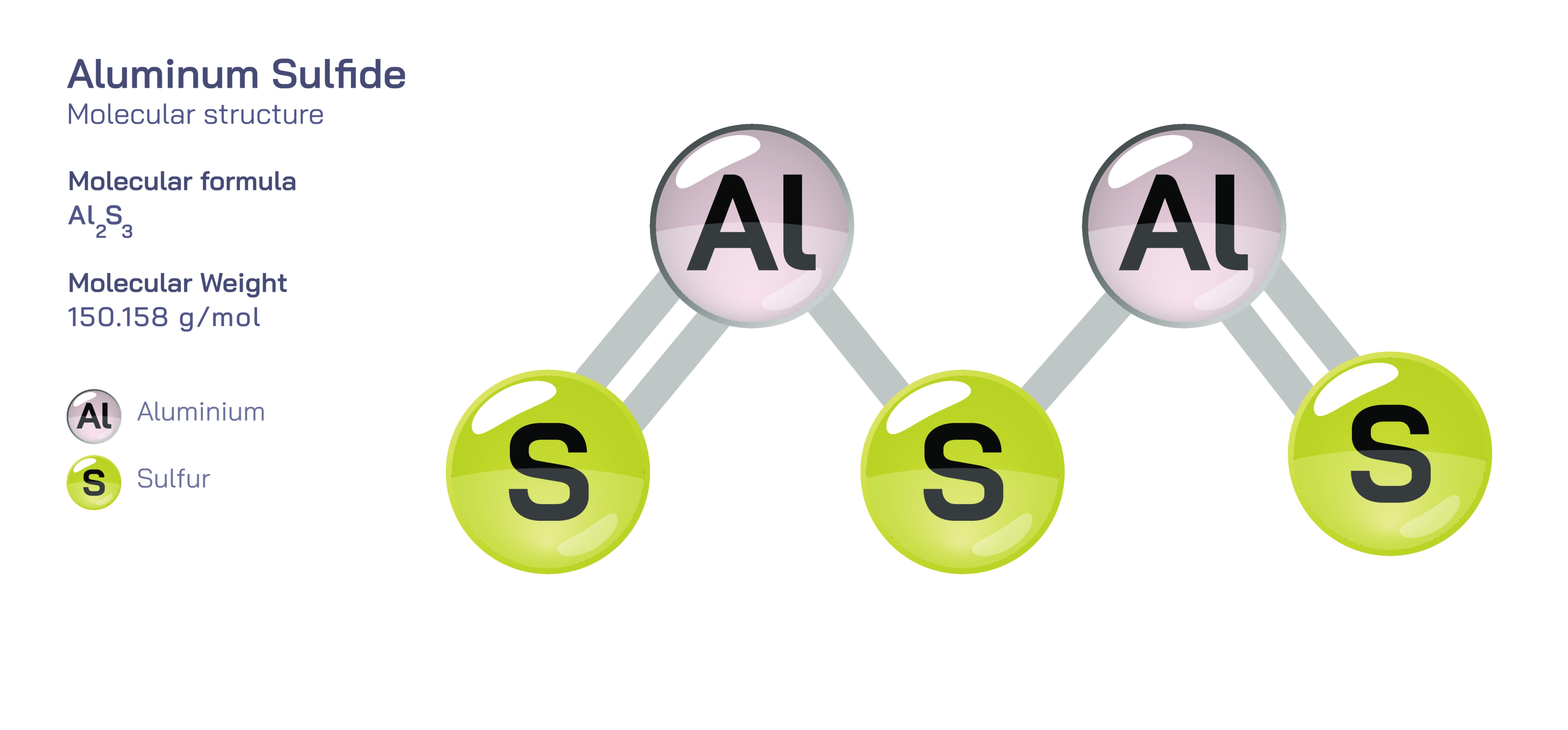
Aluminum Sulfide — Chemical Structure and Inorganic Compound Illustration
Fundamental Identity and Structural Basis of Aluminum Sulfide
Aluminum sulfide, commonly expressed by the formula Al₂S₃, is an important inorganic compound formed through the combination of trivalent aluminum and divalent sulfur. Although seemingly simple in composition, its structural behavior, reactivity, and industrial relevance extend far beyond what the formula suggests. In its solid state, aluminum sulfide occupies a complex lattice arrangement that reflects the strong electrostatic interactions between the positively charged aluminum ions and negatively charged sulfide ions. This compound naturally crystallizes in a form similar to other metal sulfides, but it exhibits variations depending on temperature, preparation method, and impurities. Aluminum sulfide is well known for its instability in moist air, as it reacts immediately with water to produce aluminum hydroxide and hydrogen sulfide gas. This sensitivity to moisture underscores its highly reactive character, distinguishing it from the more stable aluminum oxides, phosphates, or fluorides. Despite this reactivity, aluminum sulfide serves as a crucial precursor in several inorganic synthesis routes, a functional intermediate in metallurgical reactions, and a valuable teaching compound for understanding metal–sulfur interactions in solid-state chemistry.
