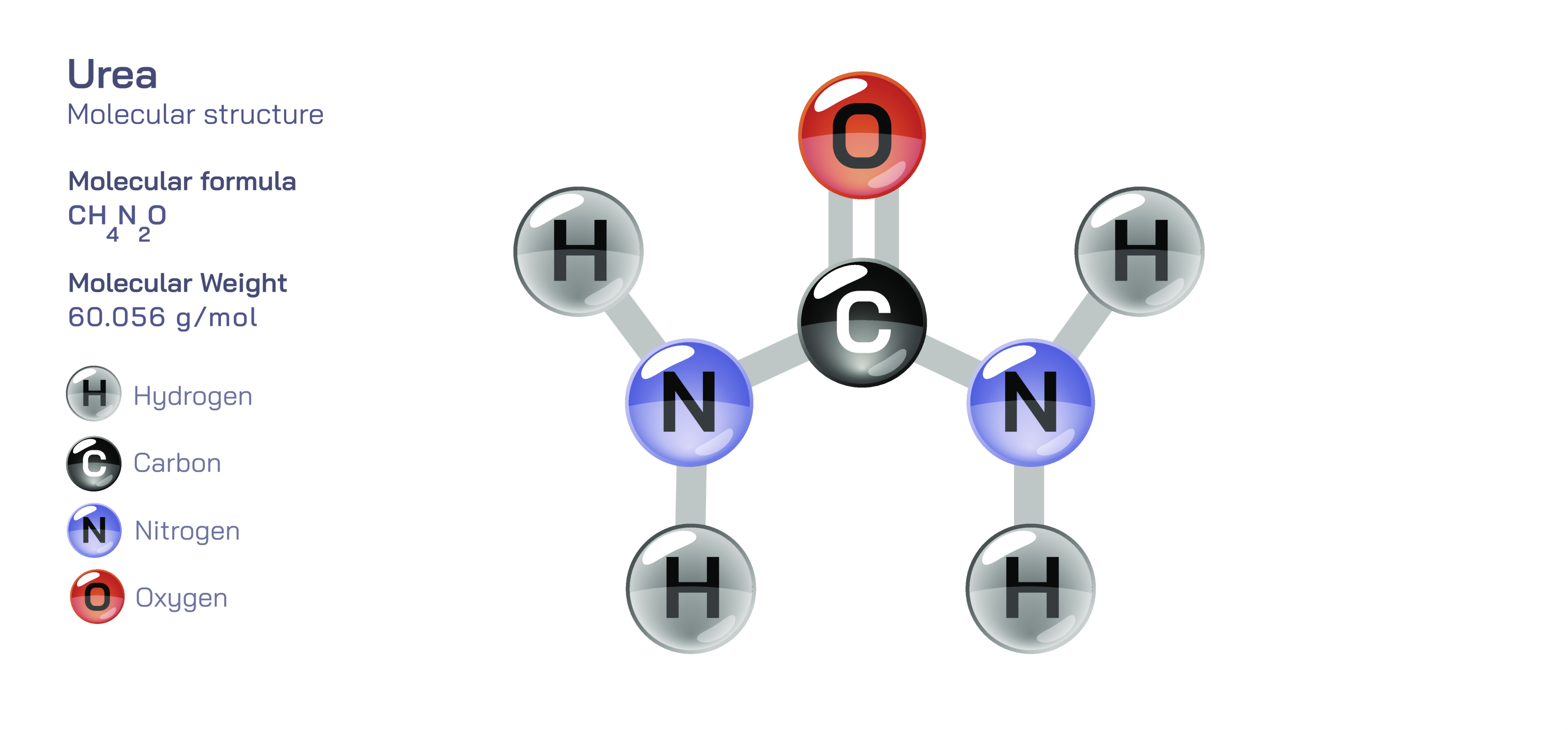
Urea — Organic Nitrogen Compound
Urea is one of the most important organic nitrogen compounds in biological systems, industry, agriculture, and environmental chemistry, and its significance stretches across medicine, physiology, biochemistry, and global food production. It is a simple molecule in structure yet profoundly influential in function, acting as a key product of protein metabolism in humans and other animals while also serving as a vital nitrogen-rich fertilizer that supports modern agriculture. Understanding urea requires a close look at how the body manages nitrogen waste, how metabolic cycles maintain internal chemical balance, how industries synthesize and use nitrogen compounds on a massive scale, and how this substance bridges the worlds of living organisms and chemical technology. Its simplicity conceals a complex role in maintaining health, facilitating global food supply, supporting chemical manufacturing, and illuminating core processes in biology.
