Stages of the Combustion Process in a Candle Flame Explained
A burning candle displays one of the most elegant demonstrations of combustion, energy transformation, phase change, and heat-driven chemical processes, all happening simultaneously within a flickering flame that most people take for granted. Although the candle flame looks simple, it is an active laboratory in which wax molecules melt, vaporize, react with oxygen, and release heat and light through a self-sustaining cycle. Each stage of combustion is linked to the physical arrangement of the candle itself — a solid wax body, a woven wick, and an exposed flame — all of which work together through capillarity, evaporation, chemical reactivity, and convection. What appears to be a stationary and motionless flame is actually a dynamic region with constantly circulating gases, flowing molten wax, rising hot air, and newly formed reaction products. Understanding the stages of candle combustion shows how a renewable heating process allows burning to continue as long as fuel and oxygen remain available and how microscopic molecular events drive a visible flame that illuminates and warms a room.
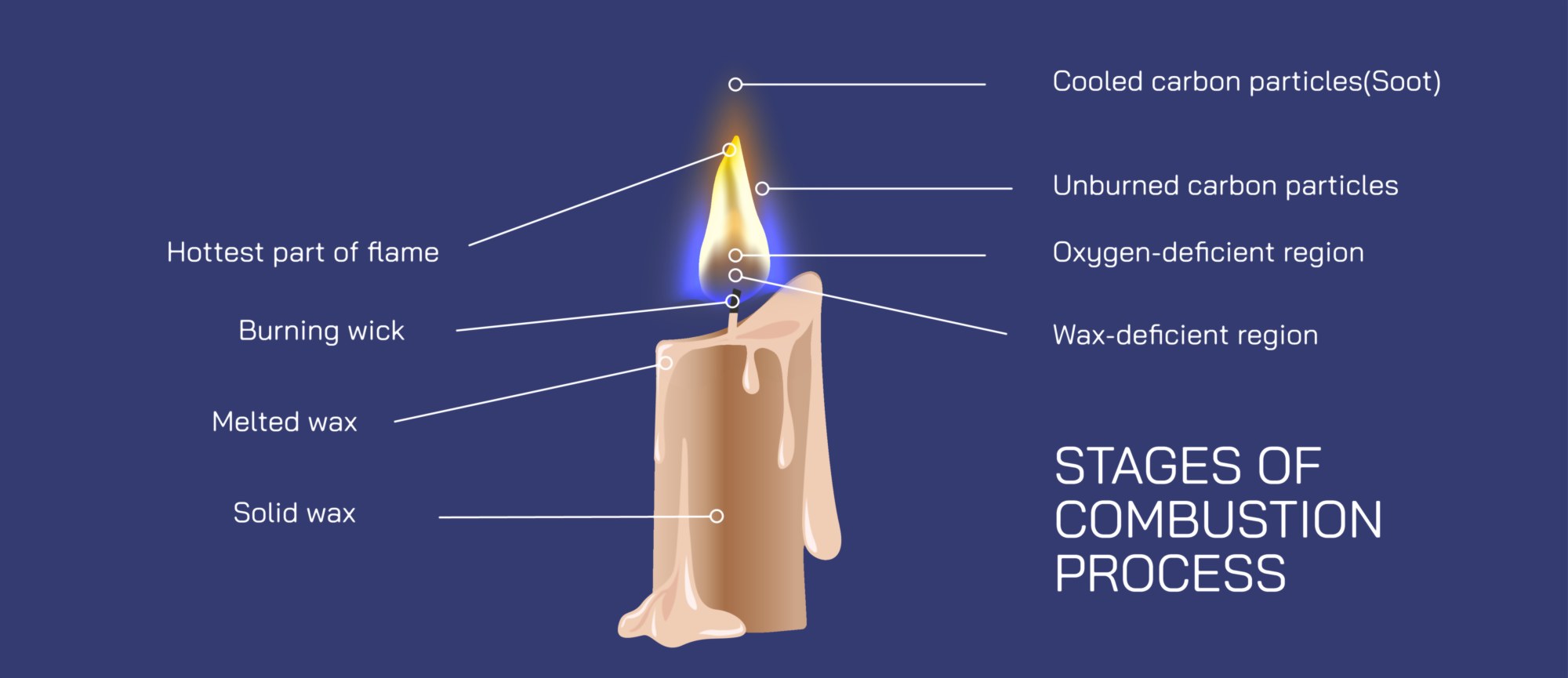
The combustion process begins the moment the wick is lit. At first, the flame does not feed on the wax itself; instead, it burns the wick’s flammable fibers coated with traces of paraffin and air deposits. As these initial fibers ignite, the flame produces enough heat to melt the solid wax near the base of the wick. This is the melting stage, in which heat travels by conduction through the wick and convection from the flame tip to the surrounding wax. Because candle wax is composed primarily of long-chain hydrocarbons — typically paraffin — it undergoes a physical phase change rather than immediate chemical decomposition, transforming from solid to liquid. The wick’s porous structure plays an essential role here. Through capillary action, liquid wax is drawn upward through the wick against gravity, forming a supply path that transports fuel to the burning zone. Without this transport mechanism, the flame would extinguish once the wick’s surface fuel was consumed, illustrating that the wick is not burned away so much as it serves as a fuel carrier.
