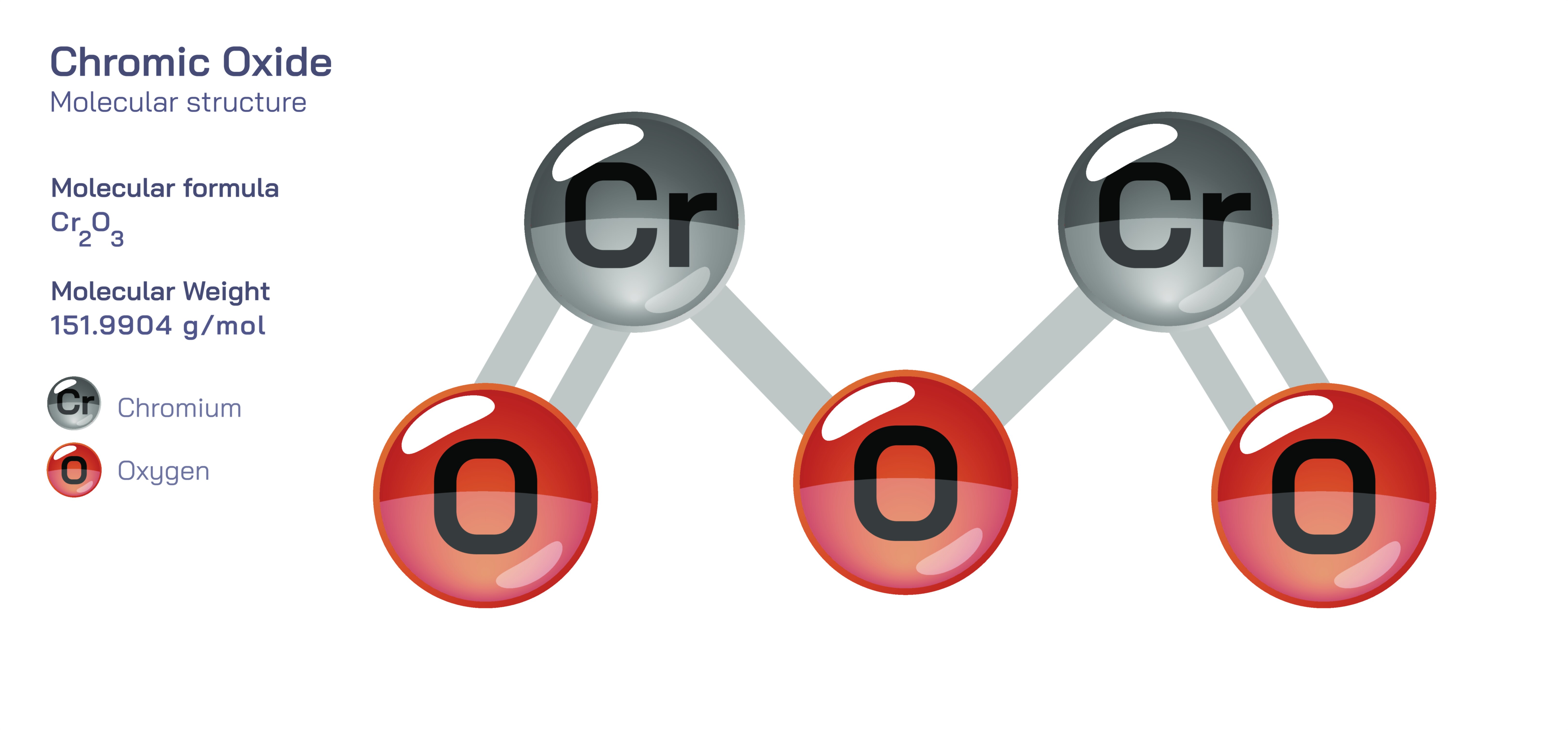
Chromic Oxide — Inorganic Pigment Compound
Chromic oxide is one of the most enduring, stable, and culturally significant inorganic pigment compounds known to science, celebrated for its characteristic deep green color, remarkable thermal and chemical stability, and wide-ranging usefulness across art, industry, ceramics, metallurgy, coatings, refractories, and material engineering. As an oxide of trivalent chromium with the formula Cr₂O₃, chromic oxide possesses a crystalline structure and a robustness that allows it to withstand extreme temperatures, resist corrosion, endure abrasive environments, and maintain its vivid coloration under conditions that would degrade or discolor many other pigments. Its story is woven into centuries of artistic creation, technological development, and scientific exploration. Understanding chromic oxide requires examining its structural characteristics, the origins of its intense color, its physical resilience, its diverse applications, and the ways in which this material has shaped industries while maintaining a reputation as one of the safest and most reliable chromium-based compounds. Its chemically inert nature and relative biological compatibility stand in stark contrast to the hazards associated with many hexavalent chromium species, making chromic oxide a rare example of a chromium compound valued for both its stability and its practicality.
