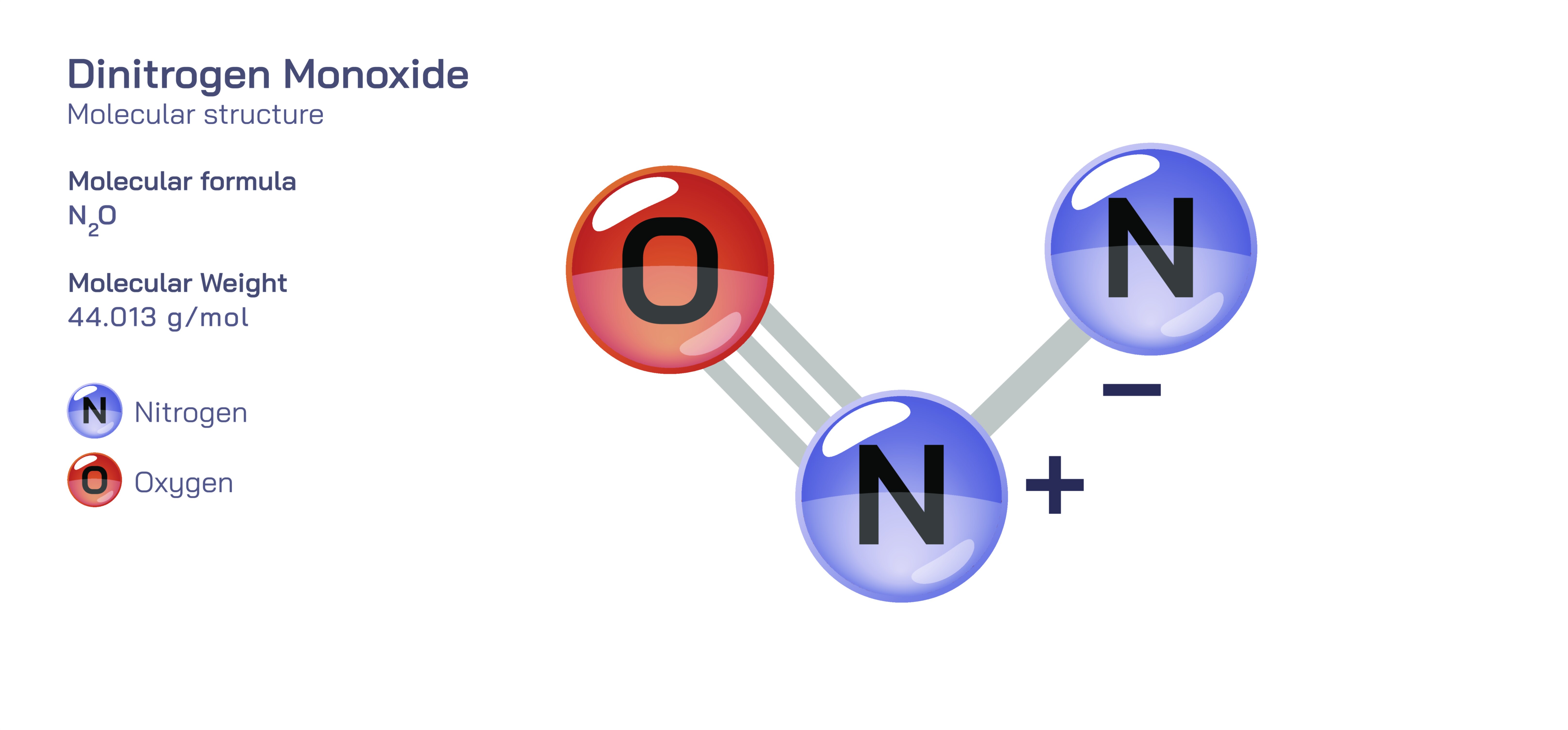
Dinitrogen Monoxide — Nitrous Oxide Gas Molecular Structure
Dinitrogen monoxide, more commonly known as nitrous oxide or laughing gas, is a chemically intriguing molecule whose structure, behavior, and wide range of effects make it one of the most fascinating substances in both natural and applied chemistry. Despite its simple formula, consisting of two nitrogen atoms and one oxygen atom, nitrous oxide possesses a combination of physical, chemical, physiological, and environmental characteristics that arise directly from the way its atoms are arranged and bonded. Its molecular identity governs everything from its mild anesthetic effects in medicine to its role in atmospheric chemistry, its reactivity patterns in combustion systems, and its unexpected environmental impact as one of the most potent greenhouse gases. Understanding nitrous oxide therefore requires a deep appreciation of how molecular structure determines chemical behavior and how that behavior cascades into large-scale effects in biological, industrial, and ecological systems.
