Boron Element Illustration: Atomic Structure, Properties, and Uses Explained
Boron stands out in the periodic table not simply as a lightweight element but as a remarkable building block of chemistry, materials science, industrial processes, and even biological function. When represented in an illustration, boron is usually shown as a small atom with a compact nucleus and a modest cloud of electrons, yet this simplicity hides a wealth of chemical behavior. Boron occupies a unique position between metals and nonmetals, giving it properties that sometimes resemble carbon, sometimes silicon, and sometimes neither. In its atomic form, boron is represented by atomic number five, meaning its nucleus contains five protons supported by a matching constellation of five electrons orbiting in arranged energy levels. The diagram of the atom emphasizes two electrons in the inner stable shell and three in the outer shell—also known as the valence shell—which determines bonding nature and reactivity. This trio of valence electrons shapes boron’s ability to participate in covalent bonding and cluster formation, its unusual affinity for forming electron-deficient structures, and its keen tendency to form rigid, heat-resistant networks. A simple atomic illustration might give the impression of modest complexity, yet anyone familiar with the substance soon discovers that boron’s power is far greater than its size.
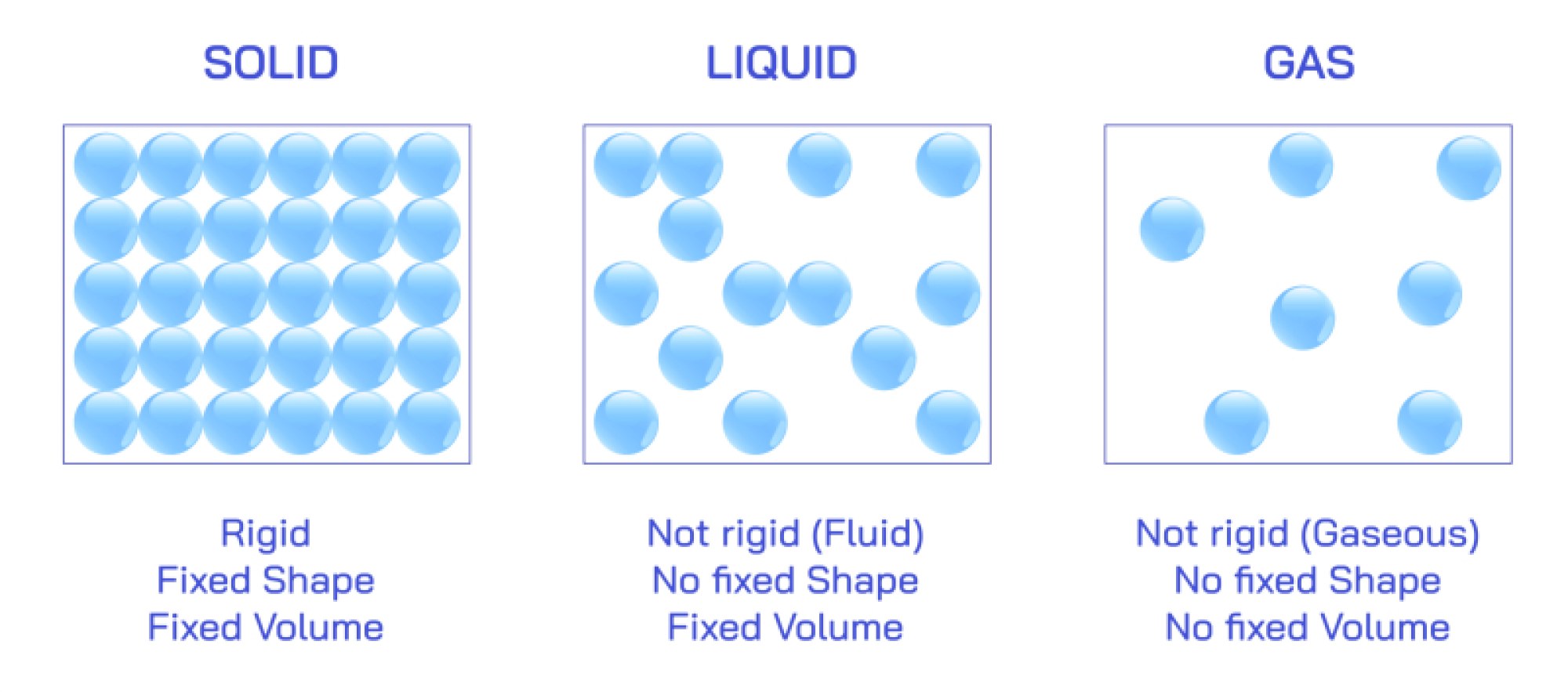
In structural illustrations showing solid boron, the element is often depicted forming stiff, triangular arrangements and interconnected networks. Unlike metallic structures, which pack atoms closely in predictable repeating patterns, and unlike classical nonmetals such as oxygen or carbon, boron creates structures that optimize bonding despite electron deficiency. Its atoms cluster into icosahedral units—geometric objects composed of twenty triangular faces that pack together in rigid formations. These geometric features explain why crystalline boron is extraordinarily hard, resisting changes in shape and earning a place among some of the most difficult natural and synthetic materials to break. Boron’s electron sharing is not straightforward; it forms bonds that distribute electrons through three centers at once, creating stable networks even when the number of electrons is lower than what classical bonding theory would require. When this microscopic architecture is stretched across millions of atoms, the result is a substance that endures extreme conditions. Illustrations of boron’s metallic-like dark crystals frequently emphasize the close packing of these icosahedral shapes, helping explain why the element appears brittle yet tough, lightweight yet nearly indestructible.