At the atomic level, chromic oxide’s properties originate from chromium in its +3 oxidation state, combined with oxygen atoms arranged in a compact and stable lattice. This structure forms a dense, tightly bonded material that is difficult to oxidize further and even more difficult to reduce under normal conditions. As a result, chromic oxide behaves as one of the most chemically inert oxide pigments in the inorganic palette. The green color that makes chromic oxide so iconic arises from electronic transitions involving chromium(III) ions in the oxide lattice. These transitions absorb light in specific regions of the visible spectrum, particularly red wavelengths, which in turn produce the deep, earthy green that artists and manufacturers have prized for centuries. The stability of these electronic arrangements means that the pigment does not fade when exposed to sunlight, heat, moisture, or industrial chemicals—traits that make it invaluable in applications where durability is essential.
Historically, chromic oxide gained prominence in the nineteenth century as a superior alternative to earlier green pigments, many of which were either toxic, unstable, or both. Prior to chromic oxide’s widespread adoption, green pigments such as verdigris, malachite, and Scheele’s green were used in paints and artistic works, but they suffered from poor stability or alarming toxicity due to arsenic content. When synthetic chromic oxide became commercially available, it quickly replaced these earlier materials in many artistic and industrial contexts. Artists valued it for its opacity, richness, and resilience, while manufacturers appreciated its ability to impart long-lasting color to ceramics, glass, roofing materials, plastics, and even high-performance coatings. Chromic oxide’s resistance to fading or weathering allowed artworks and architectural decorations to retain their color fidelity for generations, shaping the visual identity of countless products and artistic movements.
In ceramics and glassmaking, chromic oxide plays a central role as a coloring agent capable of producing stable green hues that endure even when subjected to high firing temperatures. Ceramic glazes incorporating chromic oxide retain their color across a range of kiln atmospheres, whether oxidizing or reducing, although the final shade can vary depending on the presence of other metal ions and fluxing agents. In some cases, chromic oxide participates in complex interactions that yield deep green, blackish-green, or olive tones, offering ceramicists a diverse palette of stable hues. In glass formulations, chromic oxide is used to create green bottles, tinted architectural glass, and specialized materials such as UV-protective glass where chromium ions contribute both color and functional optical properties. The pigment’s ability to withstand extreme thermal conditions highlights its loyalty to form and function, even when subjected to the harshest manufacturing environments.
Industrial coatings represent another major domain in which chromic oxide’s unique properties shine. Because it resists UV degradation, chemical attack, and mechanical wear, chromic oxide is used in coatings designed for machinery, vehicles, marine structures, and industrial facilities. Protective paints enriched with chromic oxide maintain their color and integrity when exposed to sunlight, rain, humidity, saltwater, and corrosive chemicals. This has made the pigment a longstanding component of protective finishes that safeguard metal infrastructure from environmental damage. While many chromium compounds are regulated due to toxicity, chromic oxide—being a trivalent chromium compound—poses significantly lower risk, allowing it to remain a preferred option in applications where stability and safety must coexist. Even in harsh industrial environments, chromic oxide coatings preserve both aesthetic appeal and protective function.
Chromic oxide is also essential in the manufacturing of refractory materials—substances capable of withstanding extraordinarily high temperatures without melting, deforming, or reacting. In the steel, glass, and cement industries, chromic oxide-containing refractories line furnaces, kilns, and reactors where temperatures can exceed 1500°C. The compound’s high melting point, structural rigidity, and resistance to chemical erosion make it an ideal component of refractory bricks, castables, and monolithic linings. These materials endure thermal shocks, mechanical stress, molten metal contact, and corrosive slag interactions while preserving their integrity. As a result, chromic oxide contributes directly to the efficiency, longevity, and safety of industrial thermal systems. Its presence in refractory technologies exemplifies how pigment chemistry can expand into structural and engineering roles far beyond coloration.