The next stage of the combustion process is vaporization, the key step that allows the wax to become chemically reactive. As the liquid wax travels upward and encounters regions of higher temperature near the flame, it begins to vaporize into a hot gaseous mixture of hydrocarbon molecules. These wax vapors — not the liquid wax — are the true fuel of the flame. This stage is crucial because hydrocarbons in gas form mix with oxygen far more rapidly than in liquid or solid form. The vapor stream emerging from the wick enters the flame envelope, where oxygen rich air is continually fed by convection currents. At this point, the combustion becomes self-sustaining. Heat from the flame vaporizes more wax, vaporization fuels more combustion, and combustion produces more heat. As long as the cycle continues, the flame remains alive.
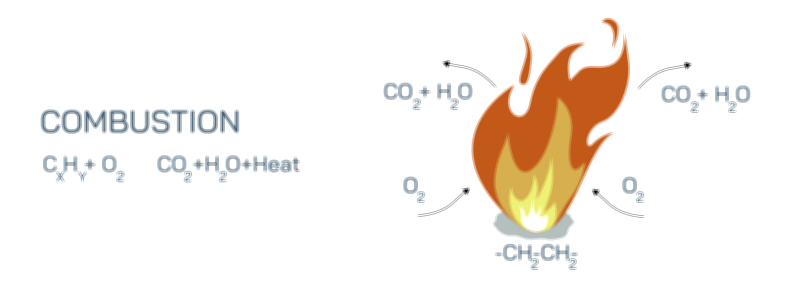
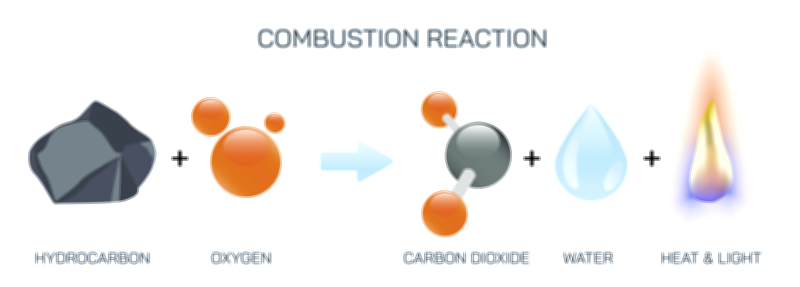
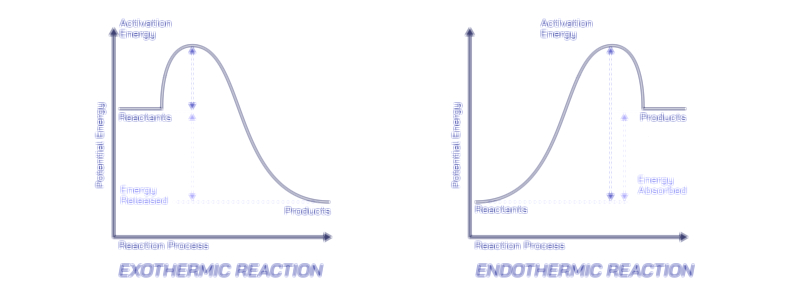
Once the wax has been vaporized and mixed with oxygen, the primary combustion stage begins. In this high-temperature zone surrounding the wick, hydrocarbon molecules break apart into fragments containing carbon and hydrogen, which rapidly react with oxygen in exothermic (heat-releasing) reactions. Hydrogen combines with oxygen to form water vapor, while carbon combines with oxygen to form carbon dioxide. The release of heat in turn drives further vaporization of wax and maintains flame temperature. However, not all combustion is perfectly complete, especially near the outer surface of the flame. In the hottest core, combustion is efficient and nearly smokeless, but toward the outer luminous region, tiny carbon particles form. These incandescent carbon particles heat up and glow, producing the familiar yellow-orange light of a candle flame. The color of the flame therefore reflects the temperature distribution within it: a nearly invisible blue zone near the base where full combustion occurs, and a bright yellow region above it where glowing carbon soot particles emit visible light.
