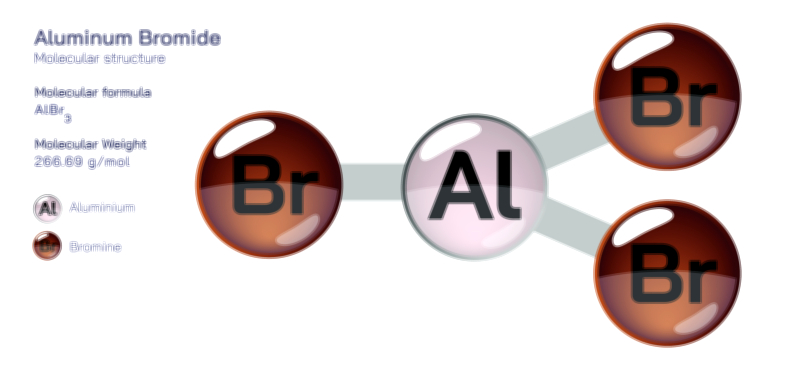
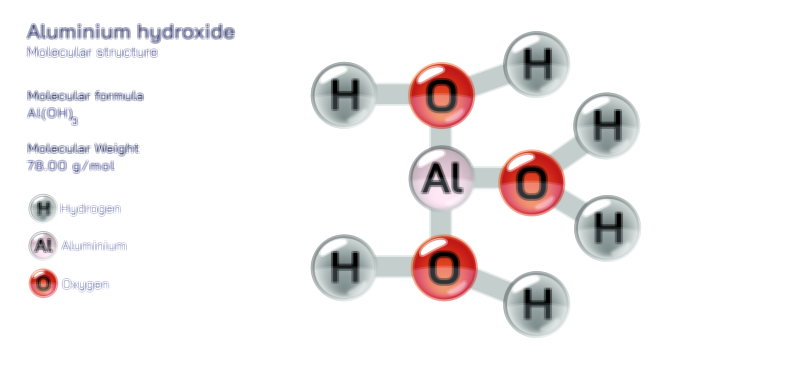
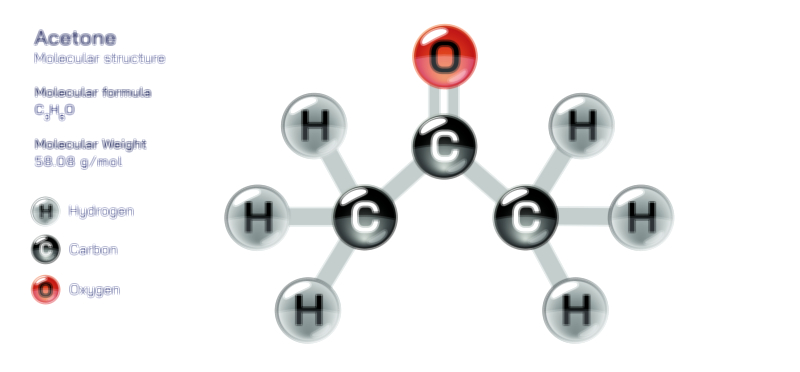
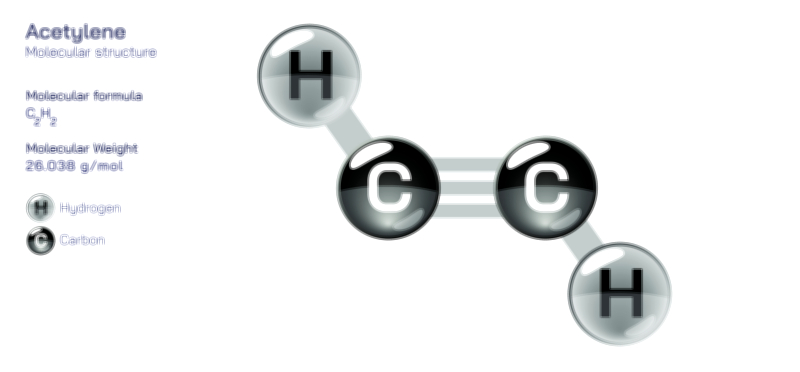
Aluminium Oxide — Chemical Structure and Industrial Compound Illustration
Fundamental Structure and Chemical Identity of Aluminium Oxide
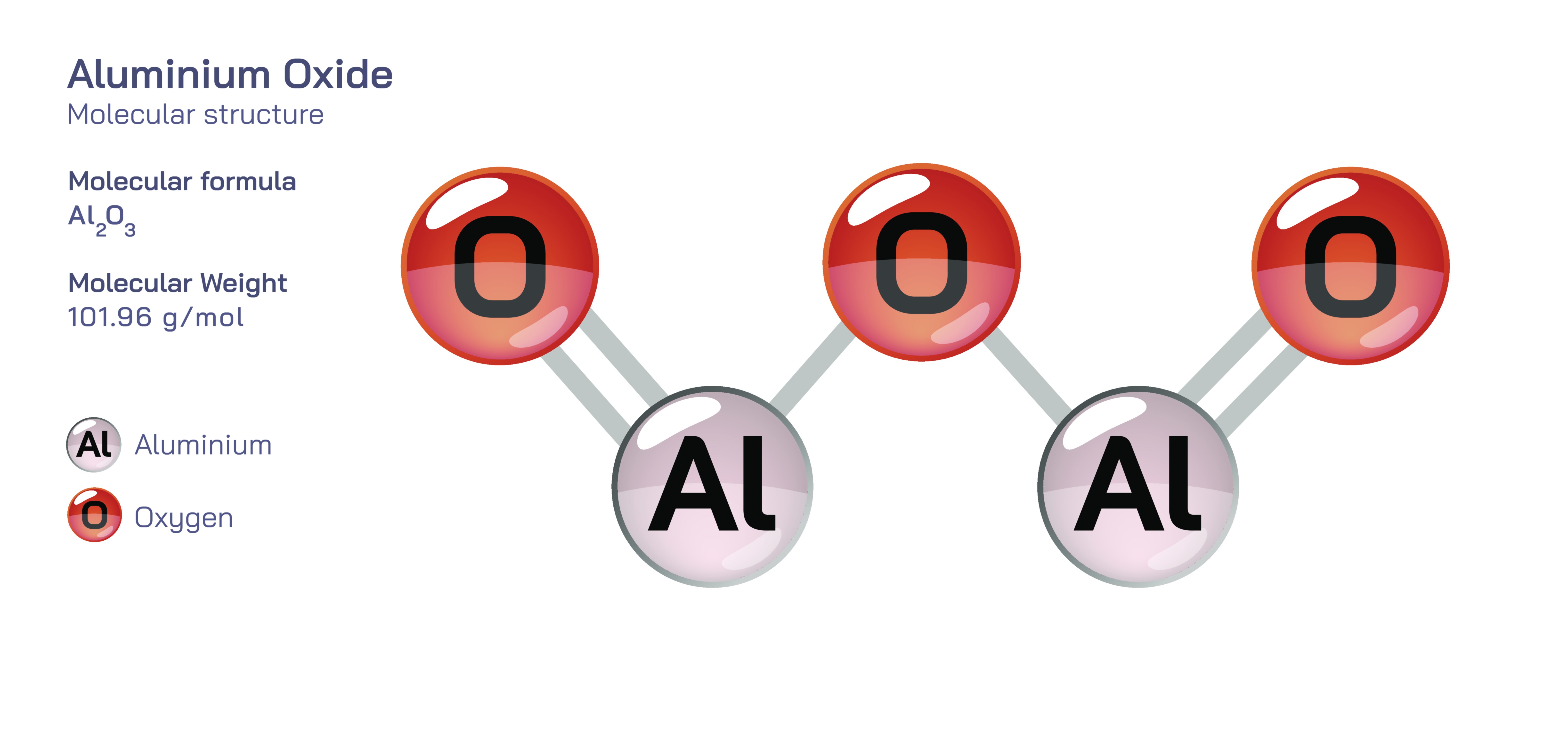
Aluminium oxide, commonly written as Al₂O₃ and widely known as alumina, is one of the most important inorganic compounds used across countless industrial, technological, and environmental applications. Structurally, it represents a robust oxide formed from aluminium in its +3 oxidation state, bonded to oxygen atoms through strong ionic and partially covalent interactions. In nature, aluminium oxide appears in multiple crystalline forms, with the most stable and well-known variant being corundum—a form so hard and durable that it is used as both an abrasive and a gemstone. The chemical simplicity of the Al₂O₃ formula belies the structural complexity of its crystalline arrangements, where aluminium ions nest within octahedral or tetrahedral pockets created by oxygen atoms. This network produces a lattice so strong and thermally stable that it resists melting even at temperatures exceeding 2000°C. Aluminium oxide plays a critical role as a foundational material for producing metallic aluminium, but its value extends far beyond metallurgy. It serves as a backbone for ceramics, insulators, catalysts, abrasives, refractories, and protective coatings, proving that its structural rigidity and chemical resilience make it one of the most indispensable materials in modern science and industry.
Chemical Bonding, Crystal Forms, and Structural Variations
The bonding within aluminium oxide blends ionic characteristics with significant covalent contributions due to the high charge density of the Al³⁺ ion. Oxygen’s ability to share electron density with aluminium creates a rigid, interconnected matrix in which each aluminium ion is surrounded by oxygen atoms arranged in a geometric framework dependent on the crystalline phase. The most common crystal structure of aluminium oxide is the corundum structure, in which aluminium ions occupy two-thirds of the available octahedral sites in a hexagonal close-packed array of oxygen atoms. This arrangement creates a lattice with exceptional hardness, second only to diamond among natural materials. Other polymorphs such as γ-alumina, η-alumina, δ-alumina, and θ-alumina form during thermal treatment or phase transitions, each possessing slightly different surface areas, pore structures, and catalytic properties. These metastable phases are crucial in industrial catalysis because of their large surface area and ability to host metal ions or catalytic particles. Across these polymorphs, the structure remains dominated by the strong attraction between aluminium and oxygen atoms, producing a compound noted for its stability, resistance to corrosion, and resilience under harsh chemical or thermal conditions.
