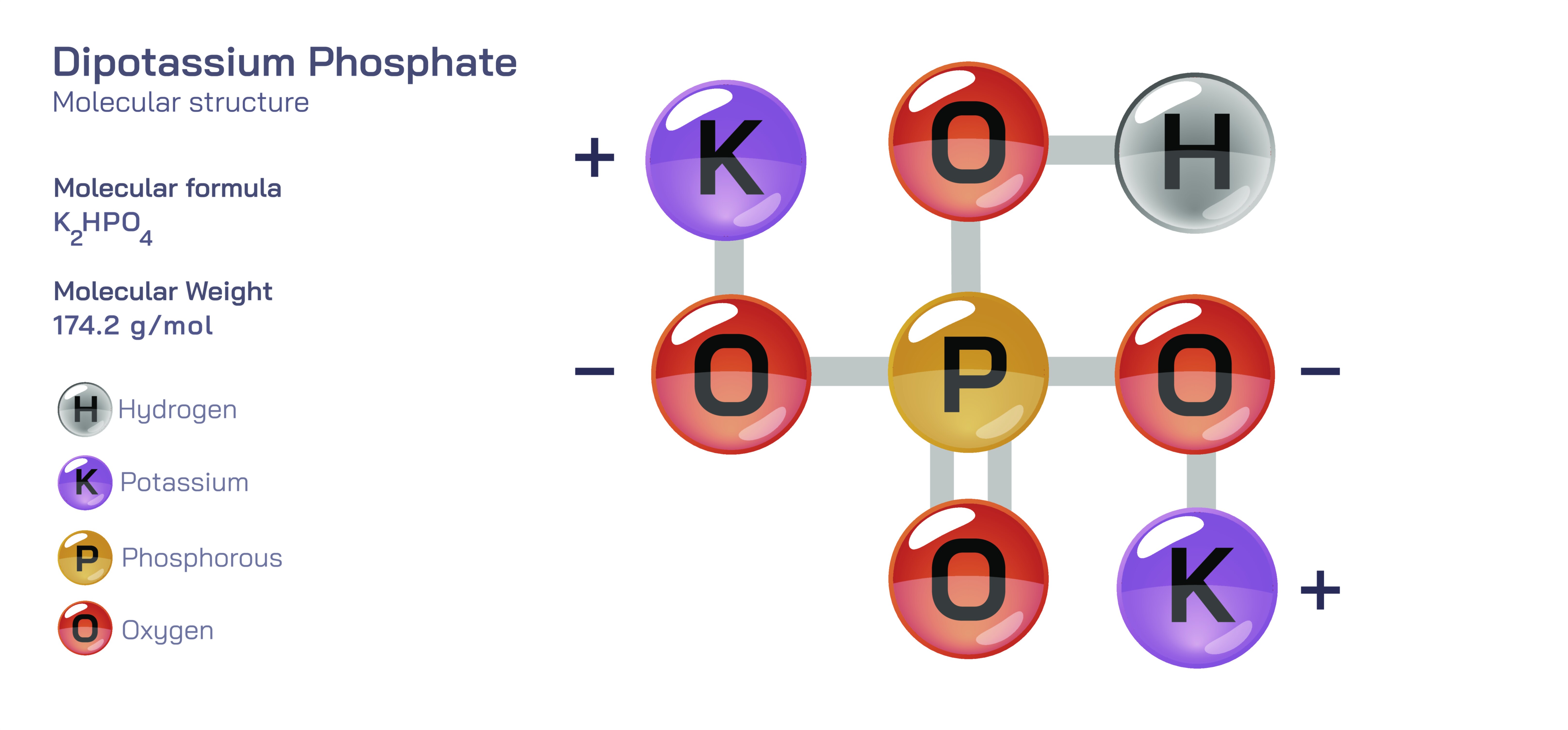
Dipotassium Phosphate — Inorganic Chemical Salt
Dipotassium phosphate is an inorganic chemical salt that plays a remarkably important role across food systems, industrial formulations, agricultural applications, biochemical processes, and numerous technological environments. Although it appears as a simple white crystalline powder with a name that suggests basic mineral chemistry, its structure and behavior reflect the deep principles of ionic bonding, acid–base equilibrium, solubility, buffering capacity, and nutrient availability. Understanding dipotassium phosphate requires looking beyond its appearance and examining the intricate ways in which the salt interacts with water, biological molecules, industrial ingredients, and environmental systems. The compound serves as a stabilizer, nutrient source, pH regulator, emulsification agent, and buffering chemical depending on the context. These varied roles arise from its underlying ionic identity, where potassium ions and hydrogen phosphate ions behave cooperatively to influence chemical environments. By exploring its formation, behavior, and applications, one gains insight into the broader significance of inorganic salts in shaping food science, agriculture, industry, and human physiology.