Chemical Bonding, Lattice Architecture, and Molecular Arrangement
The bonding in aluminum sulfide can be understood primarily through ionic interactions: aluminum carries a +3 charge, while sulfur carries a -2 charge. This charge difference leads to the formation of a repetitive ionic lattice in which each Al³⁺ ion is surrounded by sulfide ions in tetrahedral coordination. The most common crystalline form adopts a structure analogous to β-Al₂O₃ or defect spinel frameworks, where the cations and anions interact through geometrically arranged tetrahedral and octahedral sites. The arrangement is influenced by the fact that aluminum, being small and highly charged, prefers a tetrahedral environment when coordinating with larger anions like sulfur. In many structures, the aluminum sulfide lattice appears as alternating layers where tetrahedra connect in complex patterns that maximize charge balance and minimize electrostatic repulsion. Unlike covalent metal sulfides such as MoS₂ or WS₂, aluminum sulfide remains largely ionic, with directional bonding governed by lattice geometry rather than delocalized electron clouds. The compound exhibits several polymorphs, some of which form under high temperatures, further demonstrating that aluminum sulfide’s structure is adaptable and dependent on external conditions, even though its fundamental ionic nature remains intact across variations.
Physical Properties and Their Functional Implications
Aluminum sulfide is a solid that ranges from off-white to grayish in color depending on purity and preparation method. Its most defining physical characteristic is its extreme sensitivity to moisture: even atmospheric humidity causes it to decompose, releasing hydrogen sulfide, a toxic gas with a strong rotten-egg odor. This reactivity makes it difficult to store and handle under normal environmental conditions, requiring sealed containers or inert atmospheres. The compound possesses a relatively high melting point for an ionic sulfide, reflecting the strength of the electrostatic interactions within its crystal lattice. It is insoluble in most organic solvents and only undergoes solubilization through decomposition reactions in aqueous environments. Aluminum sulfide's density and hardness depend on its crystalline form, but regardless of variation, it maintains the typical rigidity expected of an ionic solid with a tightly bound lattice. These physical characteristics—reactivity with moisture, thermal stability in dry conditions, and solid-state rigidity—shape its use in controlled environments where its chemical properties can be harnessed safely and effectively.
Chemical Reactivity and Transformation Pathways
The reactivity of aluminum sulfide is dominated by its rapid and spontaneous hydrolysis. Exposure to water produces aluminum hydroxide and hydrogen sulfide gas according to the reaction:
Al₂S₃ + 6H₂O → 2Al(OH)₃ + 3H₂S
This transformation is exothermic and practically instantaneous, making aluminum sulfide an efficient generator of H₂S in laboratory and industrial synthesis when carefully controlled. Beyond hydrolysis, aluminum sulfide participates in high-temperature reactions with metal oxides, halides, and other inorganic materials, often acting as a sulfiding agent that introduces sulfur atoms into metal centers. In metallurgy, it may appear as an intermediate in refining processes or be used to produce metal sulfides with specific stoichiometries. Aluminum sulfide can also react with strong acids to release hydrogen sulfide and form aluminum salts. Under certain furnace conditions, it decomposes thermally, releasing sulfur vapors and transitioning into alumina-rich phases. Although not known for catalytic behavior in the same way as transition metal sulfides, it still displays particular reactivity patterns that make it suitable for sulfur-containing synthesis routes and inorganic transformations that depend on strong ionic movement or sulfide transfer.
Industrial, Environmental, and Practical Applications
Aluminum sulfide, while not as widely used as aluminum oxide or aluminum fluoride, remains important in specialized industrial contexts. Its primary industrial significance lies in its role as a precursor for manufacturing other aluminum-containing compounds, particularly those requiring the introduction of sulfur. In some chemical processes, aluminum sulfide is used as a sulfiding agent to convert metal oxides into their corresponding sulfides. In the realm of materials science, research continues into the behavior of aluminum–sulfur systems at high temperatures, particularly in connection with ceramics, refractories, and high-performance composites. In controlled laboratory environments, aluminum sulfide serves as a starting point for generating hydrogen sulfide in small-scale synthesis, allowing controlled production without relying on unstable or hazardous storage of gaseous H₂S. Environmental considerations center mostly on containment, since accidental exposure to moisture can result in the release of toxic hydrogen sulfide, making careful handling and storage essential. Despite its reactive nature, aluminum sulfide remains a valuable inorganic compound that supports both experimental chemistry and specific industrial processes requiring the unique interplay of aluminum and sulfur.
Illustrating the Structural and Chemical Behavior of Aluminum Sulfide
Illustrations of aluminum sulfide typically emphasize its tetrahedral aluminum coordination, showing Al³⁺ ions surrounded by four S²⁻ ions in a three-dimensional lattice. These diagrams often highlight the contrast between the compound’s dry stability and its explosive hydrolysis upon exposure to moisture. Structural illustrations may also depict the layered or networked arrangements of the lattice, comparing different polymorphs and their geometric variations. Reaction pathway illustrations often show the rapid decomposition into aluminum hydroxide and hydrogen sulfide, visualizing the release of gas and transformation of the solid. In industrial contexts, diagrams may portray aluminum sulfide as a reactant in high-temperature reactors, or as part of sulfiding sequences that convert metal oxides into sulfides. Through these visual representations, the compound’s ionic architecture, reactivity, and industrial significance become clearer, demonstrating how aluminum sulfide functions both as a structurally intriguing solid and a highly reactive participant in inorganic chemistry.