Physical Properties and Their Functional Implications
The physical properties of aluminium oxide are a direct reflection of its structural strength and bonding patterns. It is an exceptionally hard, crystalline solid with extremely high melting and boiling points, making it ideal for environments involving intense heat or mechanical stress. Its hardness lends itself well to abrasive applications—sandpaper, grinding wheels, polishing compounds, and cutting tools often incorporate alumina to harness its ability to wear down other materials without degrading itself. The compound is electrically insulating yet thermally conductive, a rare combination that makes it invaluable in electronics where heat dissipation is necessary without electrical conduction. Its resistance to weathering, chemical attack, and corrosion enables its use as a protective coating on metals, especially aluminium itself. The thin oxide layer naturally formed on aluminium surfaces—sometimes only a few nanometers thick—prevents further oxidation, giving aluminium its characteristic durability and corrosion resistance. Aluminium oxide is also insoluble in water and most solvents, making it chemically inert under most environmental conditions. These physical traits illustrate how a single material can extend its usefulness from simple abrasion tasks to high-tech insulators, fuel cells, optical windows, and refractory linings.
Reactivity and Transformation Pathways in Industrial Processes
While aluminium oxide is remarkably stable, it participates in several essential chemical processes, especially at elevated temperatures or under highly reactive conditions. One of the most significant transformations is its reduction to metallic aluminium through the Hall–Héroult process. In this method, alumina is dissolved in molten cryolite and subjected to electrolysis, allowing aluminium ions to migrate to the cathode while oxygen is released at the anode. This process forms the backbone of modern aluminium production and relies on alumina’s ability to withstand extremely high temperatures without decomposing. Aluminium oxide also plays a role in petroleum refining, where its high surface-area polymorphs serve as catalytic supports for processes such as hydrodesulfurization, cracking, and reforming. In ceramics manufacturing, alumina undergoes sintering—high-temperature fusion of fine particles into solid structures—resulting in durable components used in engines, aerospace machinery, and medical implants. Alumina’s interaction with other metal oxides allows it to form spinels, specialized compounds used in high-strength ceramics and refractory materials. Even though chemically inert, aluminium oxide contains reactive surface sites that can interact with water, acids, or metal ions under controlled conditions, granting it chemical versatility in synthesis, adsorption, and purification tasks.
Industrial, Environmental, and Technological Applications
Aluminium oxide’s wide-ranging applications reflect its unmatched adaptability. In metallurgy, it is indispensable for extracting aluminium metal, one of the world’s most important structural materials. In ceramics, it forms the basis of spark plugs, cutting tools, furnace linings, and biomedical devices such as artificial joints due to its biocompatibility and wear resistance. As an abrasive, it shapes and polishes materials from metals to gemstones. In environmental applications, alumina acts as a filtration medium, removing impurities and contaminants from water or gases. Its nanoporous forms serve in chromatography, gas purification, and adsorption processes, where its capacity to bind molecules supports analytical and industrial purification. The optical transparency of sapphire—a crystalline form of aluminium oxide—makes it suitable for high-pressure windows, scratch-resistant screens, LED substrates, and infrared optical components. In electronics, alumina substrates support high-power circuits because they efficiently disperse heat while maintaining electrical insulation. Across every technological sector—from energy systems and transportation to manufacturing and healthcare—aluminium oxide remains a fundamental chemical compound that bridges raw materials with advanced engineering demands.
Illustrating the Structure, Function, and Behavior of Aluminium Oxide
Illustrations of aluminium oxide typically highlight its crystalline lattice, showing aluminium ions nestled in their octahedral or tetrahedral positions within a dense oxygen framework. Visual representations may depict the corundum structure as layers of tightly packed oxygen atoms with aluminium occupying interstitial sites, emphasizing the compound’s hardness and thermal resistance. Diagrams often show how alumina transitions between polymorphs when heated, or how surface area increases in catalytic forms such as γ-alumina. Industrial illustrations may demonstrate its role in the Hall–Héroult cell, highlighting alumina dissolution and ion migration during electrolysis. Other illustrations show its protective oxide layer on aluminium metal, helping to visualize why the metal resists corrosion so effectively. Through such imagery, the link between structure and function becomes clear, revealing how aluminium oxide’s internal arrangements govern its mechanical strength, chemical behavior, and industrial versatility.