Iron (Fe) Element Illustration: Atomic Structure, Properties, and Uses Explained
Iron (symbol Fe) is one of Earth’s most abundant and economically important elements, and an illustration of its atomic structure, physical properties, and wide-ranging uses helps reveal why it has shaped human civilization as profoundly as it has shaped the planet itself. From the metallic core of Earth to the tools and infrastructure of modern life, iron represents a foundation of industry, technology, and biology. A well-designed diagram of iron brings together its atomic-scale identity, bulk characteristics, and real-world applications in a unified visual narrative.
At the atomic level, iron is shown with 26 protons in its nucleus, corresponding to its atomic number, along with 26 electrons arranged in orbitals around the nucleus. A typical electron-shell illustration assigns these electrons to four levels:
• K shell (1st): 2 electrons
• L shell (2nd): 8 electrons
• M shell (3rd): 14 electrons
• N shell (4th): 2 electrons
This distribution demonstrates the partially filled 3d subshell that gives iron its magnetic behavior, chemical reactivity, and ability to form multiple oxidation states, especially +2 (ferrous) and +3 (ferric). Diagrams of electron configuration often display notation such as 1s² 2s² 2p⁶ 3s² 3p⁶ 3d⁶ 4s², showing the 3d electrons responsible for iron’s characteristic bonding and metallic properties. These orbital illustrations highlight why iron is central to the formation of compounds, alloys, and catalytic reactions.
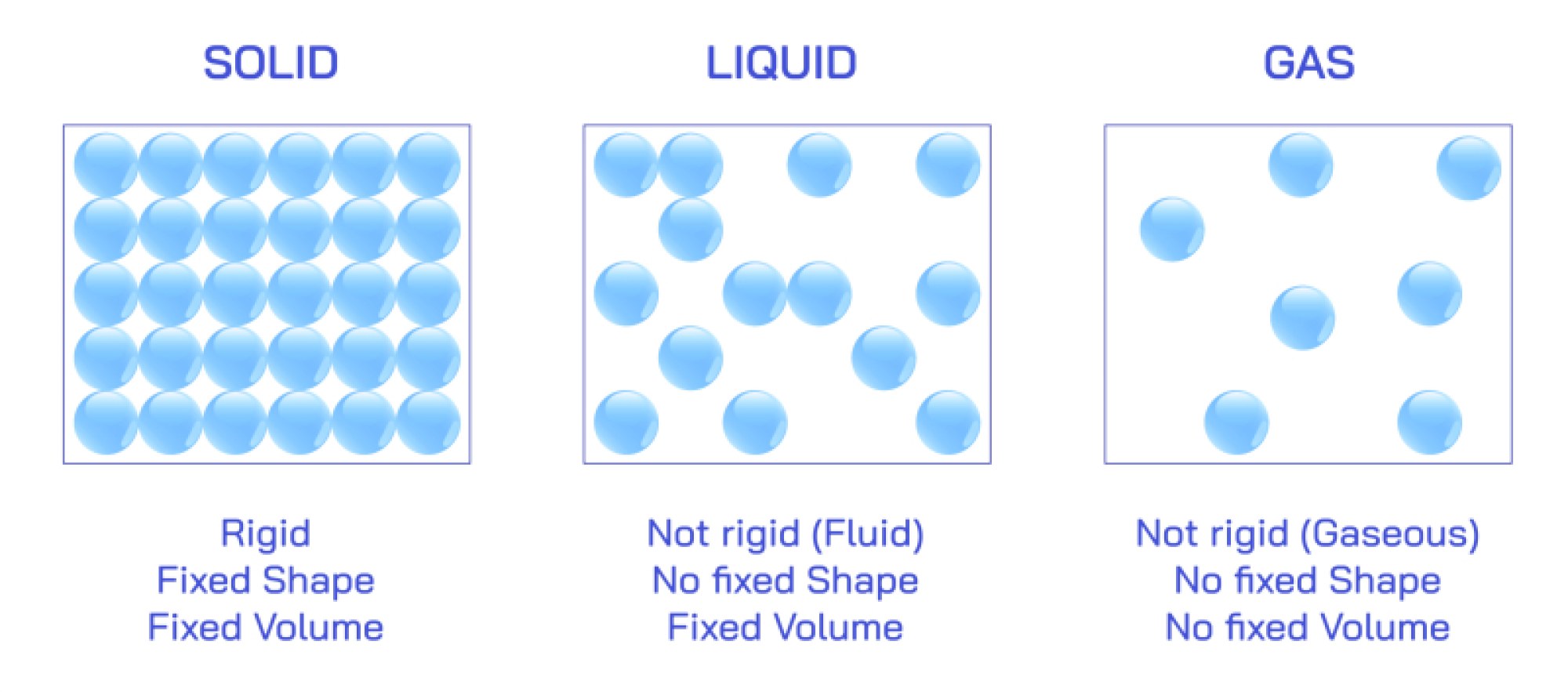
Illustrations of bulk iron properties typically include its metallic crystal structure. At room temperature, iron exists in a body-centered cubic (BCC) structure called α-iron or ferrite. When heated, it transforms to a face-centered cubic (FCC) structure known as γ-iron or austenite, before reverting to BCC at even higher temperatures (δ-iron). Industrial diagrams use these structural transitions to explain why heat treatment—such as tempering, quenching, and annealing—changes the hardness, ductility, and tensile strength of steel. Additionally, iron is magnetic in its ferrite form, and illustrations often depict magnetic domains within the metal aligning under an external magnetic field. Iron’s high tensile strength, workability, ability to form alloys, and comparatively low cost make it the dominant structural metal in engineering and architecture.