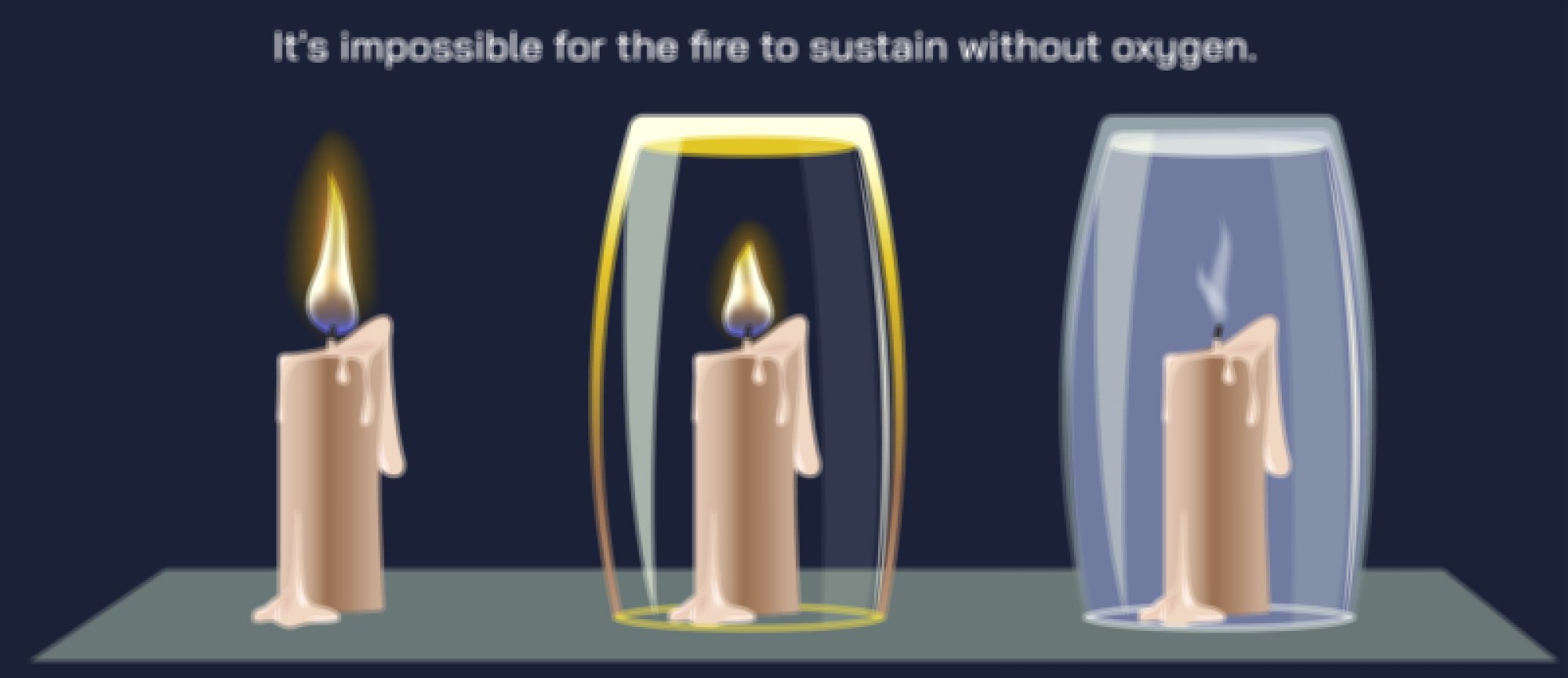
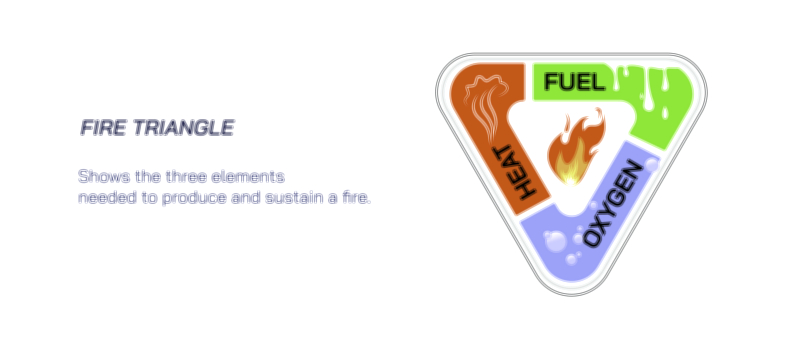
Surrounding the main combustion zone is the secondary combustion region, where oxygen concentrations are higher and partially-burned carbon particles undergo further oxidation. When the flame is undisturbed, hot gases rise upward in a teardrop-shaped envelope because convection continuously draws cool fresh air toward the base of the flame while pushing warm reaction products upward. This flow reinforces combustion by bringing oxygen into the lower part of the flame and removing heat and reaction products upward. If airflow becomes restricted — for example inside a jar — oxygen consumption occurs faster than oxygen can enter, reducing combustion efficiency. The flame dims, carbon particles accumulate, soot appears, and eventually the flame extinguishes as oxygen becomes insufficient to support chemical reactions. Extinguishing therefore happens not because fuel disappears, but because one of the three pillars of combustion — fuel, heat, and oxygen — is interrupted.
The final stage of the combustion cycle involves the formation and release of products. Water vapor and carbon dioxide rise from the flame as nearly invisible gases, while a trace amount of unburned carbon particles — soot — may escape when oxygen becomes slightly limited. The structure of the flame, including its shape, size, and brightness, constantly reflects the balance between fuel supply and oxygen availability. When the candle burns in still air, the flame settles into a stable structure in which the inward flow of oxygen, the upward flow of hot gases, and the vaporization of wax reach equilibrium. Disturbances such as a draft, increased airflow, or covering the flame disrupt this equilibrium and produce immediate visible changes in the flame.
