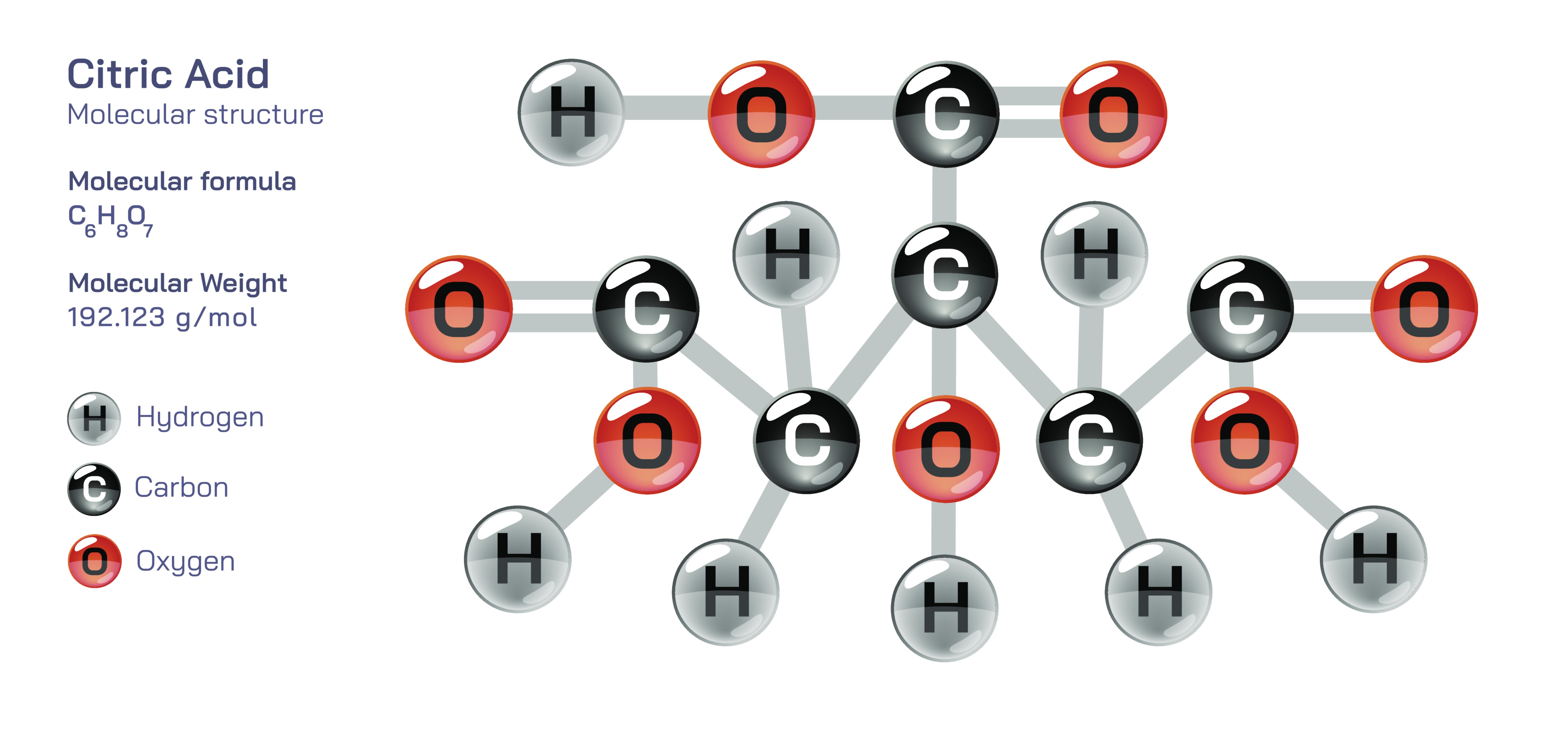
Citric Acid — Organic Tricarboxylic Compound
Citric acid is one of the most important and widely distributed organic tricarboxylic compounds in nature, known for its central role in metabolism, its characteristic sour taste in fruits, its extensive industrial applications, and its deep chemical significance in both biological and environmental systems. As a tricarboxylic acid, it contains three carboxyl groups and one hydroxyl group attached to a six-carbon skeleton, giving it a unique combination of acidity, solubility, reactivity, and complex-forming ability. Its structure, although simple in appearance, is the foundation of several essential biochemical processes, most famously the citric acid cycle—also known as the Krebs cycle or the tricarboxylic acid (TCA) cycle—which powers most aerobic life on Earth. Beyond its biological importance, citric acid is a dominant compound in the food industry, cleaning formulations, pharmaceuticals, metal treatments, cosmetics, and countless commercial products. Understanding citric acid means tracing its journey from fruit-bearing plants to cellular energy pathways, industrial reactors, and human civilization’s daily routines, revealing how deeply this single organic acid is interwoven into natural and technological systems.