On the chemical level, an illustration of boron often accompanies depictions of two of its best-known compounds: boric acid and borax. Boric acid may be shown as a network of boron bonded with oxygen and hydrogen, forming flat geometric units that dissolve into gentle antiseptic solutions often used in medicine, household cleaning, and pest control. Borax, a compound of boron, sodium, oxygen, and hydrogen, is typically illustrated as a white powder that crystallizes in translucent cubic form. It appears in detergents, flame retardants, water softeners, and chemical laboratories, and it plays a stabilizing role in many everyday materials. These compounds show a different side of boron from its hard crystalline form—this element adapts its bonding behavior depending on its environment, dissolving into cleaner, more reactive structures that participate in chemistry rather than resisting it. Through these contrasting depictions, boron appears not as a rigid one-purpose element, but as a versatile participant whose function is shaped by context.
A chart of boron’s properties highlights lightness, high melting temperature, thermal stability, and resistance to corrosion. In thermodynamic illustrations, boron is frequently shown remaining structurally unchanged even under temperatures that deform many metals. Its high melting point helps explain why boron-containing materials serve as components in heat-resistant glass, ceramic shields, and metal alloys used in spacecraft, jet engines, and laboratory equipment. One of the most famous boron-based materials is borosilicate glass, a heat-resistant glass known for its ability to withstand sudden temperature shifts. Diagrams showing ordinary glass cracking beside borosilicate glass remaining stable illustrate how boron atoms strengthen the internal structure, preventing expansion and contraction from destroying the material. Even in fiber form, such as boron fibers used in aerospace components, illustrations emphasize their exceptional stiffness despite minimal weight. This dual property—light yet strong—makes boron indispensable in fields where performance and weight must be carefully balanced.
Biological and environmental illustrations of boron reveal yet another dimension of the element. In plant physiology, boron is shown playing a vital role in strengthening cell walls, enabling proper nutrient transport, and assisting in the reproduction of root and shoot systems. Farmers and soil scientists rely on boron analysis because the element is crucial at the right concentration and toxic when applied excessively. Visual representations of leaves suffering from deficiency—curling edges, cracked stems, or stunted growth—contrast sharply with healthy, boron-balanced plants. The dual nature of boron in biology supports the broader theme that the element must exist in controlled levels rather than extremes. Humans do not require large quantities of boron, yet small amounts appear to support bone strength, metabolism, and cognitive function. Nutritional illustrations showing vegetables, nuts, and fruits as dietary sources reinforce boron’s presence as part of daily life, even if it remains invisible in the systems it supports.
