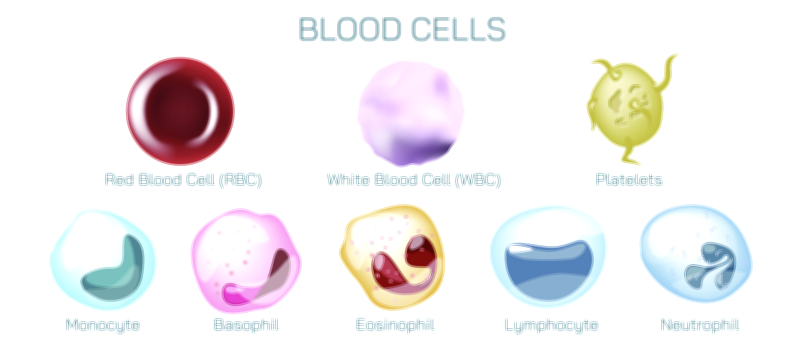
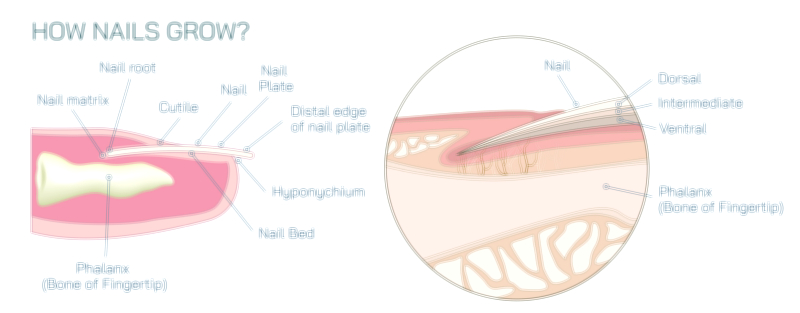
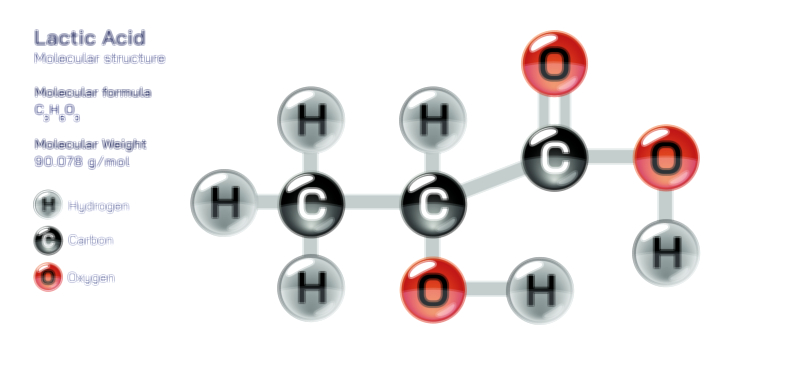
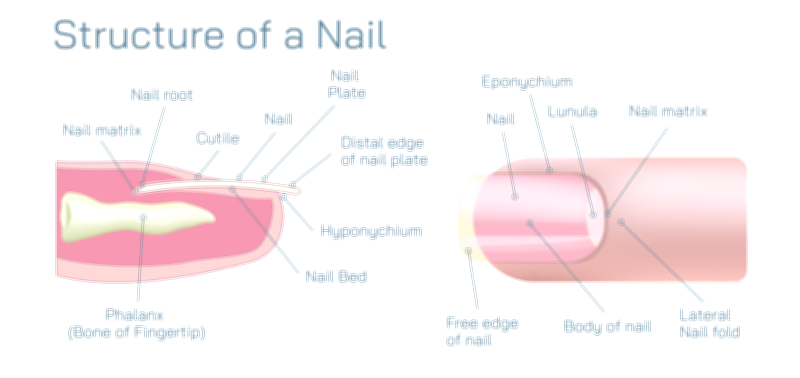
Antibody and Antigens Showing Immune Response, Antigen Binding, and Defense Mechanism in the Human Body
The interaction between antibodies and antigens is the cornerstone of the human immune defense, forming a microscopic battle that protects the body from infections, toxins, and foreign invaders. A vector illustration depicting antibodies and antigens usually places these elements in a dynamic scene where uniquely shaped antibody molecules bind to matching antigens on the surfaces of pathogens such as viruses, bacteria, or other harmful particles. Although the image may appear simple — Y-shaped antibodies attaching to differently shaped antigen markers — it represents a highly specialized biological system built upon molecular recognition and immune memory. This structural complementarity is what enables the immune system to identify a nearly limitless variety of threats with precision and speed.