Citric acid’s presence in nature is most immediately recognized in citrus fruits such as lemons, limes, oranges, and grapefruits, although it also occurs in berries, pineapples, peaches, and even many vegetables. The sour flavor associated with citrus arises from citric acid’s ability to release hydrogen ions in solution, creating a pleasing acidity that humans have valued for thousands of years. This acidity acts not only as a flavoring agent but also as a natural preservative, inhibiting microbial growth and contributing to the longevity of stored juices, jams, and fermented foods. Historically, citric acid was first isolated from lemon juice in the 18th century, but today it is produced primarily by fermentation using microorganisms such as Aspergillus niger. This method yields a high purity product at industrial scale, making citric acid one of the most widely manufactured organic acids in the world.
The biochemical significance of citric acid cannot be overstated. It is a central intermediate in the TCA cycle, the metabolic pathway through which carbohydrates, fats, and proteins are oxidized to release energy. This cycle occurs in the mitochondria of cells and supports the production of ATP, the energy currency of life. Citric acid forms when acetyl-CoA reacts with oxaloacetate, initiating a sequence of reactions that liberate energy, generate carbon dioxide, and produce reduced cofactors that feed into the electron transport chain. This cycle is fundamental not only to human and animal biology but to plants, fungi, and many microorganisms, highlighting citric acid's universal relevance. Because of its key role in metabolism, citric acid is involved in energy production, biosynthesis, cellular respiration, and the maintenance of metabolic homeostasis. It is a molecule shared across billions of years of evolution, emphasizing its timeless importance in sustaining life.
Chemically, citric acid possesses multiple functional groups that allow it to serve as a versatile reagent and complexing agent. Its three carboxyl groups and one hydroxyl group can bind metal ions, forming stable complexes that are essential in biochemistry, industrial processes, and environmental chemistry. In living organisms, citric acid helps regulate mineral balance by binding calcium, magnesium, and iron, improving their solubility and transport. This chelating ability is harnessed industrially to remove scale, dissolve rust, and control metal ion activity in formulations ranging from household descalers to high-purity laboratory buffers. The ability of citric acid to sequester metal ions also stabilizes foods, preventing discoloration and oxidation that could occur when trace metals catalyze degradation reactions. These combined chemical attributes make citric acid one of the most flexible and valuable compounds in modern industry.
In the food and beverage sector, citric acid serves numerous roles. It provides tartness to soft drinks, candies, beverages, frozen desserts, and condiments; it acts as a preservative in packaged foods; it stabilizes color and flavor; it controls pH; and it acts as an antioxidant. Its role in preventing browning of cut fruits and vegetables is particularly valuable, as it slows the enzymatic oxidation that would otherwise cause discoloration. In dairy products, citric acid enhances emulsification and prevents curdling by binding calcium ions. In baking, it activates leavening reactions when combined with bicarbonate ingredients, creating a controlled release of gas that improves texture. Through these applications, citric acid improves taste, appearance, preservation, and product stability, illustrating how a single compound can influence nearly every dimension of food design.
