric processes, industrial reactions, environmental cycles, and nitrogen chemistry as a whole. Although its formula, N₂O₃, appears simple, the substance’s structural identity, physical behavior, and reactivity illustrate the complexities that arise when atoms of nitrogen and oxygen combine in different ratios and geometries. Dinitrogen trioxide does not exist as a stable, isolable compound under standard atmospheric conditions. Instead, it occupies a transitional role within the deeper network of nitrogen oxide interconversions. In fact, it is best understood not as a conventional molecular species but as a dynamic equilibrium mixture formed from nitric oxide and nitrogen dioxide under cold, dark, or controlled laboratory environments. This fragile identity reflects the broader theme of nitrogen oxide chemistry: small changes in atomic arrangement yield dramatically different properties, lifetimes, and environmental effects.
At low temperatures, typically near or below freezing, dinitrogen trioxide can form a deep blue liquid or solid when nitric oxide and nitrogen dioxide combine in roughly equal proportions. This blue coloration arises from electronic transitions within the molecule, reflecting how the arrangement of nitrogen and oxygen atoms influences how the compound absorbs light. The vivid blue hue distinguishes dinitrogen trioxide from its related nitrogen oxides, which often appear reddish-brown, yellow, or colorless. However, even in this relatively stable low-temperature state, the compound exists in a delicate balance between its constituent nitrogen oxides. Warming the substance or exposing it to light causes it to dissociate rapidly, regenerating nitric oxide and nitrogen dioxide. This instability stems from the inherent strain within its bonding arrangement. The molecule can be viewed conceptually as a combination in which one nitrogen atom bonds to oxygen atoms in a manner that reflects partial oxidation states, with the partner nitrogen atom similarly participating in a structure that teeters between several possible resonance forms. This internal tension makes dinitrogen trioxide prone to decomposition, earning it the characterization of a transient or intermediate species rather than a stable compound.
The behavior of dinitrogen trioxide illustrates a key theme in nitrogen chemistry: oxidation states determine reactivity. Nitric oxide has nitrogen in a relatively low oxidation state, while nitrogen dioxide contains nitrogen in a higher oxidation state. When they combine to form dinitrogen trioxide, the result is a species in which nitrogen atoms effectively adopt intermediate oxidation levels. This middle ground is inherently unstable because the molecule cannot easily settle into one consistent electronic configuration. As a result, dinitrogen trioxide decomposes back into its more stable precursors. This instability also makes the compound highly reactive under appropriate conditions. When exposed to water, it hydrolyzes to form nitrous acid, an important but unstable acid that plays a role in atmospheric chemistry, especially in nighttime or polluted environments. This hydrolysis demonstrates how dinitrogen trioxide links the gas-phase nitrogen oxides with aqueous-phase nitrogen species, facilitating transformations that influence acidity, atmospheric particle formation, and the balance of reactive nitrogen species in natural systems.
In atmospheric chemistry, dinitrogen trioxide contributes to nighttime chemical pathways that differ significantly from daytime nitrogen oxide behavior. During the day, sunlight drives photochemical reactions involving nitrogen dioxide, ozone, and volatile organic compounds. These interactions shape smog formation, ozone dynamics, and pollutant behavior. At night, however, when sunlight no longer provides the energy needed for photolysis, nitrogen dioxide and nitric oxide tend to combine, forming dinitrogen trioxide as a temporary reservoir. Although short-lived, this reservoir helps regulate the concentrations of reactive nitrogen species in the atmosphere. The equilibrium between nitric oxide, nitrogen dioxide, and dinitrogen trioxide acts as a buffer that moderates how rapidly nitrogen oxides participate in further nighttime reactions. Without such intermediates, the delicate balance between ozone formation, nitrogen oxide cycling, and reactive radical species would shift more dramatically between day and night, altering atmospheric stability and pollution patterns. Even though dinitrogen trioxide rarely persists in measurable concentrations for long periods, it plays a role in smoothing the chemical transitions that occur as sunlight fades and returns.