In metallurgy, chromic oxide acts as a reducing agent and fluxing component in the production of certain alloys and metals. When combined with other elements under controlled conditions, it can facilitate reactions that yield high-purity chromium metal or contribute to alloy formation. Although technological advances have introduced alternative methods for producing chromium metal, chromic oxide retains its relevance in specific metallurgical applications that benefit from its stability and controlled reactivity.
Chromic oxide’s role in abrasives further demonstrates its multifunctionality. Known commonly as “green rouge,” finely powdered chromic oxide serves as a polishing and buffing compound for metals, gemstones, and precision instruments. Its hardness and fineness make it ideal for producing mirror-like finishes on steel, chrome-plated surfaces, knives, optical components, and jewelry. Craftspeople and industrial polishers rely on green rouge for achieving high-precision results, which require both physical durability and controlled abrasion. The compound’s stability ensures that it does not chemically interact with the materials being polished, instead providing a clean mechanical smoothing action.
In plastics, rubber, and construction materials, chromic oxide imparts long-lasting green coloration that remains vibrant even under outdoor exposure. Its ability to resist degradation from UV radiation and temperature fluctuations makes it particularly valuable for durable goods such as synthetic turf, outdoor furniture, roofing membranes, molded plastics, signage materials, and elastomer products. Many of these items are expected to maintain structural and visual stability over years of sunlight, moisture, and environmental stress. Chromic oxide’s stability ensures that these materials retain their appearance and performance without fading or deterioration.
Environmental and safety considerations distinguish chromic oxide from more hazardous chromium species. Unlike chromium(VI) compounds, which are toxic, carcinogenic, and environmentally mobile, chromic oxide contains chromium in the +3 oxidation state—a form that is significantly less soluble, less biologically available, and far less reactive with human tissues. Trivalent chromium compounds such as chromic oxide do not easily penetrate cellular membranes or produce harmful oxidative intermediates. As a result, chromic oxide is generally considered safer in occupational settings when used as intended with reasonable protective measures. Regulatory frameworks typically impose fewer restrictions on chromic oxide compared with hexavalent chromium compounds, though proper handling, dust control, and worksite hygiene remain important to prevent excessive inhalation or prolonged contact.
In soil and environmental systems, chromic oxide behaves as a stable, insoluble material that does not readily dissolve or migrate through groundwater. Its environmental persistence can be viewed through dual lenses: on one hand, its low mobility reduces concerns about contamination; on the other hand, its inertness means that it remains in the environment without breaking down. However, because chromic oxide is non-toxic in its common forms and does not release chromium(VI) under normal environmental conditions, its persistence poses significantly fewer risks than many industrial materials. Its environmental footprint is therefore shaped more by its stability than by any inherent hazard.
In cultural and artistic contexts, chromic oxide has left a vibrant imprint on history. Artists adopted chromic oxide green for its opacity, depth, and weather resistance, applying it to landscapes, decorative arts, murals, and architectural features. Its stability ensures that artworks incorporating the pigment retain their color decades or even centuries after creation. Chromic oxide allowed artists to transition away from fugitive or toxic greens, contributing to the evolution of safer and more reliable palettes.
Ultimately, chromic oxide exemplifies the power of inorganic chemistry to produce materials that combine beauty, stability, and utility. It serves as a pigment, a refractory material, a protective coating, an industrial workhorse, and an artistic tool—all stemming from the inherent properties of chromium(III) oxide. Its resilience against heat, chemicals, and environmental conditions illustrates how nature’s structural designs can yield extraordinary endurance. At the same time, its safety relative to more hazardous chromium compounds highlights the significance of oxidation state and chemical structure in determining a material’s interactions with living systems. By understanding chromic oxide, we gain insight into how elemental properties, crystal chemistry, optical behavior, and industrial innovation converge to create one of the most versatile and enduring inorganic compounds in the modern world.