Outside of food, citric acid is essential in cleaning and household care formulations. Its acidity dissolves limescale, soap scum, and mineral deposits, making it a cornerstone ingredient in descalers, bathroom cleaners, dishwasher additives, and all-purpose cleaning solutions. Unlike stronger mineral acids, citric acid is biodegradable and mild enough for use in domestic and cosmetic settings, yet powerful enough to break down stubborn calcium and iron deposits. Its chelating ability prevents minerals from redepositing on surfaces, improving cleaning efficiency. In laundry detergents, citric acid reduces water hardness, enhances stain removal, and stabilizes enzymes. Its presence in eco-friendly cleaning products reflects modern demand for high-performance chemicals with minimal environmental impact.
Citric acid also plays a vital role in pharmaceuticals and medicine. It is used as an excipient in tablets, syrups, effervescent powders, and injectable formulations. Its acidic nature stabilizes active ingredients, enhances solubility, and adjusts pH for compatibility with biological tissues. In medical diagnostics, citrate salts are used as anticoagulants in blood collection tubes, as they bind calcium and prevent clotting. In kidney stone management, citrate therapy helps reduce calcium crystallization in the urinary system, lowering the risk of stone formation. This therapeutic role highlights citric acid’s importance not only as a chemical ingredient but as a biologically active compound contributing directly to human health.
In cosmetics and personal care, citric acid acts as a pH adjuster, preservative enhancer, and stabilizing agent. Many skincare formulations require precise acidity to maintain product performance, protect microbial safety, and support compatibility with human skin. Citric acid helps achieve these conditions. In shampoos and conditioners, it smooths hair by adjusting pH and improving cuticle alignment. In bath and body products, it contributes to effervescence when combined with bicarbonate salts. These functions make it an essential part of countless grooming and wellness products.
Citric acid is also deeply integrated into industrial processes beyond consumer goods. In metal treatment, it helps remove oxides, passivate stainless steel, and prepare surfaces for welding or coating. In textiles, it adjusts pH during dyeing and improves fabric softness by neutralizing alkaline residues. In environmental engineering, citric acid is used in soil remediation and heavy metal cleanup, where its chelating properties help mobilize contaminants for removal or stabilization. In biotechnology, citric acid functions as a buffering agent, nutrient supplement, and metabolic intermediate in fermentation processes.
Environmental compatibility is another hallmark of citric acid. It is biodegradable, non-persistent, and generally regarded as safe for use in food, pharmaceuticals, and household products. Because it naturally participates in metabolic pathways, it does not accumulate in ecosystems or cause long-term ecological harm when used responsibly. This distinguishes citric acid from many synthetic additives that require extensive regulatory oversight. As industries shift toward greener formulations, citric acid continues to rise in importance as an environmentally sound alternative to harsher chemicals.
The commercial production of citric acid through microbial fermentation is an achievement that reflects the synergy between chemistry and biotechnology. Aspergillus niger converts sugars into citric acid with high yield, using pathways that mimic the metabolic processes of living cells. This biological method ensures sustainable production using renewable raw materials such as molasses, corn syrup, and sugar beets. The success of this industrial fermentation process has made citric acid one of the most readily available organic chemicals, supporting economies worldwide and contributing to the affordability of countless everyday products.
In cultural and culinary traditions, citric acid holds a place of sensory and symbolic importance. It defines the sharp brightness of lemon-based desserts, the refreshing zing in beverages, and the tanginess found in global cuisines. It serves as a natural preservative in traditional pickling techniques and in long-standing food preservation practices that predate modern refrigeration. Its presence in global food culture demonstrates its timeless resonance as both a flavor enhancer and a functional culinary tool.
Ultimately, citric acid stands as a compound that bridges nature, biology, industry, and everyday life. Its structure embodies the elegance of organic chemistry; its role in metabolism embodies the essence of biological energy; its industrial applications embody the ingenuity of human innovation; and its presence in foods and flavors embodies the sensory richness of human culture. Citric acid’s influence extends across scientific disciplines and practical domains, demonstrating how a single molecule—shaped by evolution, harnessed by biotechnology, and integrated into modern systems—can become indispensable to both life and civilization.