The stages of a candle’s combustion — melting, wicking, vaporization, ignition of wax vapor, primary combustion, secondary combustion, and release of products — form a continuous loop that maintains the flame over time. Each step depends on precise thermal and fluid interactions rather than brute consumption of solid wax. The flame determines how fast the wax melts and how quickly fuel rises in the wick. The wick, in turn, regulates how much vaporized fuel reaches the flame. Even the flame’s size adjusts naturally: if more wax is melted than the flame can vaporize, excess liquid cools the wick, reducing vaporization and shrinking the flame until balance is restored. For this reason, a candle is a self-regulating burner, stabilizing itself through feedback between heat, fuel transport, vaporization, and combustion.
Seen through scientific understanding rather than casual observation, a candle supplies a beautifully complete lesson in physics and chemistry. It demonstrates how a solid fuel can sustain a gas-phase reaction, how heat moves through conduction and convection, how capillary action enables fuel delivery, how molecular structure determines light emission, and how equilibrium governs combustion. Far from being a static image, the soft yellow flame is a three-dimensional engine powered by chemical reactions that continually transform matter into light and heat. The quiet stability of a burning candle hides an extraordinary sequence of precisely balanced stages that allow a flame to come alive, sustain itself, and eventually extinguish when its fuel or air supply is gone — a glowing reminder of how energy cycles through matter in one of the simplest yet most captivating forms of combustion.