The industrial usefulness of boron becomes even broader when diagrams display its expanded role in energy storage, electronics, and nuclear science. Boron carbide, one of the hardest synthetic substances known, is represented as a protective armor material used in body armor plates, shields, and structural reinforcements. Boron nitride appears in illustrations as a smooth lubricating material that withstands extreme heat, used in mechanical bearings and high-performance electronics. Boron’s neutron-absorbing ability makes it valuable in nuclear reactors where control rods lined with boron compounds help regulate atomic reactions and maintain safe operational stability. Illustrations showing neutrons being captured by boron atoms make this safety mechanism visible and emphasize its contribution to sustainable nuclear power systems. Even future energy designs involve boron, as research explores boron hydrides and boron-based nano-materials for hydrogen storage and energy-dense fuels. In diagrams of batteries and fuel systems, boron appears not as decorative detail but as a key to improving efficiency and reducing losses.
In context of everyday products, another type of illustration depicts boron embedded in the modern household. Boron compounds appear in laundry detergents to soften water by binding calcium and magnesium ions. They serve as fire retardants in insulation and textiles, stabilize plastics to prevent deterioration under UV radiation, and contribute to fiberglass formation used in home construction. Even children’s science toys often rely on boron compounds—for example, borax plays a role in linking polymers to create slime. Illustrations of these uses reveal how widespread boron is in daily surroundings, even if the element remains invisible to most people outside chemistry classrooms or industrial settings.
Boron’s role in technological progress is equally compelling. In semiconductor illustrations, boron appears as a doping agent used in silicon-based electronics to modify electrical conductivity. These doped materials become the backbone of integrated circuits, solar panels, and countless digital devices. Whether performing in a microscopic transistor or in the ceramic tiles shielding spacecraft from atmospheric re-entry, boron consistently appears in diagrams as a silent helper—never the largest component, yet often the most decisive in determining the structural, electrical, or thermal performance of the final material. Such images make clear that boron’s value lies not in abundance but in efficiency and precision.
In many educational illustrations, boron is placed alongside neighboring elements—carbon, nitrogen, oxygen—to show similarities and distinctions. Like carbon, boron forms complex covalent networks, yet unlike carbon it is happiest in electron-deficient configurations. Like nitrogen, it participates in strong bonding, yet unlike nitrogen it forms rigid solids rather than atmospheric gases. Like silicon, boron reinforces glass, though silicon alone cannot provide the same heat-resistant properties. These comparison diagrams lead students to recognize a broader theme: boron behaves as a bridge element, combining traits normally divided between metals and nonmetals, hardness and reactivity, structural toughness and chemical refinement. Its small atomic diagram begins to represent not only structural information but the clue to understanding its behavior across industrial, biological, and materials-science landscapes.
Ultimately, a boron illustration—whether anatomical, chemical, industrial, or biological—reveals the story of an element that quietly reinforces the world’s structures and technologies without calling attention to itself. It serves as a connective thread between laboratory chemistry, human health, agriculture, renewable energy, electronics, and mechanical engineering. Although composed of just five protons and five electrons, boron has leveraged that modest atomic blueprint into a role far larger than its atomic mass might suggest. It protects spacecraft from heat, strengthens bones in living organisms, purifies laundry water, controls nuclear reactions, and supports the most delicate tissues of plants. By making the internal structure and external functions visible through illustration, boron becomes more than a symbol on the periodic table—it becomes a model of how small atomic arrangements can yield outsized influence in both the natural world and human innovation.