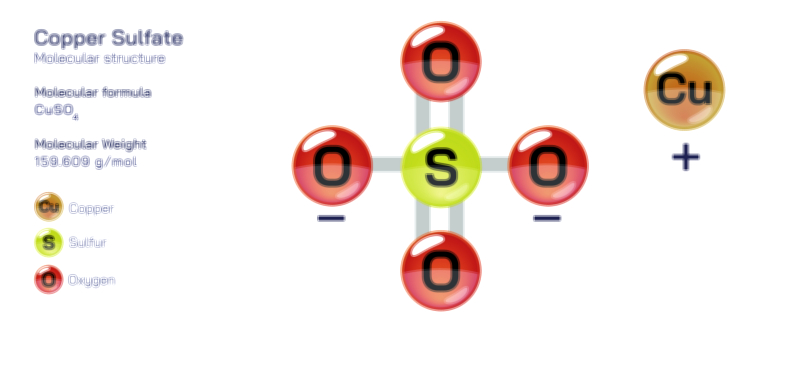
Copper(I) Oxide — Inorganic Chemical Compound
Copper(I) oxide is an important inorganic chemical compound that occupies a distinctive place in the family of copper-based materials due to its unique red coloration, its semiconducting properties, its role in redox transformations, and its relevance across industry, materials science, environmental chemistry, and fundamental inorganic studies. Unlike copper(II) oxide, which is black and contains copper in the +2 oxidation state, copper(I) oxide features copper in the +1 oxidation state, giving rise to very different chemical behaviors, structural characteristics, and applications. Its formula reflects a simple pairing between copper ions and oxide ions, yet this apparent simplicity conceals a fascinating depth: copper(I) oxide demonstrates how oxidation state, crystalline arrangement, and electron configuration all combine to shape the identity and performance of an inorganic compound. Its rich history, diverse uses, and intriguing physical properties make it a central topic in both classical and advanced inorganic chemistry.
One of the most recognizable traits of copper(I) oxide is its brilliant red or reddish-orange coloration. This distinctive appearance has long attracted scientific, artistic, and industrial interest. The color arises from the way electrons interact within the copper(I) ion’s environment and how the oxide lattice influences electronic transitions. The compound crystallizes in a cubic structure that governs light absorption and scattering, resulting in the vivid hue often associated with copper-based pigments. Historically, copper(I) oxide contributed to red and orange pigments used in ceramics, glazes, and decorative materials. When incorporated into glass or pottery and heated under the right conditions, copper(I) oxide produces warm, earthy red tones valued in traditional craftwork. These uses illustrate how inorganic chemistry directly influences human artistic expression and how the structural features of a simple compound can generate striking visual effects.
On the chemical level, copper(I) oxide plays a key role in understanding redox behavior in the copper system. Copper can cycle between oxidation states +1 and +2, and copper(I) oxide often forms as an intermediate in these transformations. For instance, when copper metal is heated in the presence of limited oxygen, it first forms copper(I) oxide before further oxidation yields copper(II) oxide under more oxygen-rich conditions. Conversely, when copper(II) compounds encounter reducing agents, copper(I) species often appear as transient or stable intermediates. These transitions highlight copper’s ability to shift electron configurations easily, making copper compounds highly relevant in catalysis, electrochemistry, and corrosion processes. Understanding how copper(I) oxide forms and transforms is essential in fields ranging from metallurgy to environmental cycles, where copper’s oxidation states govern its reactivity and mobility.
Copper(I) oxide also exhibits interesting solubility behavior that distinguishes it from copper(II) compounds. It is generally insoluble in water and only slightly soluble in dilute acids, requiring stronger acids or complexing agents to dissolve effectively. In contact with ammonia or cyanide solutions, copper(I) ions form characteristic complexes, illustrating how ligand interactions govern solubility and stability. These complexation reactions underscore broader principles of coordination chemistry, where ligand type, charge, and geometry dictate how metal ions behave in solution. The relative insolubility of copper(I) oxide makes it useful in applications where gradual release of copper ions is desired, such as in antifouling coatings for marine equipment. These coatings help prevent the buildup of algae, barnacles, and other organisms by slowly releasing copper ions that deter biological growth. Although modern environmental considerations have reduced reliance on copper-based antifouling paints, copper(I) oxide remains a key example of how inorganic materials interact with biological systems.