In human physiology, urea represents the end product of protein and amino acid breakdown. When proteins are metabolized for energy or structural turnover, they release nitrogen-containing groups that must be safely managed to prevent toxic accumulation. Ammonia is the initial nitrogen waste product, but ammonia is highly toxic to cells and cannot accumulate in the bloodstream. The liver converts ammonia into urea through a sophisticated biochemical sequence known as the urea cycle. Within liver cells, nitrogen-containing compounds enter this cycle and are transformed into urea, a far less toxic and highly water-soluble molecule that can travel safely through the bloodstream. Once formed, urea circulates to the kidneys, where it is filtered out of the blood and excreted through urine. This process is central to maintaining nitrogen balance, ensuring the safe disposal of metabolic waste, and supporting the stability of bodily chemistry. Without urea formation, ammonia levels would rise rapidly, disrupting cellular function, affecting neurological activity, and threatening survival. Thus, urea is not merely a waste product but a carefully regulated output of biochemical processes that maintain life.
The kidneys play a critical role in managing urea levels within the body. As blood passes through the kidney’s filtration structures, urea is selectively removed and directed into urine. Some urea is reabsorbed into the bloodstream to support a mechanism known as the renal concentrating system, which helps the body retain water when hydration levels are low. Urea contributes to establishing osmotic gradients in the kidney’s medulla, enabling the formation of concentrated urine. This interplay demonstrates how urea participates not only in waste elimination but also in fluid balance. Elevated blood urea levels, often measured as blood urea nitrogen (BUN), can indicate decreased kidney function, dehydration, high-protein diets, or metabolic dysfunctions. Conversely, abnormally low levels may result from liver disease or malnutrition, highlighting the relationship between urea metabolism, organ health, and dietary intake. Clinicians often rely on BUN measurements to evaluate kidney health and metabolic status, underscoring how central urea is to diagnostic medicine.
Beyond its biological importance, urea carries immense significance in the agricultural sector as one of the most widely used nitrogen fertilizers worldwide. Because plants require nitrogen to synthesize amino acids, proteins, chlorophyll, and nucleic acids, urea serves as an efficient, concentrated source of nitrogen that supports plant growth and agricultural productivity. When applied to soil, urea undergoes enzymatic hydrolysis, primarily through the action of urease, transforming it into ammonia and then into ammonium or nitrate, forms that plants can absorb. This transformation makes urea a key driver of photosynthesis, protein formation, and crop yields. Global food production depends heavily on synthetic fertilizers, many of which rely on urea or urea-based formulations. The production of urea has therefore become a foundational element of modern agriculture, supporting food security for billions of people. However, this usefulness comes with environmental considerations. Excessive or improper application can lead to nitrogen runoff, water pollution, soil acidification, and greenhouse gas emissions. Managing urea in farming practices requires careful timing, controlled application, and sometimes the use of inhibitors that slow its breakdown and reduce environmental impact. This balance between agricultural necessity and ecological responsibility illustrates the complex role urea plays in sustaining both human populations and environmental health.
Urea also serves an important role in industrial chemistry. It is synthesized on a massive scale through the reaction of ammonia and carbon dioxide under high pressure and temperature. This industrial process, tied historically to the Haber–Bosch method of ammonia production, illustrates how synthetic chemistry can mirror biological pathways while operating on vastly larger scales. The ability to produce urea cheaply and efficiently revolutionized agriculture and chemical manufacturing, supporting industries such as plastics, resins, adhesives, automotive components, pharmaceuticals, and household products. Melamine, for example, a key ingredient in laminates and durable plastics, is manufactured using urea. Urea is also incorporated into resins used in plywood, particleboard, and coatings. These industrial uses demonstrate how a molecule naturally produced in the human body becomes a versatile ingredient in technology and manufacturing.
