Combustion Reaction Showing Chemical Equation, Energy Release, and Oxygen–Fuel Interaction Process
Combustion is one of the most widely recognized and scientifically significant chemical reactions, serving as the basis for fire, heat production, and much of the world’s energy technology. At its core, a combustion reaction represents the rapid chemical combination of a fuel with oxygen, producing new substances while releasing energy in the form of heat and often light. Although flames and heat are the visible signatures of combustion, the true transformation occurs at the microscopic level, where molecules break apart and rearrange into new structures. A vector illustration showing combustion depicts this phenomenon by highlighting the interaction between fuel and oxygen molecules, the symbolic chemical equation that summarizes the transformation, and the energetic burst that results from bond breaking and bond formation. Together, these elements visualize combustion not only as an energetic display but also as a strictly governed chemical process rooted in the laws of thermodynamics.
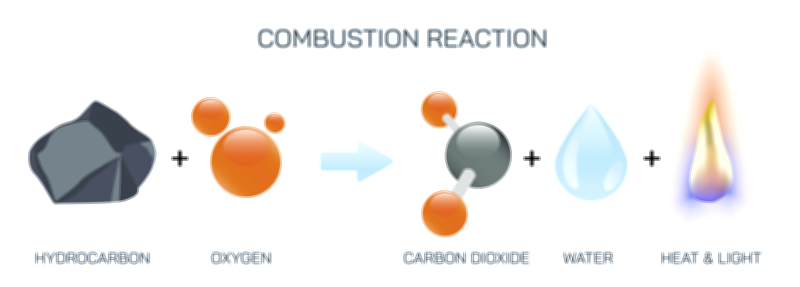
Combustion always follows a predictable pattern: a fuel rich in carbon and hydrogen reacts with oxygen from the atmosphere to produce carbon dioxide, water vapor, and energy. This simplified structure captures the essence of the reaction whether the fuel is wood, gasoline, ethanol, natural gas, kerosene, propane, or any other combustible hydrocarbon. The process begins when energy is supplied to the fuel—often through heat or a spark—initiating the breaking of chemical bonds within the fuel molecules. Once enough energy is provided to overcome the activation barrier, the exothermic chain reaction continues independently, as the energy released by one stage sustains the next. In visual terms, the start of combustion is shown not just as the ignition flame but as the structural weakening of molecular bonds and the merging of fuel and oxygen molecules, each step driving the next in rapid succession.
Although a combustion diagram may show a general fuel molecule, one of the clearest representations uses methane, a simple hydrocarbon, as an example. When methane reacts with oxygen, the rearrangement of atoms follows the pattern:
CH₄ + 2O₂ → CO₂ + 2H₂O + energy
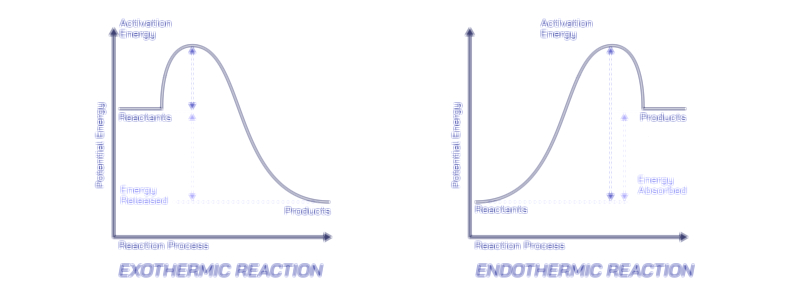
This equation represents the universal structure of complete combustion. The carbon atom in the fuel ultimately binds with two oxygen atoms to form carbon dioxide, while the hydrogen atoms bond with oxygen to form water. What the equation does not visually show—but a vector illustration can—are the energy changes that accompany the reaction. The breaking of bonds in the fuel requires energy input, while the formation of new bonds in carbon dioxide and water releases far more energy than was initially absorbed. This net release of energy is the hallmark of combustion, the reason fires emit heat and flames shine brightly. The redirection of stored chemical energy into thermal energy becomes the physical output that powers engines, heaters, power plants, and even the metabolic processes of living organisms.
