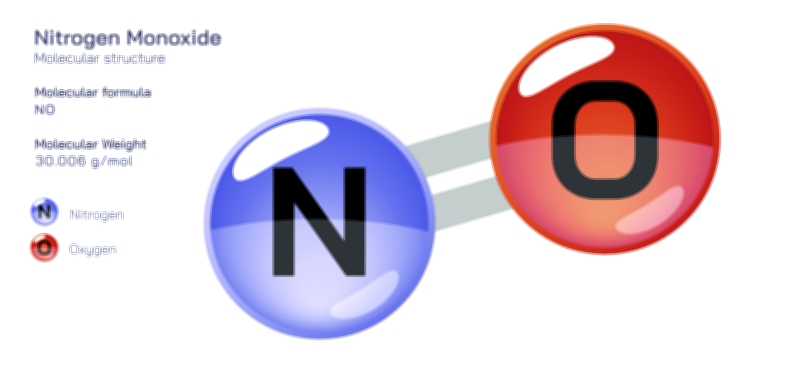
Nitrogen Dioxide — Molecular Structure and Environmental Impact
Nitrogen dioxide is a reactive gaseous compound that plays a crucial and far-reaching role in atmospheric chemistry, environmental pollution, industrial emissions, and public health. Although it appears in chemistry textbooks as a simple molecule composed of one nitrogen atom and two oxygen atoms, the true significance of nitrogen dioxide emerges from the way its structural arrangement enables it to participate in large-scale ecological and physiological effects. In a molecular vector illustration, nitrogen dioxide is typically depicted with a bent geometry rather than a linear shape, showing the nitrogen atom bonded to each oxygen atom with a mixture of single and double bond character. This shape arises from the presence of an unpaired electron on the nitrogen atom, giving the molecule paramagnetic properties and a highly reactive personality. The resonance structures drawn in such diagrams convey a dynamic distribution of electrons across the nitrogen–oxygen framework, which explains why nitrogen dioxide behaves both as an oxidizing agent and a participant in radical chain reactions in the atmosphere. Even though the structural diagram appears as just a cluster of atoms with curved lines and arrows, it represents a molecule that influences air quality, human health, climate processes, and the chemistry of Earth’s atmosphere on a global scale.
Nitrogen dioxide is most widely recognized as a major constituent of urban smog and industrial emissions, and its brownish-red color contributes to the distinctive haze that develops over congested cities. It is primarily produced during high-temperature combustion, especially in motor vehicle engines, coal-fired power plants, industrial boilers, and diesel generators. When nitrogen and oxygen in air are heated to extreme temperatures inside combustion chambers, nitrogen monoxide (NO) first forms and later reacts with atmospheric oxygen to produce nitrogen dioxide (NO₂). This transformation is the foundation of nitrogen oxide emissions collectively referred to as NOₓ, where nitrogen dioxide is the most visible and environmentally consequential component. Once released into the air, nitrogen dioxide disperses through urban and regional atmospheres, reacting with moisture, sunlight, organic compounds, and other pollutants. Its presence becomes not just a chemical event but a public health issue, because it enters the respiratory system with every breath taken by people living in dense and industrialized areas. The structural features that make nitrogen dioxide chemically reactive also determine its biological impact, because the molecule’s radical-like properties enable it to interfere with the delicate tissues of the lungs, contributing to inflammation, reduced lung function, and increased susceptibility to respiratory diseases.
One of the most significant environmental consequences of nitrogen dioxide lies in its role in the formation of ground-level ozone — not the protective ozone layer high in the stratosphere, but a harmful pollutant that forms in the lower atmosphere, directly affecting air quality and human well-being. In sunlight, nitrogen dioxide undergoes photodissociation, splitting into nitrogen monoxide and an oxygen radical. This reactive oxygen species quickly interacts with atmospheric oxygen molecules to produce ozone. Because the nitrogen monoxide formed then recombines with ozone to regenerate nitrogen dioxide, a catalytic cycle develops that continually creates and destroys ozone depending on the presence of hydrocarbons and volatile organic compounds (VOCs). When VOCs from vehicle exhaust, fuels, or industrial emissions enter this cycle, they trap nitrogen oxides in oxidation pathways that prevent ozone from being consumed, leading to an accumulation of ozone near the ground. This ozone peaks during the daytime in sunlit regions and becomes one of the key components of the photochemical smog associated with eye irritation, asthma attacks, lung inflammation, reduced crop yields, and damage to vegetation. In this sense, nitrogen dioxide does not simply coexist with ozone pollution — it enables the chemistry that makes ozone accumulate where it becomes toxic to living systems.
Nitrogen dioxide contributes additionally to the formation of fine particulate matter, particularly nitrate aerosols. In the atmosphere, nitrogen dioxide reacts with water vapor, oxidants, and ammonia to form nitric acid and ammonium nitrate particles. These particles fall into the PM₂.₅ category, referring to airborne solids and droplets smaller than 2.5 micrometers that penetrate deeply into the respiratory tract. Scientific studies link high PM₂.₅ exposure to cardiovascular disease, premature mortality, impaired child development, and systemic inflammation. The transformation of nitrogen dioxide into particulate pollutants illustrates that its impact extends far beyond the gas itself — it functions as a precursor that seeds additional pollutants capable of traveling long distances from their point of origin. Because ammonium nitrate particles can persist in the atmosphere and reflect sunlight, nitrogen dioxide indirectly influences climate processes by modifying atmospheric radiative balance. Though not a greenhouse gas in the conventional sense, its contributions to aerosols and ozone chemistry position it as a significant player in atmospheric warming, cooling, and altered cloud formation patterns.