The molecular structure of dinitrogen monoxide may appear straightforward, but its bonding arrangement is more nuanced than the simplicity of its formula suggests. The molecule exists as a linear chain of atoms arranged in sequence: nitrogen–nitrogen–oxygen. Although this alignment visually resembles the much more reactive nitrogen dioxide, nitrous oxide is far more stable because of the nature of the bonding between its atoms. The first nitrogen atom is more strongly bonded to the central nitrogen than the central nitrogen is to the terminal oxygen, creating an uneven distribution of electron density throughout the molecule. This asymmetry produces a small but meaningful polarity, with the oxygen end being slightly negative and the nitrogen end slightly positive. Because the molecule is linear, these charges are distributed along its length rather than creating bent-geometry polarity. The resulting structure allows nitrous oxide to dissolve moderately well in lipids, enabling it to interact with biological membranes and nervous system components when used medically. At the same time, this linear geometry distinguishes nitrous oxide from other nitrogen oxides that are far more reactive or toxic. Its structure positions it in a unique chemical category: stable enough to be stored and handled safely under controlled conditions but reactive enough to participate in specialized reactions when activated by heat, catalysts, or biological processes.
One of the most famous aspects of nitrous oxide is its role as an anesthetic and analgesic in medical and dental procedures. Because of its molecular structure, nitrous oxide can dissolve into the lipid components of nerve cell membranes, where it subtly alters the electrical activity of neurons. Although the precise biochemical pathways responsible for its effects remain an area of ongoing research, its influence on neurotransmitter release and neuronal firing thresholds underlies both its pain-reducing and euphoric properties. This interaction with neuronal membranes illustrates how molecular structure, polarity, and solubility combine to produce profound physiological effects. Nitrous oxide does not act in the same way as more potent anesthetics; instead, its modest molecular influence produces a mild dissociation from pain along with a sense of relaxation. Because it does not strongly depress nervous system function, it is safe to use in controlled concentrations, and it is rapidly expelled from the body once inhalation stops. These traits make nitrous oxide especially useful in dentistry, childbirth, minor surgical procedures, and emergency pain management. The molecule’s small size, moderate polarity, and ability to diffuse quickly through tissues all contribute to its rapid onset and rapid elimination, turning a simple molecular structure into a medically indispensable tool.
Beyond its medical role, nitrous oxide occupies an important place in industrial chemistry and engineering. In combustion systems, particularly those used in motorsports and aerospace research, nitrous oxide acts as a source of additional oxygen when heated or decomposed. When the molecule is subjected to high temperatures, the weaker N–O bond breaks, releasing oxygen that supports more intense combustion. This decomposition liberates oxygen in a controlled way, allowing engines to burn more fuel and generate more power. The role of nitrous oxide in such systems depends directly on the nature of its molecular structure: a combination of stability at moderate temperatures and decomposability at high temperatures. Unlike gaseous oxygen, which is highly reactive at all temperatures, nitrous oxide remains safe to store in pressurized tanks under normal conditions because its bonds require significant energy to break. This unique combination of stability and reactivity makes nitrous oxide a powerful oxidizer in specialized combustion settings, demonstrating how atomic arrangement influences mechanical performance in engineered systems.
Nitrous oxide also plays a significant role in atmospheric and environmental chemistry, where its molecular identity gives rise to effects that extend across global scales. Produced by natural processes in soil and water and by human-driven agricultural activities, nitrous oxide is a long-lived gas that accumulates in the atmosphere. Because the molecule is linear and only weakly polar, it does not react readily with many atmospheric species, allowing it to persist for more than a century before breaking down. Once nitrous oxide reaches the upper atmosphere, however, ultraviolet radiation provides enough energy to split the N–O bond, liberating nitrogen and oxygen species that participate in reactions influencing the ozone layer. Some of these nitrogen-containing fragments accelerate the destruction of ozone, contributing to subtle but meaningful changes in the structure of the stratosphere. At lower altitudes, nitrous oxide acts as a greenhouse gas. Its molecular vibrations—stretches and bends that absorb and re-emit infrared radiation—make it one of the most potent heat-trapping gases known. On a molecule-for-molecule basis, nitrous oxide has a far greater warming influence than carbon dioxide, even though its overall atmospheric concentration is smaller. The environmental effects of nitrous oxide therefore highlight how a seemingly simple molecule can shape global systems through vibrational behavior rooted in its molecular structure.
In biological and ecological contexts, the molecule plays surprising roles as well. Microorganisms in soil and sediments produce nitrous oxide as part of the nitrogen cycle. Specifically, nitrifying and denitrifying bacteria generate nitrous oxide as intermediate or byproduct during the conversion of nitrogen-containing species. These microbial processes rely on the ability of the nitrogen atoms within nitrous oxide to accept or donate electrons in enzymatic reactions. The success of these transformations derives from the unusual distribution of electron density within the molecule, where the terminal oxygen participates in redox reactions that bridge inorganic nitrogen species. This connection between molecular structure and biological function illustrates how nitrous oxide fits into Earth’s biochemical flow of nitrogen, a cycle essential for sustaining plant growth, soil fertility, and ecological balance. Human activities, particularly fertilizer use in agriculture, dramatically amplify the natural production of nitrous oxide, highlighting how disrupting microbial equilibria can alter atmospheric chemistry and climate.
