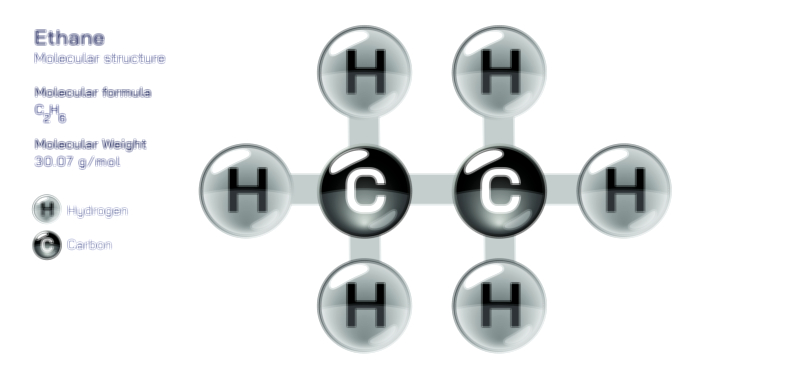
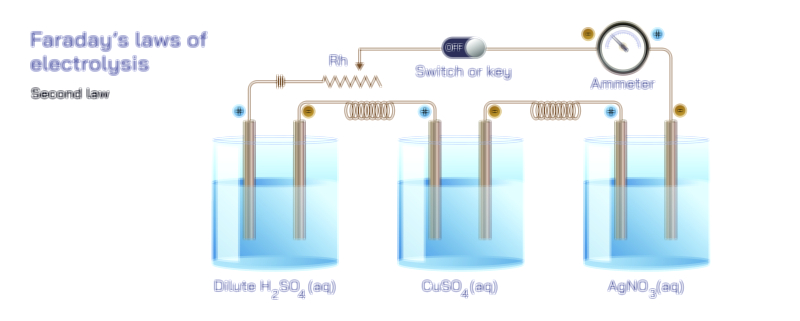
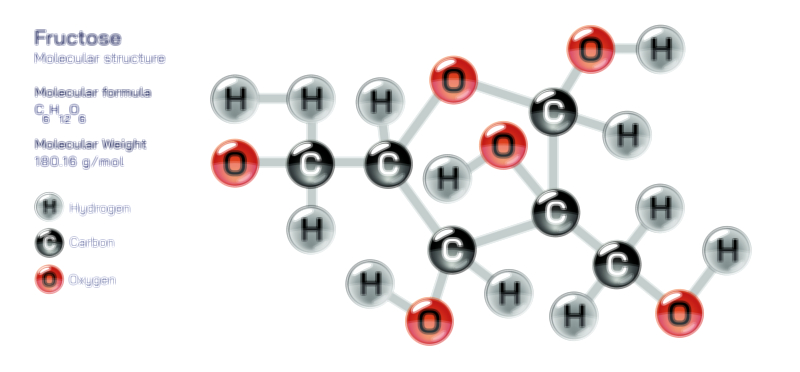
Exo vs Endo Reaction — Understanding Exothermic and Endothermic Processes
Exothermic and endothermic reactions represent two fundamental ways in which energy flows during chemical and physical transformations, shaping everything from everyday experiences to the behavior of complex industrial systems. Although the differences between them are often illustrated using simple arrows or vector diagrams, the underlying concepts involve deep insights into how matter interacts, how bonds form and break, and how energy disperses or concentrates within a system. Exploring the nature of exothermic and endothermic processes through rich descriptive explanation reveals why reactions release or absorb heat, how these energy exchanges influence the environment, why energy profiles differ, and how these processes are essential to biological life, environmental cycles, and technological applications. Even without formulas or graphical plots, the concepts become vivid when explained in detailed narrative form, allowing the idea of energy transfer to emerge as a central theme in the story of chemical change.