At its core, dipotassium phosphate emerges from the neutralization of phosphoric acid with potassium-containing bases. It is one of several potassium phosphate salts, distinguished by the number of hydrogen atoms replaced in the phosphate unit. This particular form contains two potassium ions paired with the hydrogen phosphate ion. This configuration has a profound effect on the compound’s chemical behavior. The presence of two potassium ions gives the salt excellent water solubility, allowing it to dissolve rapidly and disperse evenly in aqueous environments. The hydrogen phosphate ion, on the other hand, carries both acidic and basic characteristics, enabling it to shift its protonation state depending on the surrounding pH. This dual chemical nature forms the basis for its buffering capacity. A buffer resists changes in pH when acids or bases are introduced, and dipotassium phosphate can perform this function effectively because the phosphate ion system naturally maintains stability across a moderate pH range. This feature makes it highly valuable in food processing, laboratory environments, and biochemical systems where precise pH control is essential.
When dissolved in water, dipotassium phosphate dissociates completely into its constituent ions. The potassium ions provide nutritional value as an essential electrolyte used by organisms to regulate nerve conduction, muscle contraction, water balance, and enzymatic function. Potassium plays a critical role in maintaining cell membrane potential and supporting biochemical transport processes. The hydrogen phosphate ion, in contrast, serves as an integral part of energy production, as phosphate groups form the backbone of ATP and DNA. In this way, the structure of dipotassium phosphate reflects not only chemical stability but biological relevance. The compound is often used as a food additive to provide these essential nutrients in easily absorbed form. Because of its solubility and neutral taste, it distributes evenly without altering the sensory qualities of foods, making it ideal for fortifying beverages, dairy products, electrolyte drinks, and nutritional supplements.
In food science, dipotassium phosphate functions as a stabilizer, emulsifier, and texture modifier. It is frequently added to dairy products such as coffee creamers to prevent curdling, an effect caused when acidic conditions destabilize milk proteins. The buffering ability of the phosphate component helps maintain an environment that keeps proteins dispersed rather than clumped. This stabilization is essential for products subjected to thermal processing or exposure to acidic beverages. In non-dairy creamers, powdered drinks, and processed cheeses, the compound also interacts with proteins to create smooth textures and uniform consistency. Its ionic nature influences how fats, proteins, and water interact, supporting emulsification processes that keep ingredients from separating. In beverages, dipotassium phosphate acts as an electrolyte, helping formulate sports drinks that replace minerals lost through sweat. Its potassium content makes it preferable to sodium-containing salts for individuals seeking low-sodium formulations.
Outside the realm of food, dipotassium phosphate plays important roles in pharmaceuticals and laboratory systems. In medical formulations, it is used to adjust acidity, stabilize active ingredients, and ensure proper dissolution rates. In intravenous solutions, it provides a source of phosphate and potassium for patients with deficiencies or metabolic imbalances. In laboratory buffers, particularly those used in cell culture, chromatography, or biochemical assays, the compound helps maintain pH stability essential for reproducible results. Many biological reactions require narrow pH conditions to ensure protein stability or enzymatic function, and dipotassium phosphate provides the stability needed for complex systems to behave predictably. Because phosphate ions participate in energy transfer and metabolic cycles, their presence can support cellular health in controlled environments.
The compound also finds significant application in industrial processes. It is used in corrosion control, where phosphate ions help form protective films on metal surfaces that inhibit oxidation. In manufacturing detergents and cleaning products, dipotassium phosphate improves cleaning efficiency by softening water, stabilizing formulations, and interacting with soil particles. In fertilizers, it provides plants with both potassium and phosphorus, two of the most important nutrients required for growth. Potassium helps regulate water balance and stress tolerance in plants, while phosphorus supports root development, flowering, energy transfer, and genetic processes. Its high solubility ensures rapid nutrient availability, making it valuable in hydroponic systems, foliar applications, and soil-based agriculture. Because of its balanced nutrient profile, it is often blended with other fertilizers to create formulations targeted at specific crop needs or soil conditions.
