Ethanol Chemical Structure — Alcohol Compound Diagram Explained
Ethanol, one of the most widely recognized and extensively studied organic compounds, occupies a central position in chemistry, biology, industry, and everyday human experience. Its structure, although simple, reveals essential principles of organic chemistry, including carbon bonding behavior, functional group characteristics, molecular geometry, and intermolecular interactions. Understanding the chemical structure of ethanol not only clarifies why it behaves the way it does but also provides a foundational framework for understanding the entire family of alcohols. When examined in detail, ethanol becomes more than just a common molecule found in beverages or fuels—it becomes an elegant example of how atomic arrangement dictates physical properties, reactivity, biological effects, and technological usefulness. Describing its structure in rich conceptual detail allows the molecule to be understood far beyond the lines and symbols of a typical diagram, transforming it into a fully meaningful entity within the chemical world.
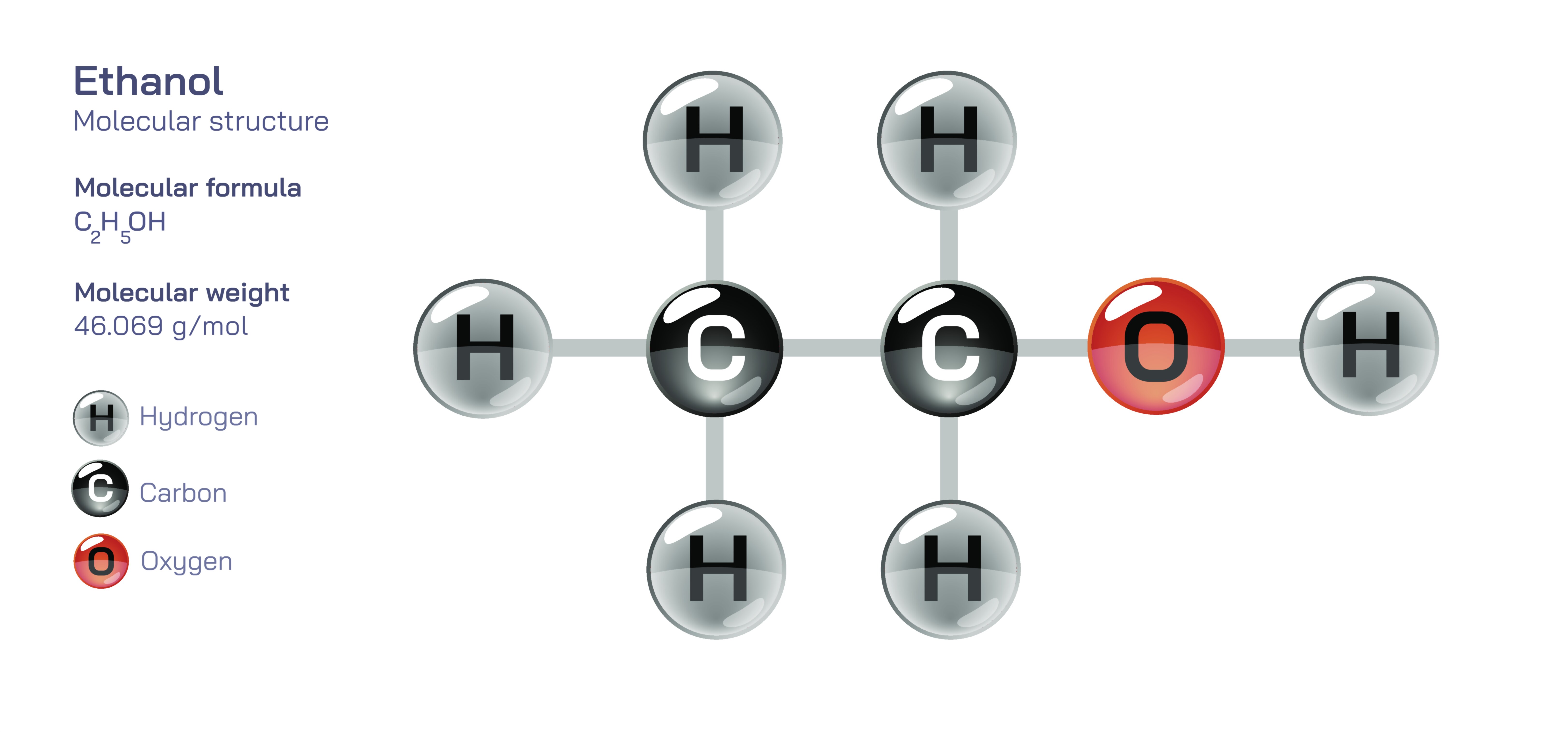
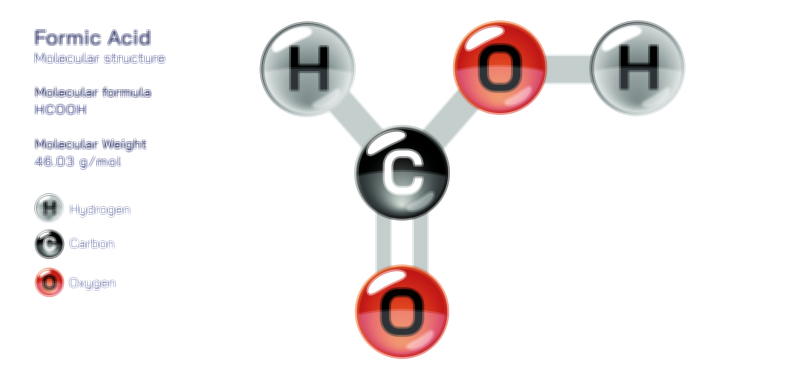
At its core, the ethanol molecule consists of two carbon atoms, six hydrogen atoms, and one oxygen atom, forming the molecular formula C₂H₅OH or, more structurally descriptive, CH₃–CH₂–OH. The molecule belongs to the alcohol family, defined by the presence of a hydroxyl functional group—a combination of an oxygen atom bonded directly to a hydrogen atom. This hydroxyl group is the feature that transforms ethanol from a simple hydrocarbon into a compound with distinctive chemical and physical behavior. Despite the small size of the molecule, this functional group introduces polarity, hydrogen bonding capability, reactivity with acids and bases, and biological activity. In diagrams, the hydroxyl group is typically drawn clearly and prominently to represent the functional aspect of the molecule, distinguishing ethanol from related hydrocarbons like ethane.
The ethanol molecule begins with a carbon–carbon single bond connecting a methyl group (CH₃) to a methylene group (CH₂). The methyl group, located at one end of the molecule, represents a carbon atom fully saturated with three hydrogen atoms. The adjacent methylene carbon connects not only to the methyl carbon but also to two hydrogen atoms and the oxygen of the hydroxyl group. As in all saturated organic molecules, each carbon atom in ethanol adopts a tetrahedral geometry, meaning its four substituents are oriented in three-dimensional space approximately evenly apart. This tetrahedral structure minimizes electron repulsion around each carbon atom’s valence shell, creating the most stable arrangement possible under typical conditions. Diagrams often flatten this three-dimensional configuration into a two-dimensional representation, but a deeper understanding emphasizes that the molecule is spatially dynamic rather than planar.