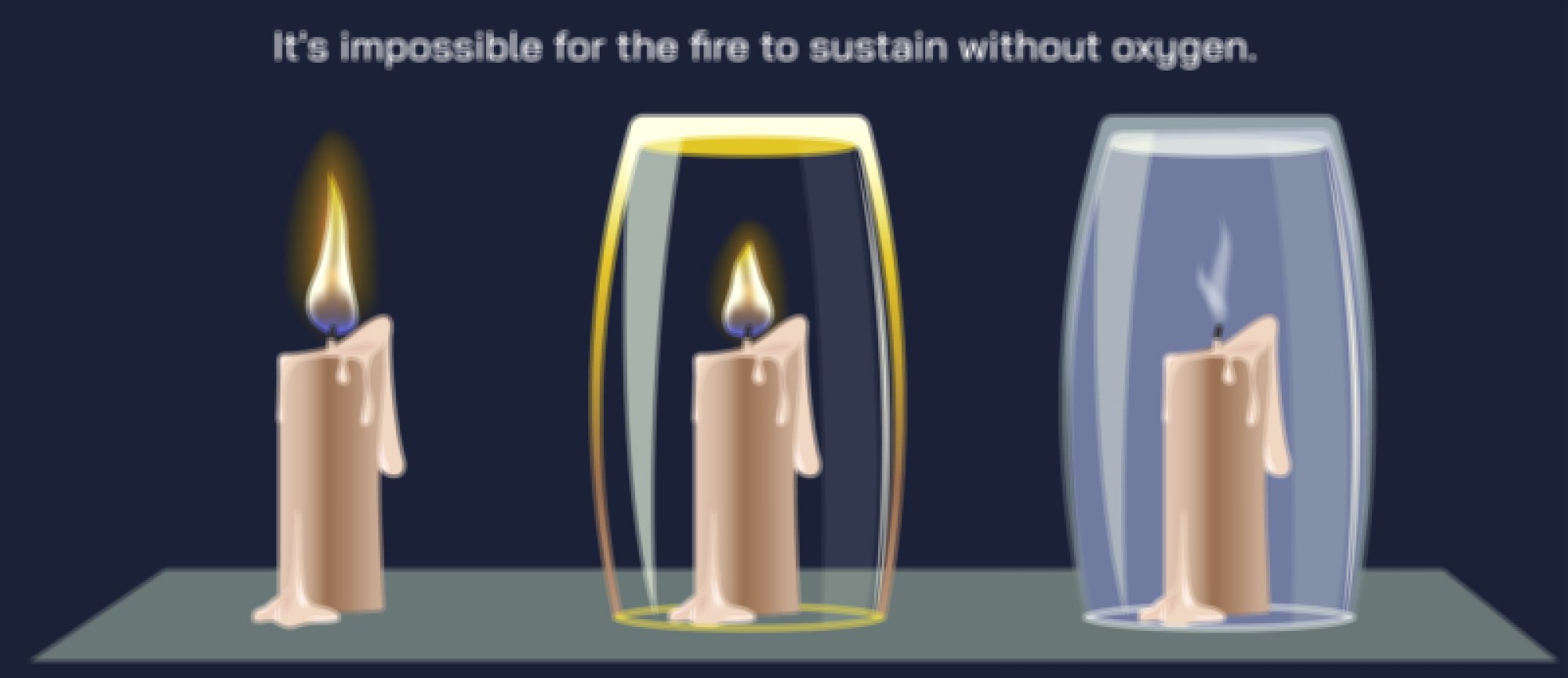
The rate and intensity of combustion depend heavily on the oxygen–fuel interaction. Combustion cannot proceed without oxygen—in fact, reducing the oxygen supply slows the reaction, producing incomplete combustion that forms soot (carbon particles) and carbon monoxide instead of carbon dioxide. When oxygen is abundant, the flame burns clean and hot; when limited, combustion becomes cooler and less efficient. In illustrations, this contrast is often shown using two reaction pathways: complete combustion leading to clear gas products and incomplete combustion producing smoke and harmful byproducts. This visualization underscores how oxygen availability shapes the outcome of combustion and why proper air supply is critical in everything from gas engines and furnaces to campfires and rocket combustion chambers.