In materials science, copper(I) oxide has gained significant attention due to its semiconducting properties. It is considered a p-type semiconductor, meaning it conducts electricity primarily through the movement of positive charge carriers known as “holes.” This property arises from slight deficiencies in copper ions within the lattice structure, which create electron vacancies that facilitate electrical conduction. Because of this, copper(I) oxide has been studied for use in photovoltaic cells, sensors, photocatalysts, and various electronic devices. Although it does not match the efficiency of silicon in mainstream solar technology, copper(I) oxide’s abundance, low toxicity, and relatively simple production methods make it an appealing material for low-cost or experimental solar applications. Researchers also explore its use in photoelectrochemical water splitting, where sunlight triggers chemical reactions that split water into hydrogen and oxygen. The study of copper(I) oxide in these contexts demonstrates how inorganic materials with modest structures can contribute to renewable energy research.
Copper(I) oxide plays a central role in catalysis as well. Its surface structure provides active sites that facilitate chemical transformations involving hydrogenation, oxidation, or coupling reactions. In organic chemistry, copper(I) oxide serves as a catalyst or co-catalyst in reactions forming carbon–carbon or carbon–nitrogen bonds. It helps initiate transformations that would otherwise require more complex systems. Its ability to switch oxidation states, interact with reactant molecules, and stabilize reaction intermediates makes it valuable in modern synthetic methodologies. In industrial contexts, copper-based catalysts incorporating copper(I) oxide contribute to processes such as gas purification, polymer manufacture, and chemical synthesis. These applications highlight how inorganic compounds provide foundational tools for controlling and accelerating chemical reactions.
The compound also offers insights into corrosion processes involving copper and its alloys. When copper surfaces are exposed to mild oxidizing conditions—such as damp air or weak oxidants—copper(I) oxide frequently forms as an initial corrosion product. Over time, depending on moisture and environmental composition, the layer may convert into copper(II) oxide or basic copper salts. As a thin film, copper(I) oxide sometimes provides partial protection to underlying copper surfaces by acting as a barrier to further oxidation. However, its stability depends heavily on environmental factors. In aqueous or chloride-rich environments, copper(I) oxide may dissolve or transform into other corrosion products. Understanding these processes is essential in fields such as plumbing, electronics, marine engineering, and artifact conservation, where copper materials must withstand long-term exposure to varied conditions.
In environmental chemistry, copper(I) oxide participates in natural cycles of copper mobility and transformation. As part of weathering processes, copper minerals may break down to form oxide layers or carbonate coatings, which influence how copper moves through soil and water. Its interactions with organic matter, dissolved oxygen, and microbial populations help determine whether copper remains immobilized or becomes bioavailable. Because copper is both an essential nutrient and a potential toxin at high concentrations, understanding the formation and behavior of copper(I) oxide contributes to environmental assessments, pollution mitigation, and ecological modeling.
In educational contexts, copper(I) oxide provides a useful demonstration of redox chemistry, precipitation behavior, and thermal transformations. It can be synthesized in the laboratory by reducing copper(II) salts with suitable reagents, illustrating how electron transfer alters oxidation state and compound identity. Conversely, heating copper metal in limited air supplies shows how controlled oxidation steps yield different copper oxides. These demonstrations help students grasp how oxidation state, stoichiometry, and environmental conditions influence the structure and appearance of inorganic materials. The compound’s distinctive red coloration provides a clear visual cue that reinforces learning about transition metals and oxidation processes.
Copper(I) oxide also finds limited use in ceramics and glass production, where it can impart subtle red hues. In glazes, the final color produced depends heavily on firing temperature, kiln atmosphere, and the presence of reducing agents. Achieving consistent coloration can be challenging because copper(I) oxide may transform into copper(II) oxide or even metallic copper during heating. These transformations offer a powerful reminder of how sensitive copper compounds are to oxidation conditions and how artists and craftspeople must understand chemistry to control ceramic finishes.
Structurally, copper(I) oxide highlights how coordination environments influence stability and properties. Its cubic arrangement, known as the cuprite structure, places each oxygen ion at the center of a tetrahedron of copper ions, forming a distinctive network. This structure contributes to its semiconducting behavior and optical characteristics. Understanding this arrangement has helped researchers design materials with similar frameworks but varied chemical compositions, aiming to tailor electronic and catalytic properties. In this way, copper(I) oxide serves as a blueprint for materials engineering efforts that draw inspiration from inorganic crystalline structures.
Ultimately, copper(I) oxide exemplifies the multifaceted nature of inorganic compounds, showing how a material defined by a simple chemical composition can express profound variability in behavior depending on environment, oxidation state, structural arrangement, and application. Its bright red form serves as a visual anchor for understanding copper’s chemistry, while its roles in catalysis, semiconducting technology, ceramics, corrosion science, environmental behavior, and education illustrate its broad significance. As both a classical compound with historical uses and a modern material with ongoing scientific relevance, copper(I) oxide demonstrates how inorganic chemistry continues to bridge tradition and innovation, connecting fundamental principles with evolving technological needs.