Equilibrium and Non-Equilibrium — Understanding Balanced and Dynamic Systems
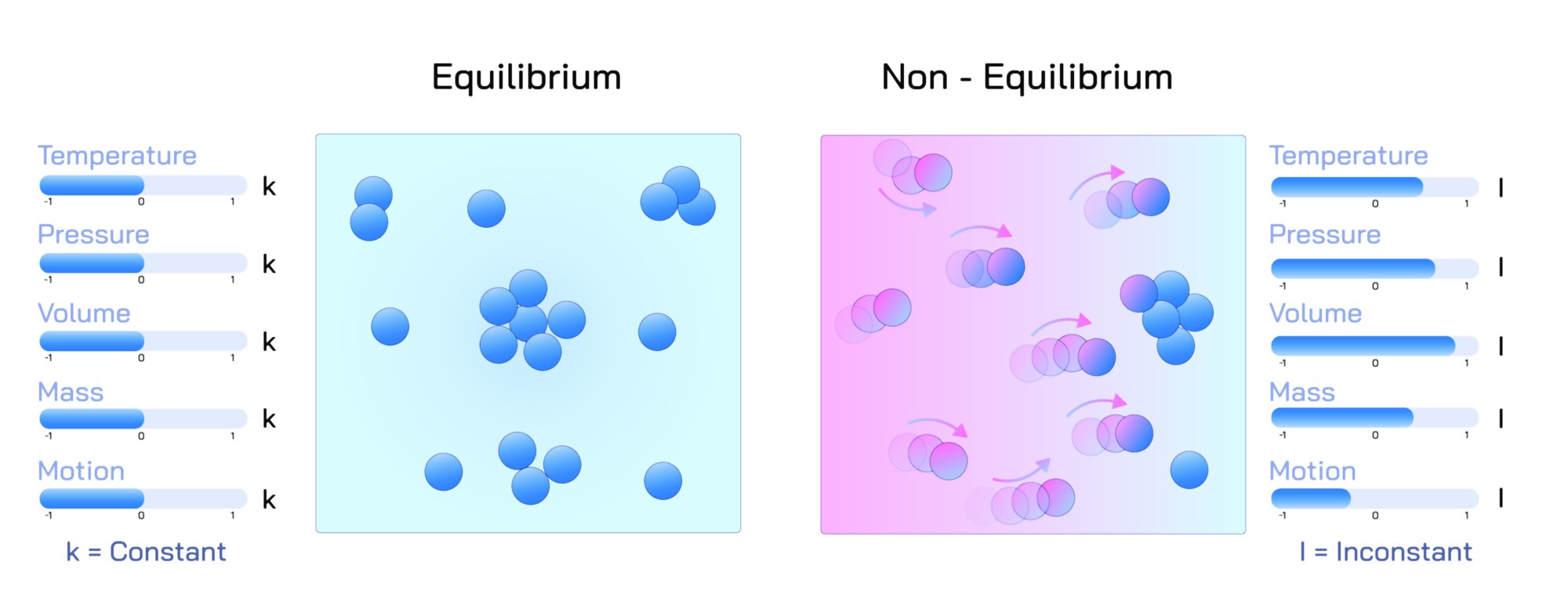
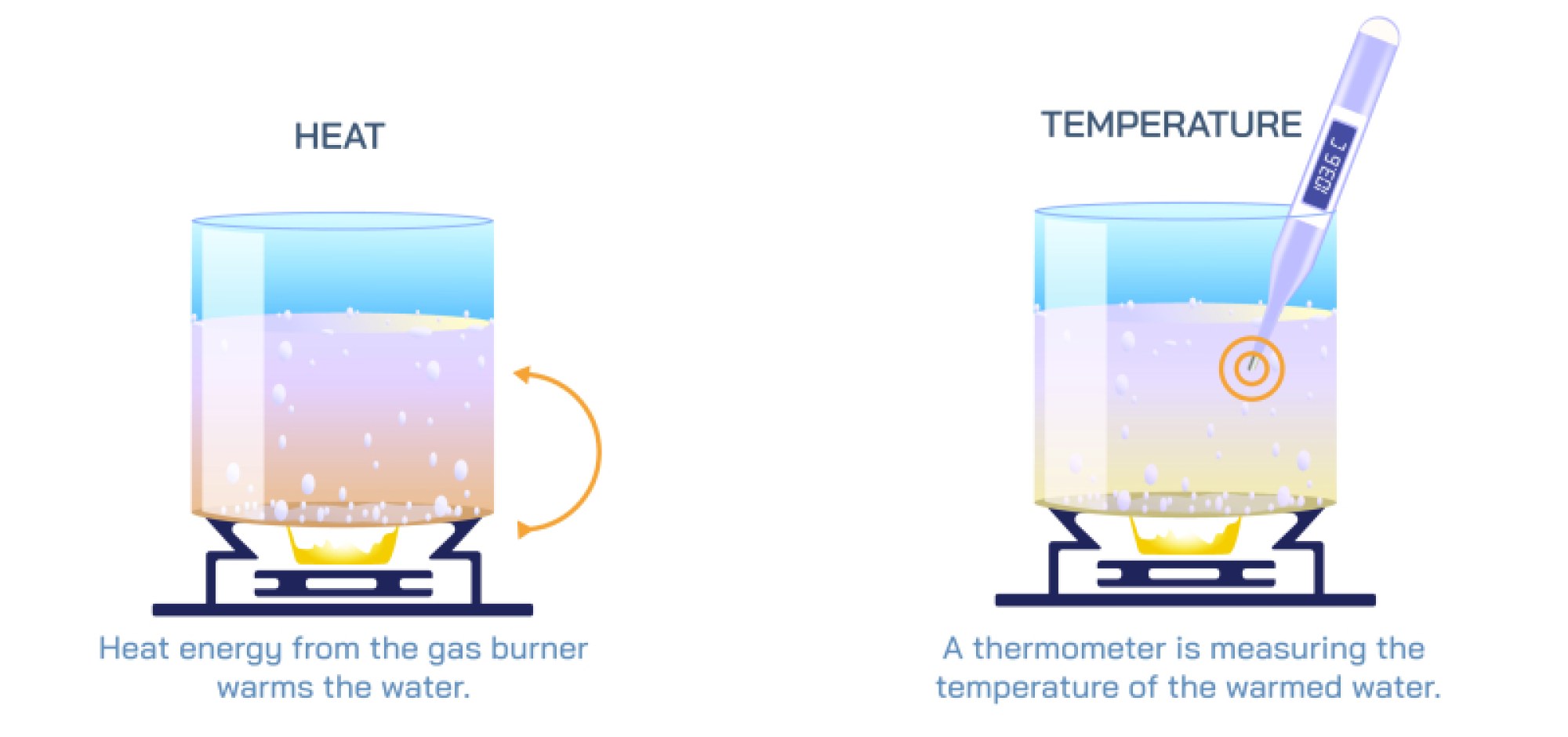
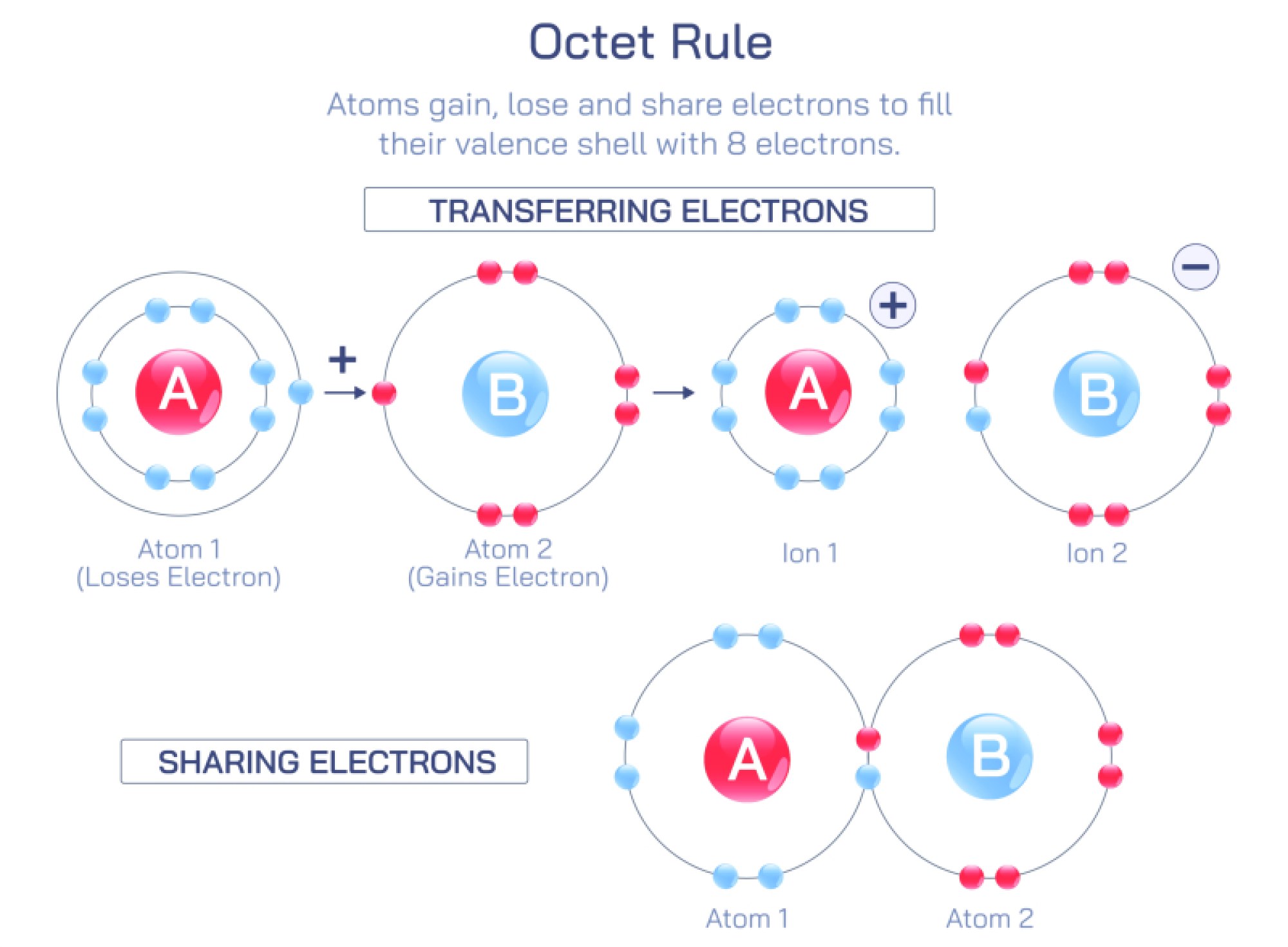
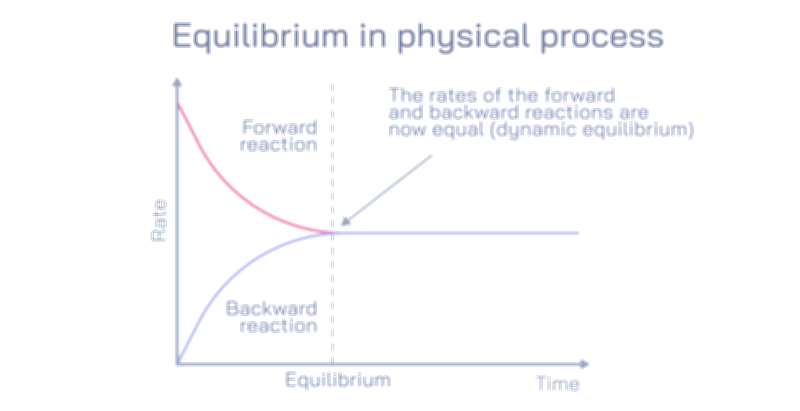
Equilibrium and non-equilibrium are fundamental concepts in physics, chemistry, and biology, describing the state of a system in terms of balance, stability, and change. An equilibrium state occurs when a system’s properties remain constant over time because the competing processes are balanced. In chemical equilibrium, for instance, the rate of the forward reaction equals the rate of the reverse reaction, resulting in no net change in the concentration of reactants and products. Physical equilibrium refers to situations like phase changes where energy transfer occurs without changing macroscopic properties, and biological equilibrium can describe homeostasis where organisms maintain internal stability despite environmental changes.