Antigens are molecules, typically proteins or polysaccharides, found on the surface of invaders that the immune system identifies as foreign. Every pathogen carries antigens that serve as biological identity markers. In illustrations, antigens are often shown as protruding shapes on the outer membrane of microbes. The human body is constantly exposed to these foreign structures through inhalation, ingestion, injury, and environmental contact. When antigens enter the body, immune surveillance cells recognize them as non-self, setting off a chain of responses. Importantly, antigens do not attack; instead, their presence triggers the defensive action, similar to how a warning flag alerts a security system. The specificity of each antigen determines which antibodies can bind to it because only a matching antibody configuration can attach to an antigen’s surface features — a biological equivalent of a lock-and-key system.
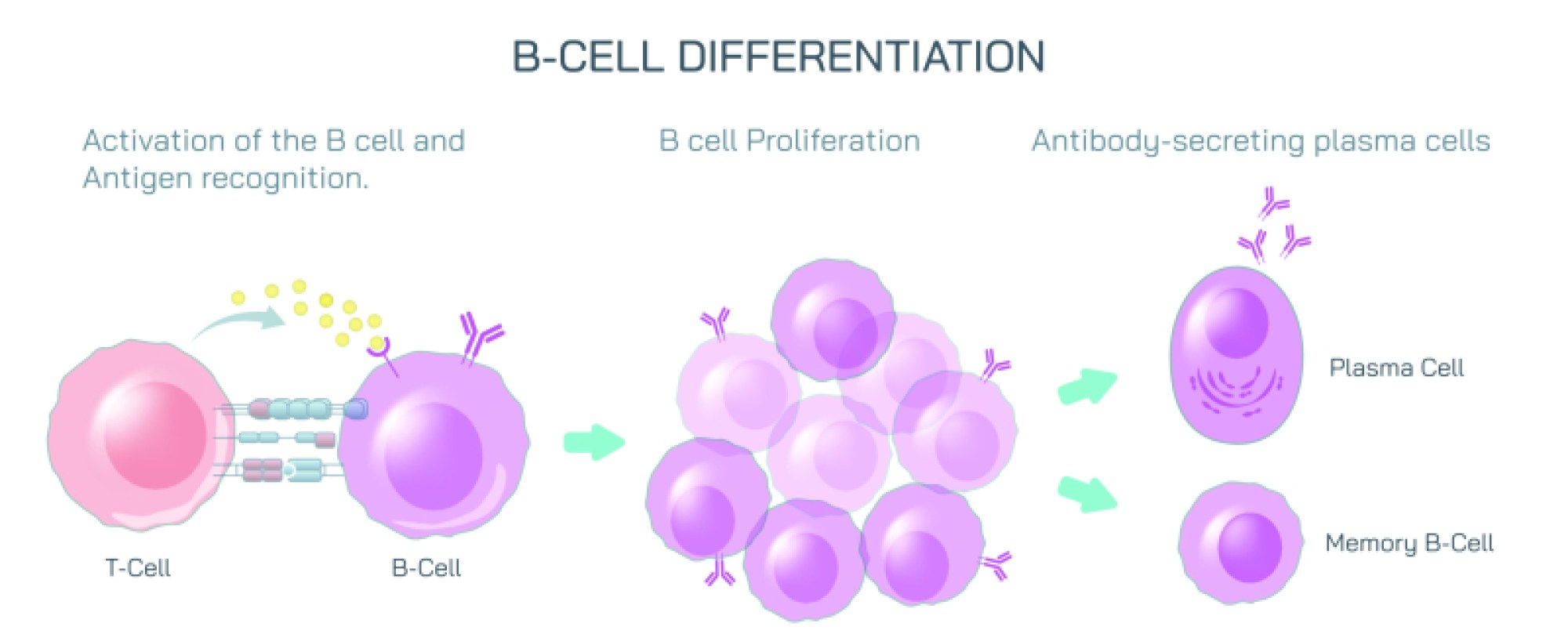
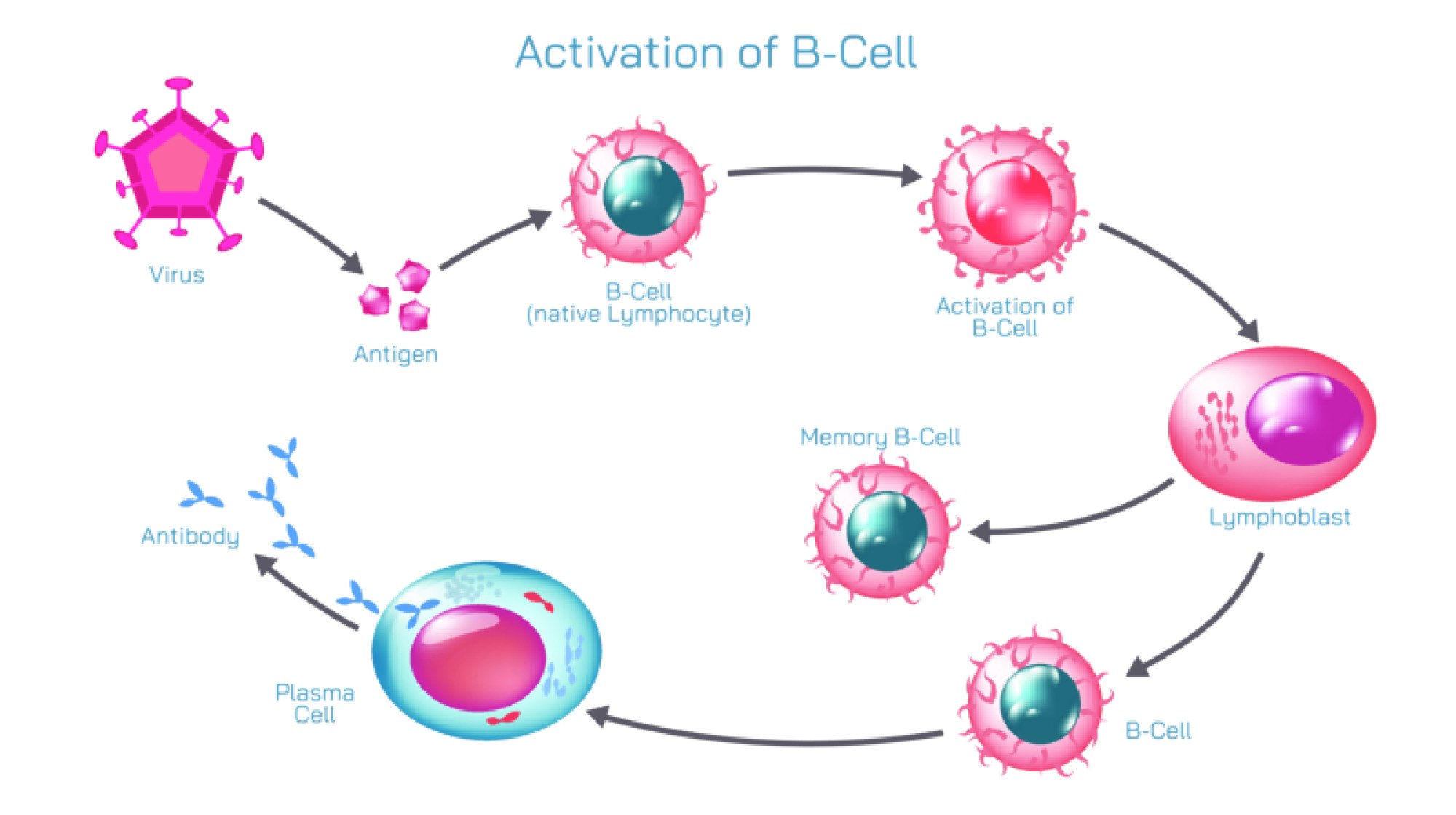
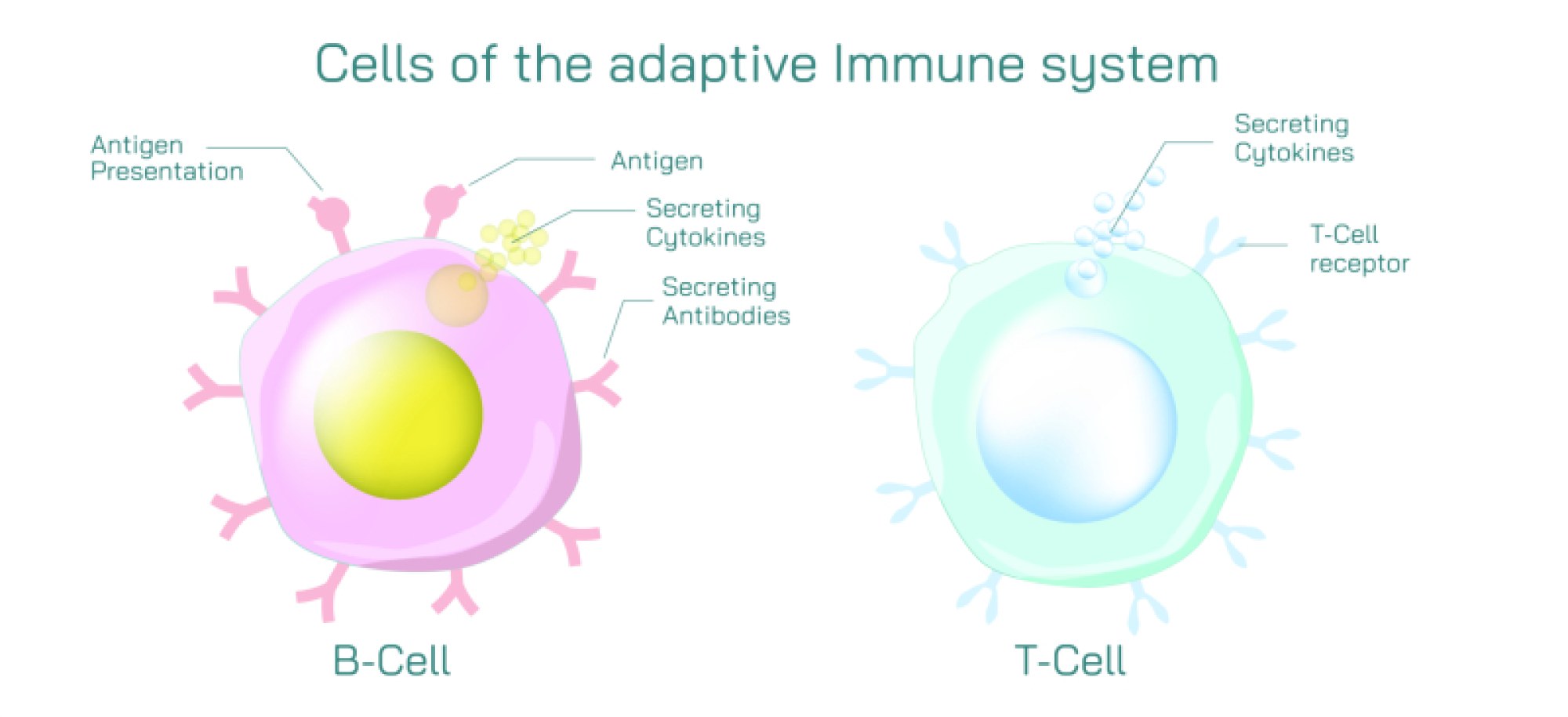
Antibodies, also known as immunoglobulins, are Y-shaped proteins produced by B cells in response to antigen exposure. In diagrams, antibodies extend their two binding arms toward the antigen, illustrating how their structure is built to latch onto invaders. The tips of each antibody contain variable regions — areas that can undergo enormous molecular diversity so that the immune system can produce antibodies for potentially millions of different antigens. When an antibody binds to an antigen, it does not simply attach; it marks the target for destruction and initiates multiple immune strategies. Binding may neutralize toxins directly by blocking their active sites, prevent viruses from entering cells, clump pathogens together to make them easier to engulf, or signal phagocytes and complement proteins to attack. Thus, antibody-antigen binding is both recognition and activation, identifying harmful particles and igniting the broader immune network.
