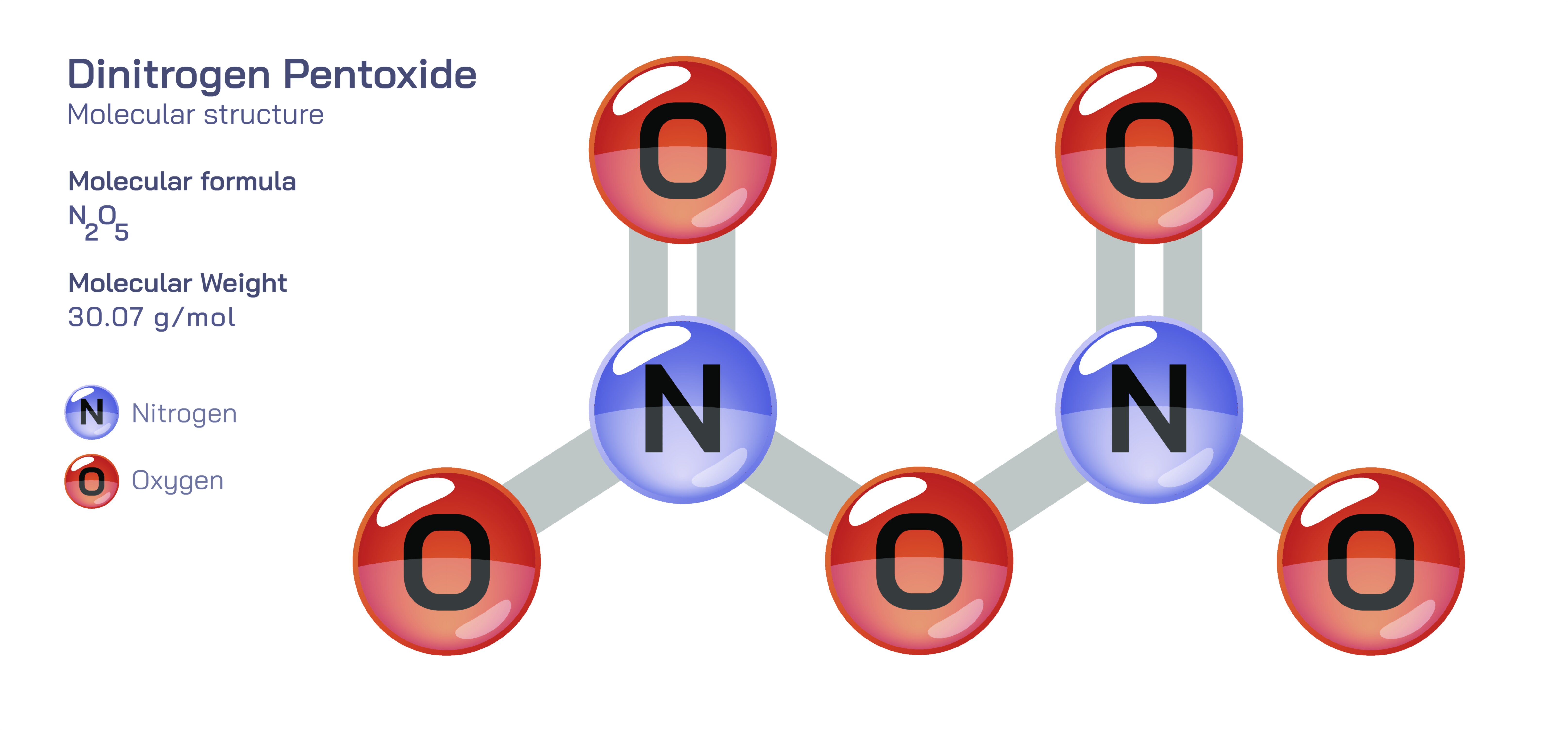
Dinitrogen Pentoxide — Nitrogen Oxide Compound
Dinitrogen pentoxide is one of the most intriguing members of the nitrogen oxide family, a group of compounds that collectively play central roles in atmospheric chemistry, industrial processes, and the environmental cycles that regulate Earth’s climate. Although its chemical formula appears simple—with two nitrogen atoms and five oxygen atoms—the molecule’s behavior is anything but ordinary. Dinitrogen pentoxide occupies a conceptual space between molecular chemistry and ionic structure, sitting at the crossroads of reactivity, instability, and environmental influence. Its properties reflect deep principles of bonding, decomposition, acid formation, and atmospheric transformation. As one of the anhydrides of nitric acid, dinitrogen pentoxide helps link the behaviors of nitrogen oxides with the formation of nitrates, the balance of reactive nitrogen species, and the processes that govern pollution and climate interactions. Understanding this compound reveals how molecular structure determines not just chemical reactivity but also large-scale processes reaching far beyond the immediate environment.