A geological illustration of iron emphasizes that it is the fourth most abundant element in Earth’s crust and the primary component of the planet’s inner and outer core. The magnetic field surrounding Earth arises largely from convection of liquid iron in the outer core—a fact often visualized by swirling arrows and magnetic field lines. Mineral diagrams commonly show iron present in ores such as hematite (Fe₂O₃), magnetite (Fe₃O₄), limonite, and siderite, which must undergo extraction and smelting in blast furnaces. Illustrations of the smelting process typically show iron oxide + carbon (coke) → molten iron, with slag separating from the metal. These industrial graphics help explain how raw ore becomes usable iron and subsequently steel.
The most iconic use of iron is in steel production, and illustrations often depict steel as an alloy of iron with carbon (up to ~2%) and small quantities of other elements such as chromium, nickel, manganese, vanadium, or molybdenum. Diagrams of microstructures such as pearlite, martensite, ferrite, and cementite help explain how steel’s performance characteristics—whether hard, flexible, tough, or corrosion-resistant—depend on composition and heat treatment. Stainless steel diagrams highlight chromium-oxide passivation layers that prevent rust. These visuals connect atomic-scale chemistry to massive architectural structures such as buildings, bridges, ships, pipelines, heavy machinery, and automobile frames.
Iron’s significance extends far beyond industry. In biology, iron is essential for life and is portrayed in diagrams as the functional metal at the center of hemoglobin, the protein responsible for transporting oxygen in the blood. Hemoglobin illustrations commonly display an iron atom nestled within the heme group, where it binds oxygen reversibly—picking it up in the lungs and releasing it into body tissues. Iron is also vital in myoglobin, cytochromes, and metabolic enzymes, and anatomical illustrations often show how iron deficiency can lead to anemia, fatigue, and impaired immunity. Nutrition charts note rich dietary sources such as red meat, leafy greens, beans, and fortified grains, contrasting heme iron (more bioavailable) with non-heme iron.
Illustrations of iron’s chemical reactions reinforce its reactive nature. When exposed to oxygen and moisture, iron undergoes oxidation to form iron oxides, commonly known as rust. Corrosion diagrams show the stepwise breakdown of the metal surface, often accompanied by electron-transfer visuals that link oxidation to reduction reactions elsewhere. Protective coatings, galvanization using zinc, and alloying strategies are drawn to compare rust-prone material with corrosion-resistant versions. These comparisons help clarify why pure iron rusts easily while steel formulations and protective methods mitigate deterioration.
In the context of technology and electromagnetism, diagrams of iron in electric motors, transformers, generators, speakers, and magnetic storage devices demonstrate the element’s high magnetic permeability. Iron cores concentrate magnetic fields, enabling efficient conversion between electrical and mechanical energy. Magnetic hysteresis loops, sometimes included in engineering illustrations, explain how soft iron responds to alternating magnetic fields with minimal residual magnetism—making it ideal for energy transfer systems.
From a cultural and historical perspective, some educational illustrations trace human progress through the Iron Age, when the ability to smelt and forge iron revolutionized agriculture, warfare, and construction. These timelines visually connect iron to human development, showing tools and weapons that replaced earlier bronze technology and enabled widespread social, economic, and engineering change.
Ultimately, an illustration of the element iron integrates atomic identity, material properties, geological availability, biological function, and technological importance into a single visual explanation. It depicts iron not only as a metal but as a chemical and cultural cornerstone—from the atoms that transport oxygen in blood to the alloys that support bridges and skyscrapers to the magnetic forces driving modern machinery. By visualizing iron across these scales, the illustration demonstrates how one element can shape both the inner workings of the human body and the infrastructure of the entire world.