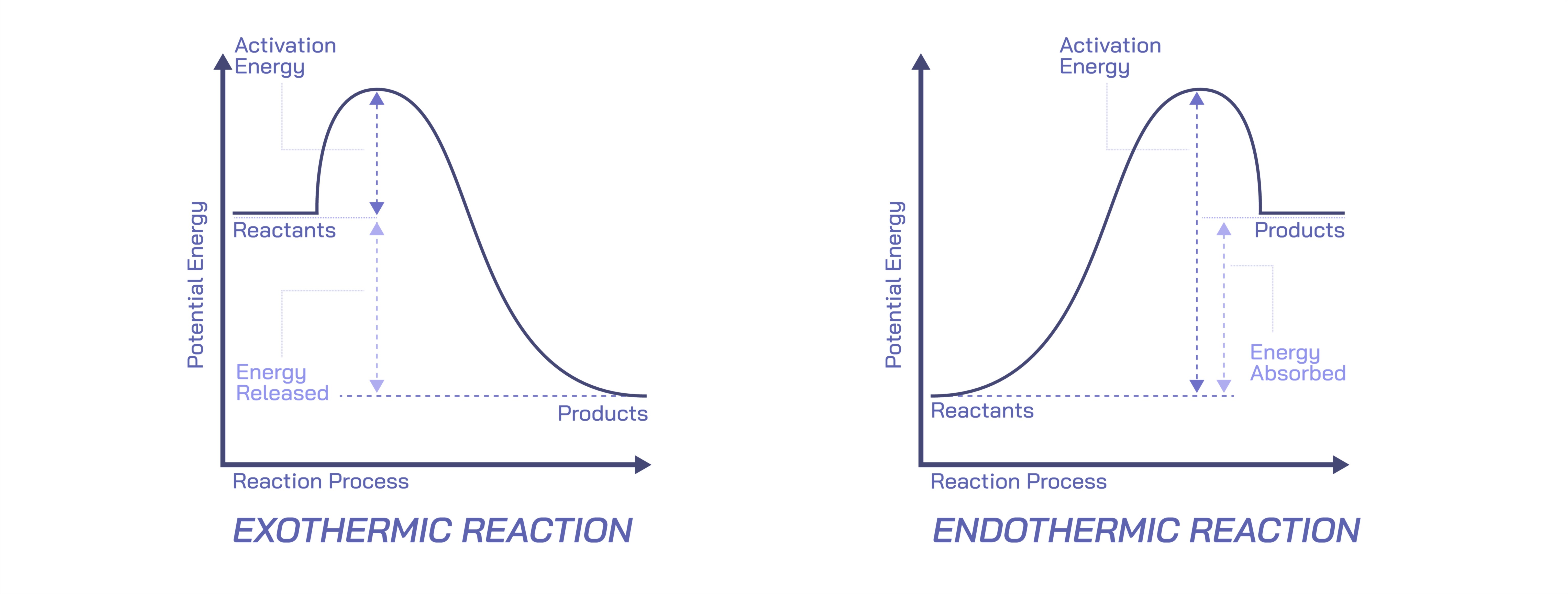
An exothermic reaction is one in which energy is released into the surroundings, usually in the form of heat but sometimes as light, sound, or other forms of energy. This release of energy occurs because the products of the reaction are lower in internal energy than the reactants. In other words, when new bonds form in the products, they release more energy than was required to break the bonds in the reactants. The system loses energy to the environment, resulting in an increase in temperature or a warming effect. This behavior is familiar in countless everyday phenomena: the heat felt during combustion, the warmth generated by rusting iron, the energetic release from fireworks, and even the mild heat produced when certain salts dissolve in water. Each of these examples illustrates how exothermic reactions transfer energy outward, influencing their surroundings through noticeable temperature changes or dynamic physical effects.
By contrast, an endothermic reaction absorbs energy from the surroundings. This absorption occurs because the reactants require more energy to break their bonds than is released when new bonds form in the products. As a result, the system becomes less energetic and must draw energy from its environment to proceed. This energy input often produces a cooling effect, as the surroundings lose heat to the reaction. Everyday examples of endothermic processes include the melting of ice, the evaporation of water, the dissolving of certain salts that lower solution temperature, and the thermal decomposition of calcium carbonate in lime kilns. These processes illustrate the nature of energy absorption: the surroundings lose heat, causing a noticeable drop in temperature, which demonstrates that the system is taking in energy to support the transformation occurring at the molecular level.
Despite these differences, both exothermic and endothermic processes share a common foundation: energy changes result from the rearrangement of matter at the microscopic scale. Bonds between atoms and molecules contain stored energy, and when these bonds break and form, energy transfers into or out of the system. Exothermic reactions release energy because the products form stronger bonds than those broken, reflecting a shift toward more stable molecular arrangements. Endothermic reactions require energy because the products either contain weaker bonds or exist in states that demand greater molecular freedom or separation. Understanding these shifts in bonding strength clarifies why energy transfer during reactions is not arbitrary but directly tied to the structure and interactions of the molecules involved.
In exothermic reactions, the energy release often manifests as a sudden increase in temperature. The warmth felt when lighting a candle, the intense heat produced when gasoline burns in an engine, and the glow emitted by a burning log all arise from exothermic processes. These reactions often proceed rapidly once initiated because the energy released helps sustain the reaction. For instance, combustion reactions can become self-propagating because the heat generated by one stage of the reaction ignites the next. This self-sustaining nature makes exothermic reactions powerful and sometimes dangerous, especially when fuels or reactive chemicals are involved. However, it also makes them incredibly useful, allowing humans to generate heat, light, motion, and electricity through controlled exothermic processes such as combustion engines, power plants, and heating systems.