At standard conditions, dinitrogen pentoxide exists as a white, crystalline solid that is inherently unstable. This instability arises from the way oxygen and nitrogen atoms are arranged within the molecule. While the formula suggests a simple N₂O₅ arrangement, the solid-state structure is more accurately represented by an ionic framework composed of nitrate and nitronium ions. In this form, the compound can be thought of as containing NO₃⁻ (nitrate) and NO₂⁺ (nitronium) units. The nitronium ion, in particular, is an important electrophilic species in many reactions involving nitration, especially in organic chemistry. This ionic representation highlights how dinitrogen pentoxide bridges molecular and ionic chemistry depending on temperature and physical state. Its identity therefore cannot be captured fully by a single structural description. Instead, its behavior must be understood as a delicate balance between its ionic arrangement in the solid state and its more loosely associated, covalent-type structure when in gaseous or dissolved forms.
Because of its unstable nature, dinitrogen pentoxide decomposes easily, especially when exposed to moisture, heat, or light. A key transformation occurs when the compound comes into contact with water, forming nitric acid through a process that reflects its role as an acid anhydride. This reaction connects dinitrogen pentoxide to fundamental acid–base chemistry and to the broader environmental cycles that involve nitric acid formation. In atmospheric systems, nitric acid contributes to acid rain, influences aerosol formation, and affects the chemical reactivity of the troposphere and stratosphere. Although nitric acid itself is a stable product, the transformation from dinitrogen pentoxide illustrates the interlinked pathways through which nitrogen oxides shift between reactive and stabilized forms. Dinitrogen pentoxide therefore acts as a transient species—a chemical intermediate that mediates transformations between NOₓ gases and acid products. Its structure and reactivity help regulate the atmospheric concentration of nitrogen dioxide and nitric oxide, enabling the atmosphere to buffer and redistribute reactive nitrogen.
Dinitrogen pentoxide’s instability also makes it an important participant in nighttime atmospheric chemistry. During the day, sunlight drives reactions involving nitrogen oxides that influence ozone levels and photochemical smog. At night, however, when light-driven reactions subside, dinitrogen pentoxide becomes a key reservoir molecule. It forms reversibly from nitrogen dioxide and ozone through intermediary species, effectively storing reactive nitrogen until sunlight returns. This nighttime storage role is essential: without such reservoir molecules, reactive nitrogen species would participate in a continuous series of reactions that could drastically shift atmospheric composition. Dinitrogen pentoxide therefore contributes to the daily rhythm of atmospheric chemistry—not by persisting for long periods, but by forming and decomposing in patterns that regulate reactivity. This dynamic behavior highlights how molecular identity influences environmental timing as much as environmental outcomes.
When dinitrogen pentoxide breaks down, one of its main decomposition pathways yields nitrogen dioxide and oxygen. This decomposition reflects internal strain in the molecule and the energy imbalance within the nitrogen–oxygen framework. Although this breakdown limits the stability of the compound under standard conditions, it is precisely this decomposition tendency that gives dinitrogen pentoxide practical utility in certain industrial and laboratory environments. In controlled settings, its ability to release reactive nitrogen dioxide or nitronium ions enables specialized nitration reactions, particularly in organic synthesis. The nitronium ion produced from dinitrogen pentoxide functions as an electrophile that attacks aromatic rings in nitration processes. These reactions help create nitroaromatic compounds, which are crucial intermediates in the production of dyes, pharmaceuticals, explosives, and agriculture-related chemicals. The connection between molecular instability and chemical utility illustrates a broader principle in chemistry: substances that are difficult to store or handle under standard conditions often become exceptionally powerful reagents when used with precision.
