Acetonitrile (Ethanenitrile) — Chemical Structure and Solvent Illustration
Molecular Identity and Structural Foundation of Acetonitrile
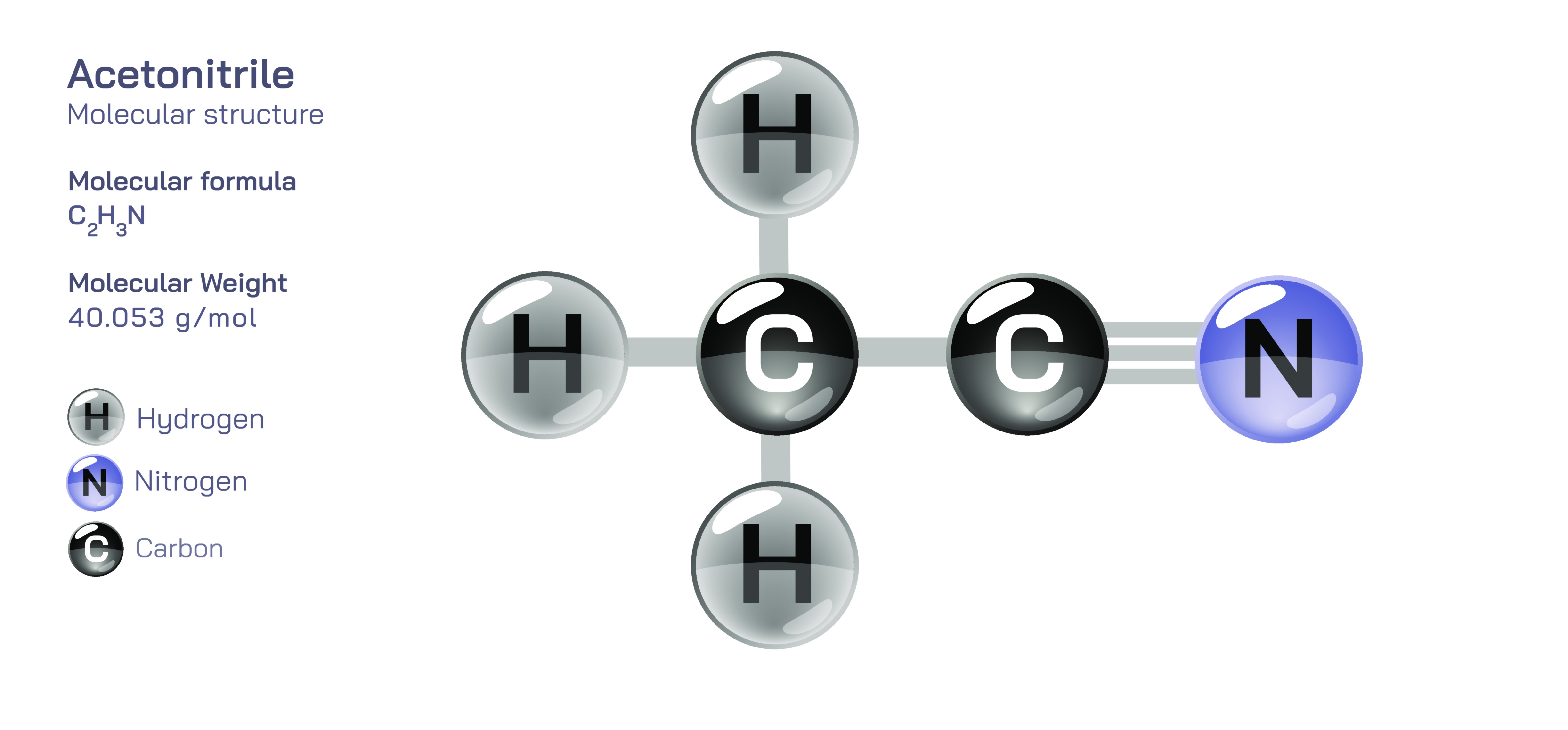
Acetonitrile, also known by its systematic name ethanenitrile, is one of the simplest organic nitriles, represented by the formula CH₃CN. Despite its small size and minimal structural complexity, it plays an outsized role in organic chemistry, analytical science, industrial synthesis, and biochemical research. The acetonitrile molecule consists of a methyl group attached to a nitrile functional group, forming a linear and highly polar structure that significantly influences its physical behavior and chemical reactivity. The nitrile group itself, featuring a carbon triple-bonded to a nitrogen atom, is the heart of the molecule’s identity. This carbon–nitrogen triple bond creates a rigid linear arrangement, giving the molecule a characteristic geometry that affects how it interacts with other molecules, solvents, and reactive species. The triple bond not only imparts substantial stability but also contributes to its strong dipole moment, making acetonitrile one of the most valuable solvents in situations where both polarity and chemical inertness are required. Understanding the molecular composition reveals why acetonitrile has become indispensable in chromatographic techniques, synthetic pathways, and industrial processing across many scientific fields.