The formation of nitrous acid through the hydrolysis of dinitrogen trioxide has broader implications for atmospheric behavior. Nitrous acid is a key precursor for hydroxyl radicals, which form when the acid is exposed to sunlight. Hydroxyl radicals are often referred to as the “detergents” of the atmosphere because they initiate reactions that degrade pollutants and volatile organic compounds. Thus, the formation and decomposition of dinitrogen trioxide indirectly influence daytime atmospheric cleansing processes, linking nighttime chemistry with daytime environmental behavior. This connection highlights a recurring theme in environmental science: even short-lived, unstable molecules can exert significant influence by serving as bridges between chemical cycles, affecting long-term behavior through transient interactions.
The transient nature of dinitrogen trioxide also makes it important in laboratory research involving nitrogen oxide chemistry. Researchers studying reaction mechanisms often rely on the behavior of intermediates like dinitrogen trioxide to understand how molecules transition between stable states. Even though the compound cannot easily be isolated at room temperature, its formation under controlled conditions helps reveal how electron density shifts between nitrogen and oxygen atoms during chemical reactions. These insights support broader investigations into redox chemistry, energy barriers, reaction kinetics, and the fundamental principles governing molecular stability. In particular, dinitrogen trioxide serves as a model for understanding how molecules with multiple resonance structures behave, how electron delocalization stabilizes or destabilizes compounds, and how chemical environments—such as temperature, light, or solvent—determine whether a molecule persists or dissociates.
In industrial contexts, dinitrogen trioxide itself is not typically used as a reagent because of its instability, but its behavior helps guide processes involving nitrous acid formation, nitrite chemistry, and nitrogen oxide control. Industries concerned with emissions, chemical synthesis, or environmental compliance benefit from understanding how nitrogen oxides interconvert, especially in situations involving low temperatures or moisture. For example, controlling the formation of nitrous acid during flue gas treatment may require knowledge of pathways involving dinitrogen trioxide. Similarly, designing technologies to reduce nitrogen oxide emissions from combustion systems often involves modeling how intermediate species form or decompose under varying temperature and pressure conditions. Dinitrogen trioxide’s formation patterns, although transient, help predict these behaviors and guide mitigation strategies.
In the context of environmental health and pollution control, the hydrolysis products of dinitrogen trioxide contribute to processes that influence air quality and atmospheric acidity. Nitrous acid, produced through this hydrolysis, can deposit onto surfaces or react further to create other nitrogen-containing products. These transformations influence the nitrogen cycle in urban environments, affecting ecosystems, plant growth, and soil chemistry. Understanding how dinitrogen trioxide participates in these pathways helps environmental scientists investigate how pollutants transform as they move through air, water, and surfaces.
Conceptually, dinitrogen trioxide offers a powerful reminder of how chemical species can exist not as fixed entities but as dynamic participants in equilibrium systems. It challenges the notion that molecular identity must be stable to be significant. Instead, the compound demonstrates that even unstable intermediates exert major influence through the reactions they mediate. In this sense, the molecule embodies the principles of transient-state chemistry: that what matters is not the longevity of a species, but the role it plays in connecting one chemical state to another. This concept extends into biochemical systems, catalytic cycles, and environmental pathways, showing how intermediates guide the flow of matter and energy at all scales.
From a structural perspective, dinitrogen trioxide reflects the diverse bonding possibilities within the nitrogen–oxygen system. Its existence as a low-temperature blue liquid formed from the equilibrium of two gases illustrates the continuum between molecular and ionic character, between order and instability. This structural versatility is echoed across the nitrogen oxide family, where nitrogen monoxide, nitrogen dioxide, dinitrogen tetroxide, dinitrogen pentoxide, and others showcase varying degrees of stability, reactivity, and environmental impact. Dinitrogen trioxide sits at a key midpoint in this spectrum, representing the fragile bridge between reduced and oxidized nitrogen species.
Ultimately, dinitrogen trioxide stands not as a stable compound to be isolated and stored, but as an informative and influential participant in the vast network of atmospheric and chemical transformations involving nitrogen. Its fleeting presence belies its importance. Through its formation, decomposition, and interactions with water, it helps regulate nighttime nitrogen chemistry, influences pollutant behavior, and contributes to the formation of reactive species essential for atmospheric cleansing. Its structural complexity reveals how delicate electronic balances shape stability, and its conceptual role illuminates the broader principle that intermediate species—though transient—are essential to the continuity and coherence of chemical systems.