Nitrous oxide’s reactivity profile also reflects the influence of its structure. Although far less reactive than nitrogen dioxide or nitric oxide, nitrous oxide participates in reduction and decomposition reactions under the right conditions. Because of its asymmetrical electron distribution, it can act as a mild oxidizer or serve as a nitrogen donor in certain specialized chemical processes. Its behavior in catalytic systems offers insight into activation energies and molecular interactions. For instance, metal surfaces can weaken the N–O bond, allowing the molecule to dissociate at lower temperatures. These surface-mediated reactions depend on how the molecule interacts with electron clouds on metal atoms, which in turn is governed by the arrangement of electrons within the nitrous oxide molecule itself. This interplay between molecular structure and catalytic activity forms the basis for studying its decomposition, oxidation capacity, and applications in green chemistry and energy research.
The sensory and physiological effects of nitrous oxide also stem from its molecular characteristics. When inhaled, nitrous oxide diffuses rapidly from the lungs into the bloodstream, crossing cell membranes with relative ease due to its small size and modest solubility in lipids. The molecule then disperses quickly throughout the body, affecting the central nervous system before being exhaled unchanged. Unlike chemical agents that undergo extensive biotransformation, nitrous oxide remains structurally intact as it cycles through the body. This metabolic inertness helps prevent long-term accumulation but also means that high or prolonged exposures can interfere with the body’s biochemistry in subtler ways. For example, nitrous oxide reacts with vitamin B₁₂-dependent enzymes, impairing their function in rare cases of chronic or excessive use. This reaction reflects the molecule’s ability to disrupt subtle nitrogen–metal interactions in biological systems, demonstrating again how structural features influence physiological impact.
In environmental monitoring, nitrous oxide serves an important role as a tracer gas because of its relatively stable structure and predictable behavior in the atmosphere. Scientists use it to study airflow patterns, ocean–atmosphere interactions, greenhouse gas transport, and long-term climate trends. Its presence in ice cores provides historical records of past environmental conditions, revealing how agricultural practices, industrial development, and climate change influence the balance of atmospheric gases. The molecular stability of nitrous oxide, derived from the strength of its internal nitrogen bonding, enables these long-term measurements and makes the gas a reliable indicator for studying ecological and climatic systems across centuries.
Even in material science and engineering, nitrous oxide’s behavior finds application. Controlled thermal decomposition of nitrous oxide allows researchers to generate reactive oxygen species in specialized environments, supporting processes such as advanced oxidation, catalytic testing, and surface modification. In semiconductor manufacturing, nitrous oxide has been used as a precursor for creating thin oxide layers under carefully regulated conditions. These uses depend on the molecule’s predictable decomposition pathways, which trace back to the stability of its nitrogen–oxygen bond and the ease with which that bond can be broken by heat or light.
The cultural and historical significance of nitrous oxide also illustrates how a molecule’s structural features shape human experience beyond the laboratory or atmosphere. When first discovered, nitrous oxide gained rapid popularity in public demonstrations because of its euphoric effects. These early entertainments, sometimes called “laughing gas shows,” represented one of the first public intersections of chemistry and sensation. Behind these experiences was a molecular mechanism: the ability of nitrous oxide to influence neural signaling through its structural interactions with lipid membranes. As understanding deepened, these sensational displays gave way to more formal medical uses, stabilizing its place in dentistry, surgery, and pain management. The evolution of nitrous oxide’s use—from entertainment to medicine to environmental concern—reflects how molecules with simple atomic compositions can produce profound effects when their structure interacts with human perception and technological development.
Ultimately, the story of dinitrogen monoxide is one of structural simplicity giving rise to multifaceted influence. From its linear arrangement of atoms come anesthetic properties that reduce human suffering, combustion-enhancing traits that power engines, greenhouse effects that shape climate change, and reactive patterns that guide atmospheric chemistry. Its molecular identity determines how it lives in the body, how it interacts with ecosystems, how it circulates in the atmosphere, and how it can be harnessed or mitigated by science and technology. Through nitrous oxide, one can see clearly the deep principle that defines chemical science: structure dictates behavior, behavior shapes interaction, and interaction influences both natural systems and human applications. The molecule’s gentle anesthetic touch, dramatic environmental significance, and rich chemical personality all stem from the same underlying architecture of nitrogen and oxygen atoms. In this way, dinitrogen monoxide becomes a powerful reminder of how even the simplest arrangements of matter can exert extraordinary effects, weaving through medicine, industry, climate, and biology with subtle yet far-reaching consequences.