Why Fire Cannot Burn Without Oxygen: Understanding the Role of Oxygen in Combustion
Fire is one of the most fundamental chemical phenomena observed in daily life, yet it is often misunderstood in terms of its requirements and behavior. At its core, fire is the visible manifestation of a chemical reaction called combustion, which involves a fuel source reacting rapidly with an oxidizing agent, typically oxygen, to release heat, light, and combustion products such as carbon dioxide and water. Understanding why fire cannot burn without oxygen requires an exploration of the combustion process, the role of oxygen, and the chemical and physical principles that govern flame propagation. A vector illustration explaining this concept typically integrates fuel molecules, oxygen molecules, reaction pathways, and energy release, providing a clear and visually engaging depiction suitable for both scientific and educational purposes. By combining labeled components, directional arrows, and color-coded reaction zones, such illustrations allow learners to grasp both the necessity of oxygen and the dynamics of fire.
At the center of the illustration is a fuel source, which may be depicted as wood, paper, or a hydrocarbon-based substance. Labels identify the chemical composition of the fuel, such as cellulose in wood or carbon-hydrogen chains in hydrocarbons, emphasizing the presence of combustible atoms that can react with oxygen. Arrows indicate the breakdown of molecular bonds during heating, leading to the release of volatile gases that participate in combustion. Color coding differentiates the solid fuel, released gases, and surrounding environment, visually clarifying the reaction interface.
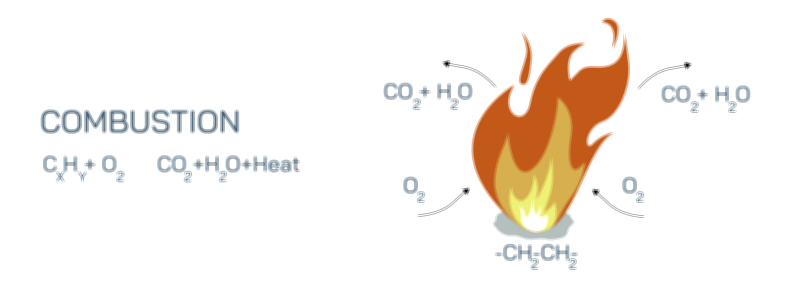
Oxygen’s role in combustion is depicted as molecular O₂ surrounding the fuel source. Labels highlight that oxygen acts as the oxidizing agent, accepting electrons from the fuel molecules during the exothermic reaction. Arrows show the movement of electrons and the formation of new chemical bonds between oxygen and fuel atoms, visually illustrating oxidation. In vector insets, individual oxygen atoms may be shown interacting with carbon and hydrogen atoms, forming CO₂ and H₂O molecules, which are the primary products of combustion. This molecular-level visualization emphasizes that without oxygen, these chemical transformations cannot occur, and therefore no fire can sustain itself.
The combustion reaction is illustrated with arrows pointing from reactants (fuel + oxygen) to products (carbon dioxide, water vapor, and heat). Labels indicate that energy is released as thermal energy and visible light, forming the flame. Color-coded gradients represent temperature zones in the flame, with blue at the base indicating high-temperature regions and yellow or orange at the tip representing cooler combustion zones. Arrows indicate heat propagation back to the fuel, which sustains the pyrolysis of additional fuel molecules and the ongoing reaction. Without oxygen, the arrows representing oxidation and heat feedback are absent, illustrating why the reaction ceases and the fire extinguishes.
Vector illustrations often include scenarios demonstrating the absence of oxygen, such as an enclosed chamber, submerged flame, or fire smothered by a blanket. Labels indicate that in these conditions, oxygen is depleted or prevented from reaching the fuel, halting combustion. Arrows are removed or grayed out in these panels to symbolize the interruption of electron transfer and molecular reactions. Magnified insets may show unreacted fuel molecules and the absence of oxidized products, visually reinforcing that oxygen is indispensable for sustaining fire.
Additional educational features may include comparative chemical reactions, showing fuels burning in oxygen-rich versus oxygen-poor environments. Arrows and labels may illustrate incomplete combustion in limited oxygen conditions, producing carbon monoxide or soot, versus complete combustion producing CO₂ and H₂O. This highlights the dual role of oxygen in not only sustaining fire but also ensuring efficient, clean energy release. Color coding may further distinguish between safe combustion and hazardous incomplete combustion products, linking chemical principles to practical consequences.
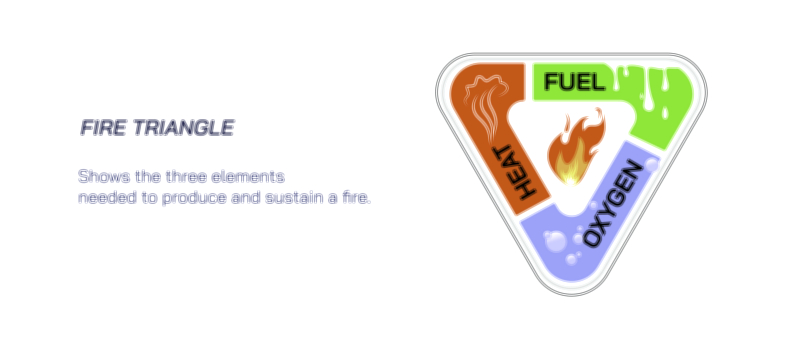
The illustration may also depict fire triangles, showing the three essential components for combustion: fuel, heat, and oxygen. Arrows and labels demonstrate that removal of any one component, particularly oxygen, disrupts the chemical reaction and extinguishes the flame. Insets may show oxygen’s role in initiating and maintaining free radical chain reactions, providing a molecular-level explanation of sustained combustion.
By combining fuel source, oxygen molecules, combustion reaction pathways, flame zones, and comparative oxygen-rich versus oxygen-depleted scenarios, a vector illustration explaining why fire cannot burn without oxygen provides a comprehensive and visually intuitive understanding. Color gradients, labeled molecules, directional arrows, and reaction products allow learners to grasp both the chemical necessity and physical manifestation of oxygen in sustaining fire.
Ultimately, understanding why fire cannot burn without oxygen demonstrates the essential role of oxygen as an oxidizing agent in combustion, linking molecular interactions to observable flame behavior. Through labeled fuel and oxygen molecules, reaction arrows, flame zones, and oxygen-deprivation scenarios, the diagram transforms a common natural phenomenon into an educational, visually engaging, and intuitive learning tool for chemistry students, physics learners, and general science education.