In industrial settings, dinitrogen pentoxide is sometimes generated in situ—meaning produced at the moment of use—rather than stored. This practice reflects the compound’s instability and the challenges associated with transporting or storing reactive nitrogen oxide solids. By generating dinitrogen pentoxide from nitrogen dioxide or nitric acid derivatives within controlled reactors, chemists can harness its reactivity without contending with its decomposition risks. This highlights the practical relationship between molecular structure and industrial strategy: the same qualities that make dinitrogen pentoxide chemically interesting also make it logistically challenging, requiring careful engineering solutions to integrate its reactivity safely into technological processes.
The environmental significance of dinitrogen pentoxide extends beyond acid formation and nighttime nitrogen storage. It plays a role in the formation of nitrate aerosols—tiny particulate matter consisting of atmospheric nitrates bound to water and other chemical components. These aerosols influence cloud formation, radiative balance, and the scattering of sunlight. Because aerosols can cool or warm different parts of the atmosphere depending on their composition and location, the transformation of dinitrogen pentoxide into particulate nitrate contributes to climate-relevant processes. Understanding this behavior requires appreciating how the compound’s molecular instability enables reactions with water vapor on atmospheric particle surfaces. These reactions serve as bridges between gas-phase nitrogen species and particulate-phase nitrogen species, illustrating how a compound’s reactivity drives transitions between different environmental forms that influence both air quality and climate.
The compound’s role in pollution chemistry is equally important. Nitrogen oxides, collectively referred to as NOₓ, are major contributors to smog formation and poor air quality. Dinitrogen pentoxide forms part of the interconversion network that distributes these oxides through various pathways, influencing their atmospheric lifetime, reactivity, and eventual removal. Dinitrogen pentoxide itself does not directly create smog; instead, it participates in pathways that temporarily remove nitrogen oxides from daytime smog-forming cycles, only to release them again under certain conditions. This nonpermanent sequestration helps moderate the pace of daytime photochemistry but also means that nighttime reactions can influence next-day pollution events. Thus, understanding the structure and behavior of dinitrogen pentoxide is crucial for environmental scientists working to predict air quality and model atmospheric reactivity under varying weather and pollution conditions.
From a laboratory perspective, dinitrogen pentoxide offers significant educational value because it demonstrates how structure dictates both reactivity and stability. Its ionic character in solid form teaches lessons about the relationship between molecular and ionic bonding. Its decomposition reactions highlight how energy imbalances create instability. Its reversibility in atmospheric cycles illustrates how dynamic equilibria govern chemical behavior in natural systems. Through dinitrogen pentoxide, students and researchers can explore deeper truths about nitrogen chemistry: the versatility of nitrogen–oxygen bonding, the transitions between oxidation states, and the fine balance between molecular identity and environmental context. The compound embodies how small variations in atomic arrangement produce vast differences in chemical behavior, even among closely related nitrogen oxides.
Dinitrogen pentoxide also ties into broader conceptual themes in chemistry: the interplay between structure and environment, the role of reactive intermediates, and the importance of transient species in sustaining longer-term cycles. Although the molecule rarely exists in large, isolated quantities outside laboratories, its presence in the atmosphere, its role in the nitrogen cycle, and its relevance in industrial synthesis amplify its impact far beyond its fleeting existence. Dinitrogen pentoxide’s molecular identity—straddling the line between stability and reactivity—allows it to serve as a bridge between chemical worlds: between gases and aerosols, between nitrogen oxides and nitric acid, between environmental reservoirs and active pollutants. In every case, its behavior reflects the core chemical principle that structure determines function, and function shapes influence.
Ultimately, dinitrogen pentoxide stands as a vivid illustration of how a compound with a deceptively simple formula can exert far-reaching effects through its molecular structure. Its identity as both a nitrogen oxide and an acid anhydride allows it to influence atmospheric chemistry, environmental processes, and industrial techniques. Its instability, while posing challenges, unlocks powerful reactivity that chemists harness in synthetic contexts. Its role in nighttime atmospheric cycles reveals how even short-lived molecules shape broader environmental dynamics. Through its diverse behaviors, dinitrogen pentoxide reveals the intricate connections between the microscopic world of chemical bonding and the macroscopic world of climate, pollution, industry, and natural cycles.