Acid rain formation is another powerful pathway through which nitrogen dioxide affects the environment. When nitrogen dioxide dissolves into atmospheric moisture or reacts with oxidants, it produces nitric acid, which returns to the surface through rainfall, snow, or fog. Acid rain disrupts the buffering capacity of soils, decreases nutrient availability, damages forests, and acidifies lakes and rivers. Aquatic ecosystems exposed to prolonged acidification experience reduced biodiversity and weakened resilience, as sensitive fish, amphibians, and microorganisms struggle to survive in lowered pH conditions. In agricultural regions, acid rain can leach essential minerals from soil and impair crop productivity. When the nitrogen component of acid precipitation enters bodies of water, it also contributes to nutrient overloading and eutrophication, encouraging excessive algal growth that depletes oxygen and suffocates aquatic life. Whether affecting soils, forests, lakes, or oceans, the environmental chanǵes initiated by nitrogen dioxide emerge from the same structural capability: the molecule readily oxidizes into nitric acid because its nitrogen center has an electron-deficient configuration that accepts additional oxygen and hydrogen in aqueous environments.
Nitrogen dioxide also interacts with the nitrogen cycle, connecting industrial pollution to biological nutrient flows. Although nitrogen is an essential fertilizer element, excess atmospheric deposition of nitrogen products generated from NO₂ disruptions can upset the balanced chemistry of natural ecosystems. Coastal and marine regions receiving nitrogen runoff — whether from agriculture or atmospheric deposition — experience harmful algal blooms, oxygen deprivation, and aquatic dead zones. In some cases, nitrogen deposition forces a shift in plant species composition in terrestrial ecosystems by favoring fast-growing nitrogen-loving plants at the expense of slow-growing species adapted to low-nutrient soils. The resulting decline in plant diversity affects insects, birds, and entire food webs. Nitrogen dioxide did not evolve as a molecule designed to cause ecological disruption, but human-driven emissions have altered its natural place in environmental chemistry, magnifying its presence beyond the levels that ecosystems are equipped to process.
Public health implications of nitrogen dioxide go beyond chemical irritation and physical pollution. Long-term exposure to even moderate concentrations has been associated with increased hospitalization rates for asthma, chronic obstructive pulmonary disease, and cardiovascular conditions. Children and older adults are especially vulnerable because developing respiratory systems and weakened immunity respond more dramatically to airborne oxidative stress. Nitrogen dioxide increases susceptibility to respiratory infections by impairing mucosal defenses and triggering inflammatory responses in lung tissue. Studies conducted in traffic-dense urban areas consistently show that proximity to emission sources correlates with elevated NO₂ exposure and poorer health outcomes, underscoring the inequities in pollution burdens between regions with differing industrial and transportation patterns. Because nitrogen dioxide serves as both a direct pollutant and a precursor to more complex pollution chemistry, its reduction is one of the most effective and immediate strategies for improving air quality and public health in polluted areas.
Despite its negative environmental impacts, nitrogen dioxide is not intrinsically harmful in scientific and industrial contexts when managed responsibly. In controlled laboratory settings, it is an important reagent for nitrate synthesis and oxidation reactions. In the upper atmosphere, nitrogen dioxide plays roles in natural nitrogen cycling driven by lightning and microbial activity. The molecule itself is not a pollutant by choice — it becomes one when its concentration in the air exceeds the capacity of the Earth’s atmosphere and ecosystems to absorb and transform it. Solutions therefore focus not on eliminating nitrogen dioxide from all chemical environments but on controlling its production from anthropogenic sources. This involves improving combustion efficiency, transitioning from fossil fuels to renewable energy, controlling industrial emissions, optimizing catalytic converters in transportation, redesigning urban mobility systems, and shifting manufacturing practices to minimize NOₓ formation. Scientific advances in atmospheric modeling and satellite-based monitoring now allow governments and environmental agencies to track nitrogen dioxide globally and evaluate the success of air-quality policies in real time.
The story of nitrogen dioxide is the story of how a relatively simple molecule can influence the functioning of modern civilization and natural ecosystems simultaneously. Its bent molecular structure, paramagnetic nature, resonance distribution, and redox flexibility make it a key agent in atmospheric reactions that shape air quality, climate processes, ecological health, and human well-being. A vector illustration of nitrogen dioxide may appear as a modest configuration of one nitrogen atom and two oxygen atoms, but embedded in that illustration is the chemical foundation for smog formation, ozone chemistry, particulate pollution, acid rain development, nitrogen cycling, and respiratory risk. Understanding the structure of nitrogen dioxide is therefore not an academic exercise but a gateway to recognizing how industrial activities intertwine with atmospheric chemistry and environmental sustainability. Through the lens of this single molecule, it becomes clear that managing air quality is not merely about reducing emissions but about preserving the chemical balance that supports life on Earth.