In consumer and cosmetic applications, urea is valued for its hydrating and keratolytic properties. Skincare formulations incorporate urea to moisturize dry skin, soften rough areas, and support the natural shedding of dead skin cells. At low concentrations, urea functions primarily as a humectant, attracting and retaining moisture in the skin. At higher concentrations, it helps loosen and exfoliate hardened or thickened skin, making it useful in treatments for calluses, psoriasis, eczema, and keratosis. Urea also enhances the penetration of topical medications, increasing their effectiveness. In shampoos and hair conditioners, it helps maintain moisture balance, soothe irritations, and enhance softness. These dermatological roles show how a molecule associated with metabolic waste serves gentle and therapeutic functions when incorporated into topical applications.
In veterinary medicine, urea aids in feeding strategies for ruminants such as cattle. Because the microorganisms in their stomachs can use urea as a nitrogen source to synthesize proteins, controlled urea supplementation supports growth and productivity. However, excessive intake can be toxic, demonstrating again how dosage and context determine whether urea acts as a beneficial nutrient or a harmful compound.
Urea’s involvement in laboratory science is equally important. As a protein denaturant, it allows researchers to break hydrogen bonds within proteins, unfolding them for analysis and experimentation. This capability supports research in molecular biology, protein chemistry, and enzymology. In medical laboratories, urea participates in diagnostic reagents and testing kits. Its chemical behavior, solubility, and reactivity make it a valuable tool for studying biochemical processes, analyzing enzymes, and exploring protein structure. These laboratory applications highlight urea’s role not only in living systems but also in the scientific investigations that help decipher biological complexity.
Environmental processes also reflect urea’s influence. When released into ecosystems through waste, runoff, or natural decomposition, urea serves as a nitrogen source for plant and microbial growth. Its presence in soils and water bodies influences nutrient cycles, microbial communities, and ecological balance. In wastewater treatment, urea undergoes breakdown that contributes to nitrogen loads, prompting treatment strategies that manage nitrification and denitrification. Understanding these environmental pathways is essential for balancing agricultural productivity with ecosystem protection.
Urea has historical significance as well. Its synthesis in the early nineteenth century marked a turning point in the development of organic chemistry. When Friedrich Wöhler synthesized urea from inorganic starting materials, he demonstrated that organic compounds could be created without biological processes. This groundbreaking discovery helped dismantle the theory of vitalism, which held that organic substances could only be produced by living organisms. The synthesis of urea paved the way for modern organic chemistry, biotechnology, and synthetic biology, illustrating how a single molecule can reshape scientific understanding.
In clinical practice, disorders related to urea metabolism highlight its importance. Conditions such as urea cycle disorders impair the body’s ability to safely eliminate nitrogen waste, leading to toxic ammonia buildup, neurological symptoms, and metabolic crises. Kidney dysfunction increases blood urea levels, signaling reduced filtration efficiency. Dehydration elevates urea concentration, while liver disease lowers it by impairing synthesis. These clinical patterns make urea a crucial biomarker for assessing metabolic and organ health, guiding diagnosis and treatment decisions.
Ultimately, urea is far more than a simple nitrogen compound. It is a centerpiece of metabolic safety, a cornerstone of agricultural productivity, a foundation of chemical industries, a tool in scientific research, a therapeutic agent in dermatology, and a molecule deeply interwoven into environmental cycles. From its role in excreting nitrogenous waste to its influence on global food systems, from its historical importance in scientific discovery to its everyday presence in skincare products, urea demonstrates how profoundly interconnected chemistry, biology, industry, and ecology truly are. Through its simplicity and versatility, urea reflects the unity of natural and technological worlds, revealing how a molecule originating from metabolic necessity can sustain life, support civilization, and inspire scientific innovation.