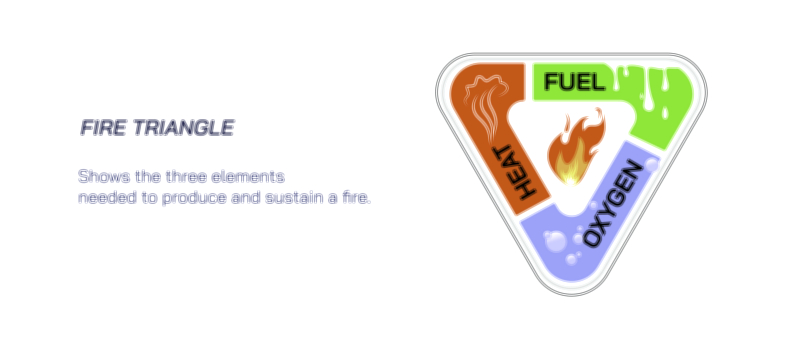
At the molecular level, the interaction between fuel and oxygen occurs through a series of intermediate radicals that propagate the reaction chain. Although these radicals form and disappear within fractions of a second, they represent the heart of the combustion mechanism. In a flame, millions of bond rearrangements happen each moment as carbon–hydrogen bonds rupture, oxygen bonds break, and new carbon–oxygen and hydrogen–oxygen bonds form. The continuous release of heat accelerates molecular motion, drawing more oxygen toward the fuel surface and stimulating further collisions. This chain reaction explains why fire spreads on its own once ignition begins. It also explains how removing heat, oxygen, or fuel — the three sides of the fire triangle — will extinguish a flame by halting at least one part of the chain.
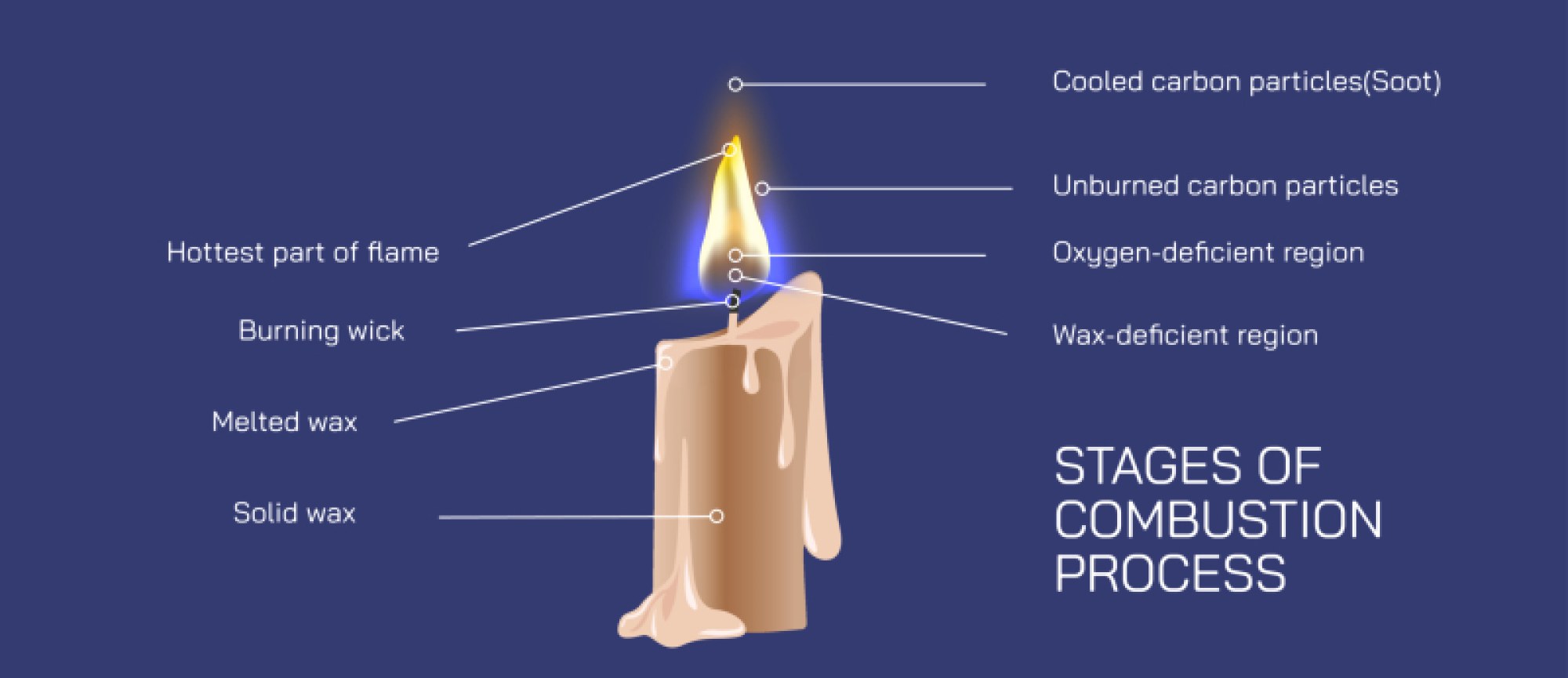
The release of large amounts of energy during combustion generates effects familiar in daily life. In controlled environments such as car engines or power plants, energy is harnessed to perform mechanical work or generate electricity. In household heating, the energy warms air or water. In open flames, it radiates outward as light and heat. The orange-yellow glow of many flames arises from incandescent carbon particles heated to high temperatures, while blue flames indicate more complete combustion with stronger heat output. Smoke color and flame shape reveal valuable diagnostic information about combustion efficiency, fuel type, and airflow — insights routinely used in engineering, fire science, and safety systems.
