Sigma and Pi Bonds Illustration Showing Molecular Orbital Overlap in Chemical Bonding
Sigma and pi bonds form the fundamental language of covalent chemistry, revealing how atoms unite by sharing electrons through overlapping orbitals to produce stable molecular structures. Understanding these bond types requires looking beyond simple line drawings on paper and imagining the three-dimensional way atomic orbitals interact in space. When two atoms approach each other closely, their electron clouds begin to overlap in ways that reduce the total energy of the system and allow electrons to exist in a shared region between nuclei. This shared density of electrons creates the attraction known as a covalent bond, and the shape and orientation of that overlap determine whether the bond is a sigma bond or a pi bond. Sigma bonds represent the strongest and most direct type of orbital overlap, forming along the axis that connects the two nuclei like a cylinder of electron density. Pi bonds, by contrast, arise from side-by-side overlap of orbitals positioned above and below this axis, forming a distributed electron cloud that complements the sigma bond and strengthens multiple bonding arrangements. A complete illustration of sigma and pi bonding highlights not only the geometry of the overlap but also the way these interactions create single, double, and triple bonds that determine molecular shape, reactivity, and physical properties.
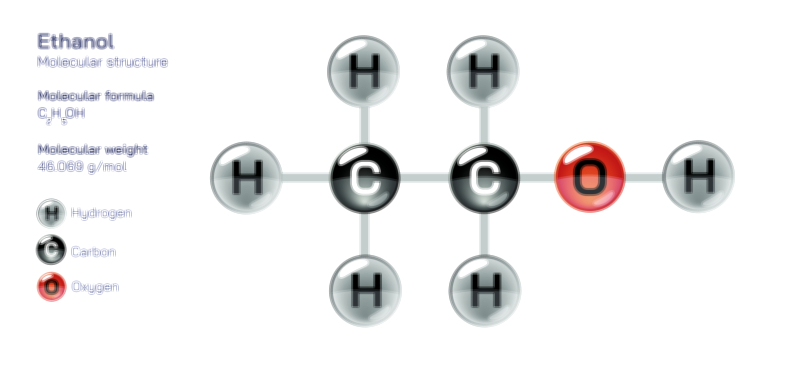
At the simplest molecular level, a sigma bond is the first and primary bond formed between two atoms because it involves head-on overlap, which brings electron clouds together in the most energetically favorable way. This can occur between two s orbitals, between an s and a p orbital, or between two p orbitals oriented along the internuclear axis. In a visual representation, the sigma bond appears as a region of electron density centered directly between the nuclei, resembling a smooth elongated cloud linking the two atoms. This arrangement allows electrons to exist symmetrically around the axis, meaning the nuclei can rotate relative to one another without breaking the bond. The strength, symmetry, and versatility of sigma bonds explain why every single covalent bond is a sigma bond. Even in double or triple bonds—where additional bonding occurs—the first bond formed is always a sigma bond because it provides the most stable foundation for atomic connection. The formation of this bond reduces the potential energy of the system significantly, pulling the nuclei toward each other and establishing the core of molecular stability before pi bonding can take place.
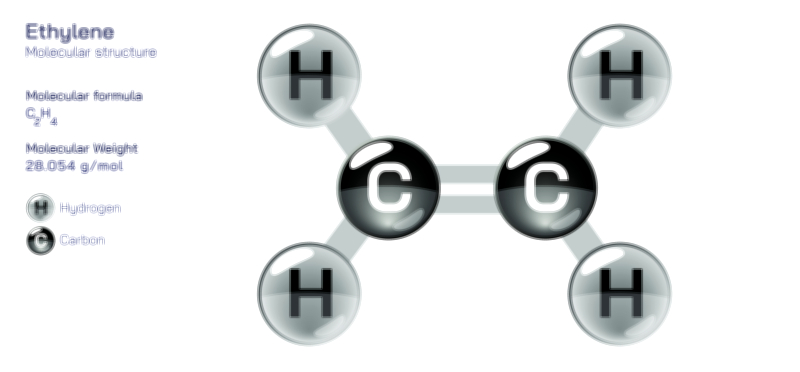
Pi bonds enter the picture when atoms form multiple bonds, such as in double or triple carbon–carbon bonds, and these cannot be explained through axial overlap alone. After the formation of a sigma bond, additional bonding capacity must come from orbitals arranged in a different direction. When p orbitals exist parallel to each other on adjacent atoms, their sideways alignment allows electron density to be shared in two lobes—one above the internuclear axis and one below it. This distribution creates a pi bond, which does not lie directly between the nuclei but wraps around the sigma bond like a belt holding the atoms more tightly together. In a diagram, this is commonly shown as two elongated electron clouds positioned over and under the bonding axis, making it visually clear that a pi bond depends on the presence of unhybridized p orbitals and that it cannot exist in isolation without a sigma bond anchoring the atomic framework. Because the overlap occurs sideways rather than head-on, the pi bond is generally weaker than the sigma bond and provides an additional but less flexible connection between the atoms.