Electronic Structure and Polarity of the Nitrile Group
At the core of acetonitrile’s chemical behavior lies the electronic configuration of the nitrile group. The carbon atom in the nitrile function adopts an sp-hybridized state, forming a linear geometry with the nitrogen. This hybridization leaves two unhybridized p orbitals that overlap with nitrogen’s orbitals to generate the triple bond, consisting of one sigma bond and two pi bonds. The nitrogen atom carries a significant portion of the electron density, while the carbon remains strongly electrophilic due to the polarity of the bond. This distribution of charge produces a pronounced dipole moment that makes acetonitrile highly effective at dissolving a wide range of polar and ionic compounds. The methyl group attached to the nitrile moderates some of the electron withdrawal but does not diminish the overall polarity of the molecule. The combination of structural rigidity, electron distribution, and dipole character results in a solvent capable of penetrating complex chemical matrices, stabilizing ions, and creating a uniform medium for controlled reactions. This electronic profile also helps explain why acetonitrile resists many types of chemical attack, allowing it to serve reliably in environments where other solvents would degrade or participate in unwanted reactions.
Physical Properties and Their Functional Significance
Acetonitrile exhibits a suite of physical properties that make it one of the most versatile solvents in modern chemistry. It is a colorless, volatile liquid with a relatively low viscosity and a distinctive, mildly ether-like odor. Its boiling point is moderate, allowing for easy removal through evaporation or distillation, while its low freezing point permits use in low-temperature reactions and specialized laboratory conditions. Its strong polarity, combined with a relatively low dielectric constant compared to water, enables it to dissolve a wide range of salts, organic compounds, and electrolytes used in electrochemistry. Importantly, acetonitrile is miscible with water, alcohols, and many other polar solvents, expanding its use across diverse applications. Its low UV absorbance makes it an indispensable component of high-performance liquid chromatography, where optical clarity is essential for accurate detection. Additionally, its low nucleophilicity and resistance to oxidation provide stability when working with sensitive reagents, electrophiles, and transition-metal catalysts. These physical traits collectively explain why acetonitrile is often selected when precision, reproducibility, and chemical neutrality are required.
Chemical Behavior and Reactivity Pathways
While acetonitrile is most widely recognized for its function as an inert solvent, it also participates in a variety of chemical transformations when used as a reagent. Under strongly basic conditions, the methyl group can undergo deprotonation to produce a reactive carbanion, enabling its use in alkylation and condensation reactions. The nitrile group itself can be converted into amines, amides, or carboxylic acids through reduction, hydration, or hydrolysis processes, respectively. Hydrogenation reduces acetonitrile to ethylamine, while catalytic hydration forms acetamide, and extended hydrolysis leads to acetic acid. These pathways demonstrate that acetonitrile can serve as a starting point for synthesizing more complex organic molecules. Its stability toward acids, bases, and oxidizing agents makes it particularly valuable as a medium for reactions requiring tight control, while its ability to coordinate weakly to metal centers grants it relevance in organometallic chemistry. Overall, acetonitrile’s reactivity profile embodies the duality of being both a stable solvent and a functional reagent capable of engaging in structured synthetic transformations.
Biological, Industrial, and Environmental Applications
In industrial contexts, acetonitrile is used in the production of pharmaceuticals, agricultural chemicals, perfumes, and synthetic fibers. Its ability to dissolve a broad spectrum of organic and inorganic compounds makes it essential in extraction processes, purification systems, and large-scale chemical manufacturing. In analytical chemistry, particularly in liquid chromatography and mass spectrometry, it is the solvent of choice because it provides sharp peak resolution, minimizes background noise, and allows precise control of mobile-phase gradients. In biological research, acetonitrile is employed to precipitate proteins, extract metabolites, and stabilize samples prior to analysis. Despite its usefulness, acetonitrile must be handled with care due to the toxic effects it can produce when metabolized, especially its conversion into hydrogen cyanide under extreme conditions. Environmentally, acetonitrile breaks down through microbial action and atmospheric chemical processes, and although it is not considered highly persistent, its presence in industrial waste requires proper management. Understanding these roles illustrates why acetonitrile is deeply integrated into modern scientific and industrial workflows.
Illustrating the Structure and Behavior of Acetonitrile
Scientific illustrations of acetonitrile typically emphasize the linear arrangement of the nitrile group, the electron distribution across the carbon–nitrogen triple bond, and the orientation of the methyl group relative to the nitrogen. Diagrams often highlight the dipole moment, showing how the negative charge density clusters around the nitrogen while the carbon remains electron-poor. These illustrations help explain how acetonitrile interacts with ions, dissolves salts, stabilizes transition states, and behaves under different reaction conditions. By visualizing the molecule’s geometry, polarity, and functional behavior, such illustrations enhance understanding of why acetonitrile acts as one of the most reliable and widely used solvents in modern chemistry.