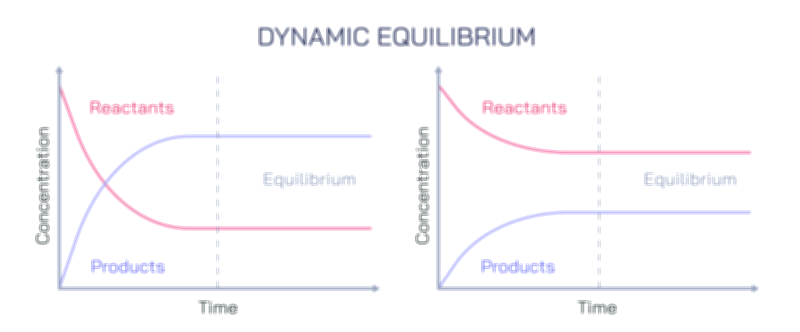
Dynamic equilibrium is a critical concept, emphasizing that while macroscopic properties remain stable, microscopic processes continue to occur. For example, in a saturated solution, dissolved particles continually move in and out of the solid phase, but the overall concentration remains constant. Equilibrium can also be classified based on its nature, such as thermodynamic equilibrium, mechanical equilibrium, or chemical equilibrium, each governed by specific laws and conditions, including temperature, pressure, and energy constraints. Systems at equilibrium are predictable and can be analyzed using mathematical models, allowing scientists and engineers to design chemical reactors, optimize industrial processes, and understand natural phenomena.
In contrast, a non-equilibrium state describes systems where properties change over time, as the processes within the system are not balanced. Non-equilibrium systems can exhibit complex behaviors such as oscillations, chaos, or transient dynamics. Examples include rapidly cooling molten metal, flowing fluids, living cells undergoing metabolic reactions, and atmospheric systems affected by changing energy inputs. Non-equilibrium thermodynamics studies how energy and matter flow through systems, providing insight into irreversible processes, transport phenomena, and the evolution of complex structures. These systems are less predictable but often exhibit fascinating patterns and self-organizing behavior that are central to understanding natural and engineered processes.
Understanding the differences between equilibrium and non-equilibrium is essential for applications across scientific disciplines. Engineers rely on equilibrium concepts to design efficient chemical reactors and industrial systems, while non-equilibrium studies help predict weather patterns, biological responses, and material behavior under dynamic conditions. Monitoring the transition between equilibrium and non-equilibrium states also informs safety measures, energy management, and system optimization. By analyzing how systems reach or deviate from equilibrium, researchers gain insight into stability, energy conservation, and the behavior of matter under diverse conditions.
In conclusion, equilibrium represents a stable, balanced state where competing processes cancel out, while non-equilibrium describes dynamic conditions with ongoing changes. Both concepts are vital in understanding natural, chemical, physical, and biological systems. Mastery of equilibrium and non-equilibrium principles enables scientists, engineers, and students to analyze system behavior, predict outcomes, and design processes that harness or control energy and matter effectively. Recognizing these states and their implications provides a foundation for advancements in chemistry, physics, biology, engineering, and environmental science.