The immune response shown in vector illustrations usually highlights this chain of events: antigens first appear on the pathogen surface, antibodies circulate through the bloodstream or tissue, and binding occurs when shapes match. This molecular recognition triggers downstream defense mechanisms. Some antibodies remain attached to B cells and act as detectors, while others circulate freely in blood and lymph, responding to threats across the body. Once binding occurs, immune signals intensify rapidly. Phagocytic white blood cells such as macrophages or neutrophils are attracted to the antibody-tagged invader and engulf it. The complement system — a cascade of plasma proteins — can be activated to punch holes in bacterial membranes or amplify inflammation at the infection site. These processes ensure that once an antigen is identified, the entire immune system joins the effort to eliminate the threat.
A vital feature of immunity illustrated in antibody-antigen diagrams is immune memory. After an initial exposure, the immune system retains memory B cells that remember the antigen’s structure for years, sometimes for life. If the body encounters the same antigen again, it launches a much faster and stronger antibody response, often eliminating the pathogen before symptoms even develop. This is why antibody imagery is commonly associated with vaccines: vaccination introduces harmless or weakened antigens so that memory cells form before actual infection occurs, ensuring rapid protection if the real pathogen appears.
The unique binding between antibodies and antigens highlights how the immune system distinguishes between self and non-self. Cells of the human body have antigens too, but these “self antigens” normally do not provoke antibody production because of immune tolerance — the training process that teaches immune cells not to attack the body’s own tissues. When tolerance fails, the immune system may mistake self antigens as foreign, leading to autoimmune diseases. This demonstrates how the accuracy of antibody-antigen binding is essential not only for defending the body but also for preventing self-damage.
In medical science, antibody-antigen interactions have become a foundational diagnostic and therapeutic tool. Laboratory tests such as ELISA detect disease by identifying antibodies or antigens in blood samples. Modern treatments, including monoclonal antibody therapy, involve engineered antibodies designed to bind specific antigens linked to cancers, autoimmune disorders, and infectious diseases. These therapeutic antibodies mimic the body’s natural binding defenses, showing how the immune mechanism can be harnessed to treat illness rather than simply resist it.
A fully detailed vector illustration of this immune concept typically displays antibodies with distinct Y shapes attaching to antigen markers on pathogens, sometimes clustering multiple invaders together to demonstrate how binding neutralizes threats. Arrows trace the subsequent immune cascade — including phagocyte activation, complement involvement, and pathogen removal. Even without text or equations, the picture communicates a powerful biological truth: the immune system relies on molecular recognition to protect the body, and the antibody-antigen relationship is the core of that recognition.
Through this interaction, microscopic proteins become guardians of health. Antigens sound the alarm; antibodies respond with matching precision; and the immune system joins the battle to neutralize, destroy, and remember. In one visual scene, the illustration captures the essence of human immunity — a defense system built not on brute force, but on structural matching, biological intelligence, and memory that protects the body across a lifetime.