Clausius Statement — Second Law of Thermodynamics
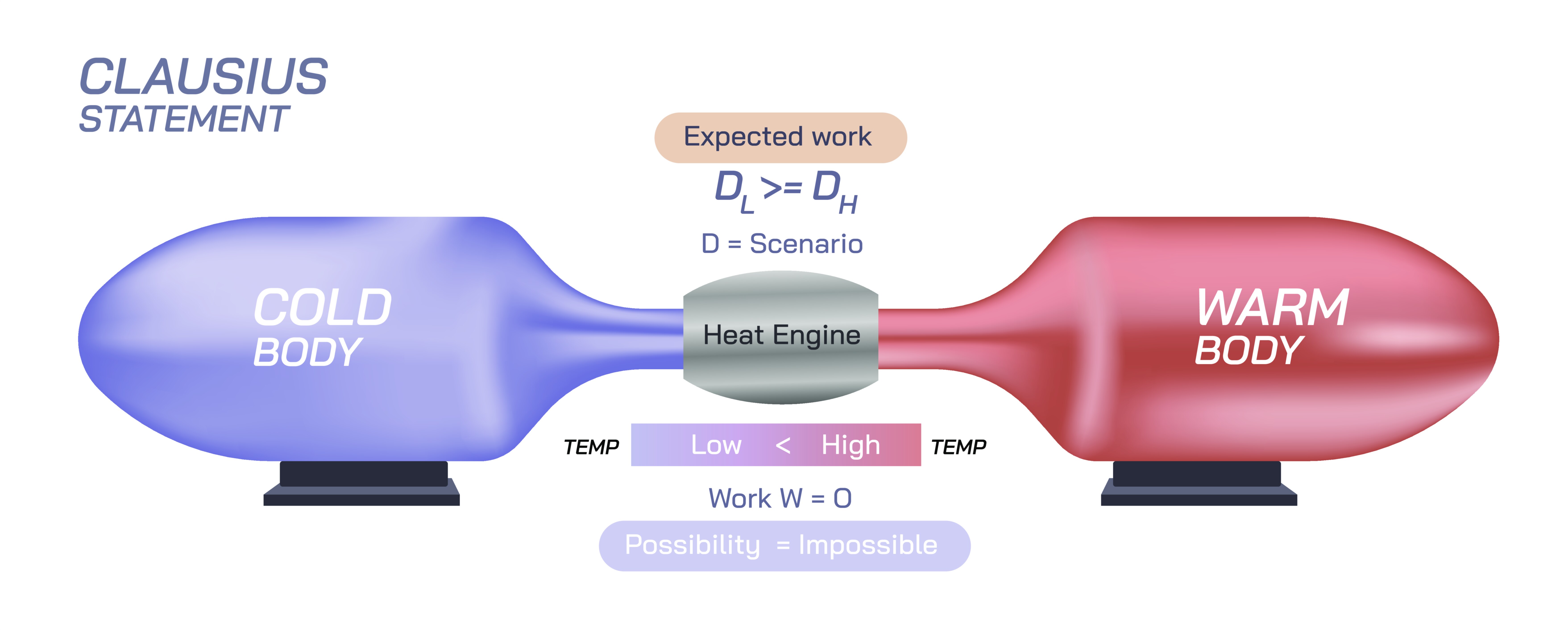
The Clausius statement of the Second Law of Thermodynamics stands as one of the most profound and foundational principles in all of physical science, shaping our understanding of energy flow, heat transfer, natural processes, and the fundamental limits of what can and cannot occur in the universe. Though formulated in simple language, its implications reach deep into physics, chemistry, engineering, cosmology, biology, and every domain where energy transformations take place. The Clausius statement expresses a universal truth about the directionality of heat: in the absence of external influence, heat will never spontaneously travel from a colder object to a hotter one. Instead, natural processes always carry thermal energy from regions of higher temperature to regions of lower temperature. This deceptively modest rule limits the behavior of engines, refrigerators, biological organisms, planetary climates, chemical reactions, and even the unfolding of time itself. The Clausius statement captures the irreversibility built into the fabric of the physical world, revealing why some processes unfold easily and naturally while their reverse forms remain impossible without deliberate intervention.