The hydroxyl group attached to the second carbon plays the most defining role in ethanol’s structure and function. Oxygen, with its high electronegativity, attracts electron density strongly toward itself, giving the O–H bond a pronounced polarity. Within this structure, the oxygen atom holds a partial negative charge, while the hydrogen carries a partial positive charge. This polarization enables ethanol molecules to engage in hydrogen bonding, both with one another and with water. Hydrogen bonding is a powerful intermolecular force, far stronger than typical van der Waals interactions found in hydrocarbons. It explains many of ethanol’s most recognizable macroscopic properties, such as its miscibility with water, relatively high boiling point for its molecular size, and characteristic solvent behavior. The presence of this polar functional group differentiates ethanol from purely nonpolar hydrocarbons that lack such strong intermolecular attractions.
The ethanol structure also reveals how functional groups govern chemical reactivity. The hydroxyl group can donate hydrogen in certain reactions, accept hydrogen in others, form esters with acids, react with oxidizing agents to form aldehydes or carboxylic acids, and participate in substitution or elimination processes under controlled conditions. These transformations are all rooted in the electronic structure of the oxygen atom and the bond polarity associated with it. The carbon chain in ethanol serves as a relatively nonreactive framework, while the hydroxyl group acts as the reactive center. In conceptual illustrations or vector representations, the hydroxyl group is often highlighted or drawn distinctly to emphasize its function as the molecule’s reactive heart.
Ethanol’s structure also introduces the concept of hybridization. Each carbon atom in ethanol adopts an sp³ hybridization state, meaning that the atomic orbitals combine to form four equivalent orbitals arranged tetrahedrally. This hybridization supports the formation of strong sigma bonds with hydrogen atoms, the adjacent carbon, and the oxygen atom. The oxygen atom also uses hybridized orbitals, retaining two lone electron pairs that influence molecular geometry. These lone pairs slightly distort the geometry around the oxygen, making the O–H bond angle narrower than the tetrahedral ideal. These small structural features have profound consequences for the molecule’s overall polarity, solubility, and interaction with biological systems.