A molecular illustration showing the difference between sigma and pi bonds becomes especially compelling when depicting a double bond. In such a structure, the sigma bond holds the atoms together with axial overlap, while the pi bond adds lateral reinforcement through parallel orbital overlap. This arrangement creates a planar molecular geometry because the pi bond restricts rotation. If one atom attempted to rotate relative to the other, the parallel p orbitals would lose alignment and the pi bond would instantly collapse. Thus, the restricted rotational freedom associated with double bonds is not a result of molecular crowding alone but of orbital geometry. This distinction explains why molecules with double bonds often exhibit rigid structural features such as cis–trans (E/Z) isomerism. A triple bond includes one sigma bond and two mutually perpendicular pi bonds, creating a linear configuration that locks the atoms tightly in place and shortens the bond length compared to both double and single bonds. Visualizing the sigma bond at the center with two pi bonds forming perpendicular overlapping regions around it helps demonstrate how multiple bonding dramatically enhances bond strength while simultaneously reducing rotational freedom.
The spatial orientation of sigma and pi bonds also connects directly to molecular hybridization, which describes how atomic orbitals reorganize themselves during bond formation. In a carbon atom forming four single bonds, hybridization results in four equivalent sp³ orbitals that create four sigma bonds in a tetrahedral arrangement. In a double bond, the carbon atoms adopt an sp² configuration in which three hybrid orbitals form sigma bonds while the remaining unhybridized p orbitals create the pi bond. In a triple bond, sp hybridization produces two linear sigma bonds, leaving two unhybridized p orbitals available for the two pi bonds that complete the triple-bond structure. When illustrated clearly, hybridization and orbital overlap become complementary ideas—the hybrid orbitals explain the directional sigma bonding, while the unhybridized orbitals explain where and how pi bonds form. Looking at a single diagram that compares sp³, sp², and sp hybridization alongside the resulting sigma and pi bonds reveals how chemical bonding shapes molecular architecture at the level of electron arrangement rather than atomic labels alone.
Beyond geometry and structural identity, sigma and pi bonds influence chemical reactivity in profound ways. The electron density concentrated directly along the internuclear axis in sigma bonds makes them deep and tightly bound, requiring high energy to break; this is why sigma bonds tend to remain intact unless severe chemical or thermal stress is applied. Pi bonds, however, with their electron density farther from the nuclei, are more exposed to external reagents and therefore far more reactive. Many characteristic reactions of organic chemistry, such as electrophilic addition to alkenes or nucleophilic attack on carbonyl groups, involve the selective breaking of pi bonds while leaving the sigma bond untouched. The pi bond becomes the entry point for reaction because its distributed electron cloud is vulnerable to attraction by electron-seeking species. Understanding this dynamic explains why compounds with double or triple bonds show higher levels of reactivity and participate in specialized reaction pathways compared to fully saturated compounds containing only sigma bonds. An illustration capturing both types of bonding not only clarifies structural differences but also foreshadows how particular molecules behave in chemical environments.
When sigma and pi bonds are represented together in an educational or professional illustration, the image becomes more than a static diagram—it becomes a visualization of the physical logic shaping chemical structure. Sigma bonds, drawn as direct overlaps along the bonding axis, convey stability, strength, and rotational freedom. Pi bonds, drawn as cloud-like regions above and below or around the axis, communicate added reinforcement, rigidity, and reactivity. Together, they demonstrate that covalent bonding is not a one-dimensional concept but a three-dimensional interaction governed by orbital orientation, electron distribution, and energy minimization. Chemists rely on this understanding to design molecules with desired shapes and functions, while biochemists use it to explain the structure of proteins, enzymes, and nucleic acids. Even fields like materials science and nanotechnology depend on sigma and pi bonding to create conductive surfaces, aromatic rings, polymers, and carbon-based nanomaterials such as graphene. In every instance, visualizing orbital overlap deepens understanding of how electrons hold the molecular world together.
Seeing sigma and pi bonds side by side connects theoretical chemistry with the practical behavior of molecules, turning symbolic chemical formulas into meaningful spatial structures. The overlapping orbitals in a diagram are not merely academic—they are graphic representations of the forces that organize matter and dictate how atoms interact, combine, and transform. By appreciating how these two types of covalent interactions complement and reinforce each other, one gains a clearer view of why molecules have the shapes they do, why they respond to different chemicals in predictable ways, and why the complexity of life and materials emerges from a handful of consistent, elegant quantum interactions. Through such illustrations, chemistry becomes less a collection of rules and more a visual architecture of interaction, where the invisible becomes thinkable and the structure of nature reveals itself through the geometry of electrons in motion.