At its core, the Clausius statement provides an intuitive way of describing the Second Law without relying on mathematical form or abstract quantities. It tells us that heat seeks equilibrium, flowing in a direction that spreads out energy rather than concentrating it. If two bodies at different temperatures are placed in contact, the warmer body cools and the cooler one warms until their temperatures equalize. This everyday experience reflects a deep natural tendency toward increasing overall dispersal of thermal energy. The statement denies the possibility of spontaneous reversal: a cold object will never suddenly warm itself further by drawing heat out of a warmer neighbor. Even though physics allows countless microscopic interactions between particles, the collective behavior of those particles always organizes itself in a way that respects the Second Law. This directionality is not imposed by artificial rules but emerges from the statistical behavior of trillions of interacting atoms and molecules.
The Clausius statement also implies that certain hypothetical machines cannot exist. A device that causes heat to flow from a cold reservoir to a hot reservoir without any external work—often called a “Clausius-violating machine”—would break the fundamental logic of nature. Refrigerators, air conditioners, and heat pumps all move heat from cold to hot regions, but they require work from outside sources, such as electrical energy, to accomplish this. Without such work, the natural direction of heat flow cannot be reversed. This limitation shapes the design of all thermal technologies, ensuring that no engine, no cycle, and no machine can circumvent the restrictions dictated by the Second Law. The Clausius statement therefore functions both as a scientific description of natural behavior and as a constraint on engineering possibilities.
This principle reveals that natural processes possess an inherent arrow of time. When heat spreads from a hot object to a cold one, the process unfolds spontaneously. But the reverse—heat spontaneously organizing itself into a more concentrated state—does not occur in nature. This asymmetry is one of the clearest manifestations of time’s directionality in the physical world. Most fundamental laws of physics do not actually distinguish between forward and backward time; gravitational interactions, electromagnetic processes, and even many atomic interactions appear symmetric when time is reversed. Yet in thermodynamics, the Clausius statement codifies an irreversible trend. It explains why melting ice never refreezes itself without cooling, why iron rusts but rust does not spontaneously reorganize into iron, and why the diffusion of heat and energy always corresponds to a progression from ordered states to more disordered ones. The Clausius statement gives the Second Law its experiential significance by linking microscopic behavior to macroscopic irreversibility.
In practical engineering, the Clausius statement guides the functioning of heat engines and refrigeration systems. A heat engine works by exploiting the natural flow of heat from a hot reservoir to a cold reservoir. This flow can be partially converted into work, such as the motion of a piston or the rotation of a turbine. Yet the Clausius statement guarantees that no engine can operate with perfect efficiency. Some heat must always be discharged to a colder body, and this waste heat is not a flaw of machine design but a requirement set by the Second Law. Similarly, when designing refrigeration systems, engineers know that energy must be supplied to move heat from a cold interior to a warm exterior. The Clausius statement forces the inclusion of compressors, pumps, or electrical inputs that provide the required work. Even advanced technologies—cryogenic coolers, heat pumps, thermal batteries—adhere to the same principle. No matter how sophisticated the machinery becomes, it cannot ignore the fundamental truth that heat only flows against its natural gradient when work is applied.
In climate science and meteorology, the Clausius statement reveals why atmospheric circulation patterns form as they do. Warm air rising from heated ground surfaces, oceanic heat flowing toward polar regions, and wind patterns that redistribute thermal energy across the globe all follow the natural direction of heat flow from warmer to cooler regions. The Clausius statement provides a conceptual backbone for understanding why weather systems behave the way they do. Thunderstorms develop when hot, moist air rises into cooler altitudes, releasing energy as it condenses. Ocean currents carry warm water from equatorial zones toward cooler latitudes. Heat always spreads outward, and the complex choreography of atmospheric and oceanic motion ultimately mirrors the fundamental thermodynamic rule embedded in the Clausius statement.
The Clausius principle also shapes chemical reactions. Many reactions occur spontaneously because they release energy into surroundings that are at a lower temperature. Exothermic reactions proceed readily as they increase overall energy dispersal. Endothermic reactions, on the other hand, require outside energy to force the system to adopt a less naturally favored state. Chemical equilibria, reaction spontaneity, and the behavior of mixtures and solutions all hinge on the directionality imposed by the Second Law. While chemists once viewed reactions only through the lens of energy release or absorption, modern thermodynamics shows that directionality is best understood in terms of energy spreading and the Clausius rule that heat does not concentrate unless driven by an external agent.