Endothermic reactions, on the other hand, often feel cold to the touch because they draw heat inward. When ammonium nitrate dissolves in water, for example, the container becomes noticeably cold as heat flows from your skin into the reaction mixture. The melting of ice is another everyday endothermic process. Although it appears passive, melting requires energy to break the orderly lattice structure of solid water, absorbing heat from the surroundings until the solid becomes liquid. In industrial contexts, endothermic reactions play essential roles in processes such as metal extraction, chemical decomposition, and certain types of material synthesis. These reactions often require external heating sources to maintain conditions necessary for chemical change, reflecting how energy input is essential to drive systems from one structural state to another.
The environmental and biological significance of these processes cannot be overstated. Many natural cycles depend on exothermic and endothermic transformations. Photosynthesis, for example, is an endothermic process that absorbs energy from sunlight to convert carbon dioxide and water into glucose and oxygen. Without this massive energy absorption occurring globally, ecosystems would collapse and food chains would fail. Conversely, cellular respiration in living organisms is largely exothermic. It breaks down glucose to release energy that fuels biological activity. This delicate balance between absorbing and releasing energy forms the foundation of life. Organisms rely on the cycle of energy flow between endothermic and exothermic processes to maintain metabolism, growth, and sustainability.
In atmospheric and geological systems, these processes play equally important roles. The evaporation of water—an endothermic process—absorbs tremendous amounts of heat from Earth’s surface, helping regulate temperature and drive weather patterns. When this water vapor eventually condenses into clouds or precipitation, the process reverses and becomes exothermic, releasing heat into the atmosphere and influencing storm development. Volcanoes, earthquakes, and rock formation also depend on exothermic and endothermic transformations deep within Earth’s crust, where energy absorption and release govern melting, solidification, and mineral changes. These large-scale examples demonstrate how the principles behind simple experimental reactions extend into the behavior of entire planetary systems.
Industrial processes harness exothermic and endothermic reactions strategically. Exothermic processes are used to generate heat or energy, such as in the formation of concrete, which warms as it cures due to energy release. Chemical plants design reactors that can safely handle exothermic behavior, often using heat exchangers to capture and repurpose the energy for other stages of production. Endothermic reactions are equally essential. Processes such as cracking petroleum molecules, refining metals from ores, producing ceramics, and manufacturing fertilizers all depend on controlled energy absorption. Industrial facilities apply heat through furnaces, kilns, or electrical systems to ensure these reactions proceed efficiently. The design of such systems must account for the continuous energy demands of endothermic transformations to maintain productivity and safety.
From an educational perspective, a vector illustration comparing exothermic and endothermic reactions typically depicts two energy pathways. In exothermic diagrams, the pathway slopes downward, symbolizing energy release, while endothermic diagrams slope upward, representing energy absorption. These illustrations often include arrows showing heat flow out of or into the system. While simple, such diagrams effectively convey the essence of energy change: exothermic reactions lead to more stable, lower-energy states, while endothermic reactions move toward less stable, higher-energy states that require external input. These diagrams help students visualize how energy levels shift during chemical processes without mathematical descriptions.
The safety implications of understanding exothermic and endothermic behavior are significant. Exothermic reactions can escalate rapidly if not controlled, leading to overheating, pressure buildup, or even explosions in extreme cases. Endothermic reactions, while generally less hazardous in terms of heat release, can produce risks associated with sudden cooling, freezing of solutions, or destabilization of materials when temperatures drop below safe thresholds. Laboratories, industries, and educational settings rely on a clear understanding of these processes to ensure safe handling, controlled reaction rates, and appropriate responses to energy changes.
Even in everyday life, awareness of exothermic and endothermic processes enhances understanding of common experiences. Hand warmers rely on exothermic oxidation reactions to provide warmth, while cold packs used in first aid operate through endothermic dissolution reactions. Cooking, baking, freezing, refrigerating, and heating all depend on transformations involving these principles. Recognizing these processes helps explain why bread rises and browns in the oven, why ice cubes absorb heat as they melt in a drink, why certain chemical mixtures heat spontaneously, and why others require external heating to react.
Ultimately, the comparison between exothermic and endothermic processes reveals a broader truth about chemical and physical change: energy must always be accounted for. Whether a reaction releases energy or requires it, the total energy of the universe remains conserved, shifting forms and redistributing as matter transforms. Exothermic and endothermic reactions are not opposites but complementary aspects of a larger, interconnected system of energy flow. They embody the principles that govern the transformation of matter, the movement of heat, and the structure of natural and engineered systems.
By understanding these processes deeply, beyond diagrams and definitions, the flow of energy becomes a guiding thread that connects molecular interactions with large-scale phenomena. The warmth of a flame, the cooling of a melting ice cube, the power of industrial reactions, and the cycles of life itself all reflect the continuous interplay between exothermic and endothermic transformations. Through this rich conceptual lens, the world becomes a dynamic landscape of energy exchange, shaped by the structural and energetic realities of the reactions occurring everywhere, all the time.