In addition to heat and light, combustion produces gases that influence environmental and safety considerations. While carbon dioxide and water are the primary products of complete combustion, real-world systems often produce nitrogen oxides, sulfur dioxide, ash, and unburned hydrocarbons depending on fuel composition. Understanding combustion chemistry therefore becomes essential not only for maximizing energy efficiency but also for minimizing pollution and reducing risk. Modern systems use catalytic converters, oxygen sensors, optimized burner design, and precise fuel–air control to approach clean combustion while avoiding incomplete burning. These improvements maintain the core chemical structure of combustion while managing its byproducts responsibly.
The visual representation of combustion in educational illustrations highlights this entire journey: oxygen molecules approaching hydrocarbon fuel, the rearrangement of atoms during ignition, and the formation of carbon dioxide and water alongside bursting energy. Heat waves, flames, sparks, and glowing bonds symbolize the exothermic nature of the reaction. Whether simplified for students or detailed for professionals, such diagrams translate the symbolic chemical equation into a vivid picture of molecular transformation.
Ultimately, a combustion reaction is much more than the appearance of fire—it is the conversion of stored chemical energy into usable thermal energy through the systematic restructuring of molecular bonds. It links microscopic atomic interactions to large-scale physical results: warmth, movement, power, illumination, and sometimes destruction. The chemical equation captures the balance of reactants and products, while oxygen and fuel dynamics determine the reaction’s efficiency, and energy release forms the driving outcome that gives combustion its practical value. Through illustration, chemistry and physics converge, offering a clear window into one of the most influential reactions shaping both human society and natural processes across the planet.