In biological systems, the Clausius statement plays a subtle yet foundational role. Life forms maintain ordered internal structures and regulate processes that depend on thermal and chemical gradients. But organisms cannot violate the Second Law. They maintain order by consuming energy from food, sunlight, or chemical resources, generating heat as a byproduct. Metabolic processes continually move thermal energy outward into the environment. Cells sustain their internal organization only by exporting disorder, releasing heat into surroundings that are cooler than the metabolic centers within the organism. This outward flow of heat ensures compliance with the Clausius statement while allowing life to maintain structural order. The principle explains why organisms cannot survive without energy input and why metabolic processes produce heat even when performing tasks that appear unrelated to temperature.
In material science, the Clausius statement governs heat treatment, welding, forging, annealing, and the thermal stability of solids. Metals heated in a furnace absorb energy as their atoms vibrate more vigorously, and once removed from the heat source, they cool naturally by releasing heat into cooler surroundings. The rate at which materials heat up or cool down is determined by thermal gradients consistent with the Clausius rule. No metal spontaneously heats itself while resting in a cooler environment, and no ceramic absorbs thermal energy from a cold region to warm a hot surface around it. Whether studying conduction through solids, convection in fluids, or radiation among surfaces, the Clausius statement asserts itself through every pathway of thermal exchange.
In environmental science, the natural flow of heat influences ecosystems, seasons, and planetary energy balance. Solar energy warms Earth’s surface, and this heat radiates outward into the cooler atmosphere and eventually into space. This outward flow is an enormous-scale example of the Clausius principle. If heat were to spontaneously move from cooler cosmic background conditions back toward Earth’s surface in concentrated form, planetary temperatures would behave unpredictably. Instead, Earth’s climate system stabilizes through a consistent exchange in which high-temperature regions lose heat to lower-temperature environments. Even global warming, driven by enhanced greenhouse effects, does not violate the Clausius statement. Instead, greenhouse gases limit the outward radiation of heat by trapping it, slowing the natural flow and raising local temperatures—but never reversing the direction of heat movement.
The Clausius statement also intersects with information theory and entropy. Claude Shannon, who formalized information theory, defined information in ways that parallel thermodynamic entropy. Both systems demonstrate a tendency toward increased uncertainty or disorder unless external work is applied. Just as the Clausius principle states that heat requires work to move from cold to hot, information systems require work to reduce uncertainty or isolate meaningful data from noise. These parallels highlight how the Second Law touches even conceptual realms of knowledge, computation, and communication.
In cosmology, the Clausius statement contributes to our understanding of the universe’s fate. As stars radiate heat into the vast coldness of space, as galaxies emit energy that disperses outward, and as all processes follow the trend of decreasing temperature gradients, the universe moves gradually toward a state of maximal uniformity. This distant future, often described as thermodynamic equilibrium, reflects the Clausius principle extended across cosmic time. The idea that no large-scale heat flows will remain, and no organized energy gradients will persist, derives directly from the natural direction of thermal movement expressed by the Clausius statement.
The significance of the Clausius principle becomes especially clear when imagining a world without it. If heat could spontaneously flow from cold to hot, refrigerators would operate without electricity, chemical reactions could reverse spontaneously, molten materials might freeze while surrounded by warmer environments, and natural processes would lose their directionality. Temperature itself would cease to be a meaningful concept. Instead, our universe exhibits order within limitation: heat disperses, energy spreads out, and systems evolve toward equilibrium in ways that make natural processes predictable, understandable, and governed by stable laws. The Clausius statement captures this universal order in a concise declaration that defines the boundary between possible and impossible phenomena.
Ultimately, the Clausius statement of the Second Law of Thermodynamics stands as a guiding principle that shapes everything from engines and weather patterns to biological life and the evolution of the universe. It represents one of the deepest truths about physical reality: that heat seeks balance, that energy disperses naturally, and that organized structures endure only through constant input of work. Its simplicity masks its profound power. From microscopic particles to galaxies, from industrial machines to human bodies, the Clausius statement governs the flow of thermal energy and sets the direction for the unfolding of time itself.