The spatial arrangement of ethanol and the polarity of its bonds create a molecule with one polar end and one nonpolar end. The hydroxyl end interacts strongly with water and other polar molecules, while the hydrocarbon portion interacts more readily with nonpolar substances. This dual character gives ethanol the ability to serve as a bridge between polar and nonpolar environments—a reason it is such a versatile solvent. It can dissolve substances that are otherwise incompatible, making it ideal for pharmaceuticals, disinfectants, laboratory solutions, and industrial formulations. The diagrammatic representation of ethanol often captures this duality implicitly, with the hydroxyl group placed distinctly to highlight its polarity.
Ethanol also demonstrates the concept of molecular flexibility. The carbon–carbon bond in ethanol is a single sigma bond, which allows rotation around its axis. This rotational freedom means that the spatial orientation of the hydroxyl group relative to the carbon backbone can vary constantly. Although subtle, these conformational changes influence how ethanol interacts with neighboring molecules, both in structure and in biological contexts. The ability of ethanol to adopt multiple conformations allows it to fit into various molecular environments, contributing to its biological and physiological effects.
The structure of ethanol reveals its phase behavior and thermodynamic properties. Because of hydrogen bonding, ethanol boils at a much higher temperature than similarly sized hydrocarbons such as ethane or propane. The presence of the hydroxyl group dramatically increases intermolecular attractions, requiring more energy for vaporization. Ethanol is also fully miscible with water because the two substances can form extensive networks of hydrogen bonds. This complete miscibility contrasts sharply with hydrocarbons, which do not dissolve in water because they lack polar functional groups capable of bonding with water molecules. Even ethanol’s flammability is linked to its structure. The hydrocarbon portion provides combustible energy, while the oxygen-containing functional group readily participates in oxidation reactions.
Ethanol’s chemical structure also influences its biological effects. In living organisms, ethanol interacts with cell membranes, proteins, and neural receptors. Its polarity allows it to dissolve into the water-filled cytoplasm, while its nonpolar end allows it to embed within lipid membranes. This dual solubility allows ethanol to travel easily through tissues, cross cellular barriers, and reach the brain rapidly. Its small size enhances this mobility. The interactions between ethanol molecules and nerve cell receptors produce the well-known psychoactive and physiological effects. While biological activity is complex and multifactorial, these behaviors begin with the molecule’s simple structural features.
In environmental and atmospheric contexts, the structure of ethanol affects how it breaks down and participates in natural cycles. Ethanol, unlike many complex organic solvents, degrades relatively quickly and does not accumulate persistently. Its hydroxyl group facilitates biodegradation, making it more environmentally friendly in certain applications compared to longer-chain alcohols or synthetic solvents. The structure’s inherent reactivity ensures that ethanol does not remain chemically inert, participating instead in numerous natural processes.
As a foundational compound in organic chemistry education, ethanol’s structure helps students and researchers understand broader principles. It acts as a gateway to learning about alcohol classification, oxidation states of carbon, functional group reactivity, hydrogen bonding, polarity, isomerism, and intermolecular forces. Its simplicity makes it accessible, while its functional significance makes it essential. In vector illustration diagrams or structural models, ethanol is often presented in a way that emphasizes clarity: a two-carbon chain with a distinct –OH group. Yet behind this straightforward diagram lies a wealth of structural detail that shapes the molecule’s properties and uses.
In industrial processes, ethanol’s structure underlies its transformation into numerous other compounds. Through dehydration, ethanol forms ethylene, a fundamental building block in polymer production. Through oxidation, it can produce acetaldehyde or acetic acid. Each of these transformations relies on the bond arrangement within the ethanol molecule and the ability of the hydroxyl group to participate in chemical changes. Ethanol is also widely used as a renewable fuel, and its structural characteristics influence its combustion behavior and energy output. The presence of oxygen within the molecule contributes to cleaner burning compared to purely hydrocarbon fuels.
Ultimately, the chemical structure of ethanol illustrates how a small, simple molecule can carry immense scientific, practical, and societal significance. Each bond, each atom, and each geometric arrangement contributes not only to the molecule’s physical form but to its chemistry, its interactions, its usefulness, and its effects. By understanding ethanol’s structure deeply and descriptively, it becomes clear that this common alcohol is a finely tuned arrangement of atoms whose properties flow naturally from its design. Whether depicted as a vector illustration, a skeletal formula, or a three-dimensional model, ethanol’s structure stands as a powerful example of how molecular architecture determines behavior, shaping everything from chemical reactions to biological responses and technological applications across the world.