Environmental science also intersects with dipotassium phosphate in important ways. Phosphate-containing compounds must be used with care because excessive runoff can contribute to eutrophication—an over-enrichment of water bodies that stimulates algal growth. This environmental challenge highlights how essential nutrients become pollutants when introduced in excessive amounts. Understanding dipotassium phosphate thus requires awareness of nutrient cycles and ecological balance. Its behavior in soil and water demonstrates how inorganic salts influence biological systems, from plant growth to aquatic ecosystems. Responsible management ensures that its benefits to agriculture and industry do not lead to unintended environmental consequences.
Dipotassium phosphate’s chemical stability contributes to its longevity in storage and versatility in formulations. It resists degradation under normal conditions, remaining effective even when exposed to heat or varying humidity. This stability makes it reliable in powdered mixes, pharmaceutical tablets, and laboratory reagents. When incorporated into products, it interacts predictably with other ingredients, simplifying formulation design. The ionic nature of the compound also influences how it behaves in mixtures, where it can adjust osmotic balance, electrolyte strength, and molecular interactions. These features reflect the broader principle that inorganic salts, despite their apparent simplicity, exert profound influence on the physical and chemical properties of complex systems.
In more specialized scientific contexts, dipotassium phosphate plays a role in energy research, biochemistry, and molecular biology. Phosphate groups are central to phosphorylation reactions within cells, processes that regulate enzyme activity, signal pathways, and metabolic cycles. Although dipotassium phosphate does not directly participate in these reactions the way organic phosphates do, its dissociation into hydrogen phosphate contributes indirectly by stabilizing environments where these pathways occur. Buffer systems containing phosphate ions form part of the foundation for biochemical experimentation, from DNA analysis to protein purification. The predictable behavior of phosphate buffers supports experiments requiring consistency across repeated procedures. This makes dipotassium phosphate a key ingredient in many foundational reagents used in academic and industrial research laboratories worldwide.
On the physiological level, the compound is associated with two essential nutrients: potassium and phosphorus. Potassium supports heart function, nerve transmission, fluid regulation, and muscle contraction. Phosphorus is essential for bone health, cellular structure, and energy storage molecules such as ATP. When used as a supplement or ingredient, dipotassium phosphate delivers these nutrients in a form that dissolves easily and is absorbed efficiently. This feature underscores the interplay between chemistry and biology, where the form of a nutrient influences how the body receives and uses it. The balance between potassium intake and other electrolytes is especially important in individuals with kidney conditions or metabolic disorders, making the regulation of compounds like dipotassium phosphate essential in medical and nutritional contexts.
Despite its stability, the compound reflects the dynamic nature of chemical systems. In solution, its ions participate in subtle interactions with other components, influencing reactions, solubility, protein structure, and even sensory properties in food. The phosphate ion system, consisting of multiple protonation states, is one of the most versatile buffering systems in chemistry. Dipotassium phosphate contributes to this system by occupying a middle point between more acidic and more basic phosphate salts. This intermediate behavior enables it to shift proton balance and maintain environments required for sensitive processes, whether in living cells, cooking sauces, or laboratory assays. Its effectiveness in such diverse environments shows how the structure of a chemical salt plays a foundational role in shaping both natural and engineered systems.
Ultimately, dipotassium phosphate stands as a testament to the importance of inorganic salts in modern life. It links agricultural productivity, food stability, industrial processes, laboratory precision, and physiological function through a single chemical identity. Its influence stems from the interaction of its ions with water, biological molecules, and complex mixtures. Understanding dipotassium phosphate reveals how fundamental chemical principles—ionic bonding, solubility, acid–base equilibrium, buffering, and nutrient transport—appear consistently across the systems that sustain society. From a simple crystalline powder emerges an essential contributor to nutrition, chemistry, industry, and environmental balance. Through this compound, one can appreciate the profound role that inorganic substances play in shaping technology, biology, and daily life, demonstrating that even the most unassuming materials have far-reaching significance when examined through the lens of molecular behavior.