Activation of B Cell Process: Antigen Recognition and Immune Response Explained
The activation of B cells is one of the most essential and complex processes in the adaptive immune system, responsible for producing antibodies that neutralize harmful pathogens and provide long-term immunity. Although people often associate immunity with white blood cells in general, B cells execute a remarkable and highly coordinated response that begins with the simple act of recognizing a foreign molecule and ends with the creation of specialized cells that remember the pathogen for future protection. The process is not random or passive; it follows a well-regulated sequence involving antigen recognition, cellular signaling, clonal expansion, antibody production, affinity maturation, and memory formation. At every stage, the body ensures that resources are directed only toward threats rather than innocent or self-derived molecules. Understanding B cell activation reveals why the immune system can defend against countless infectious agents while still maintaining the ability to respond rapidly to pathogens encountered years later. It also helps illustrate how breakdowns in this system can lead to autoimmune disease, allergies, immunodeficiency, or chronic inflammation when the immune system functions incorrectly.
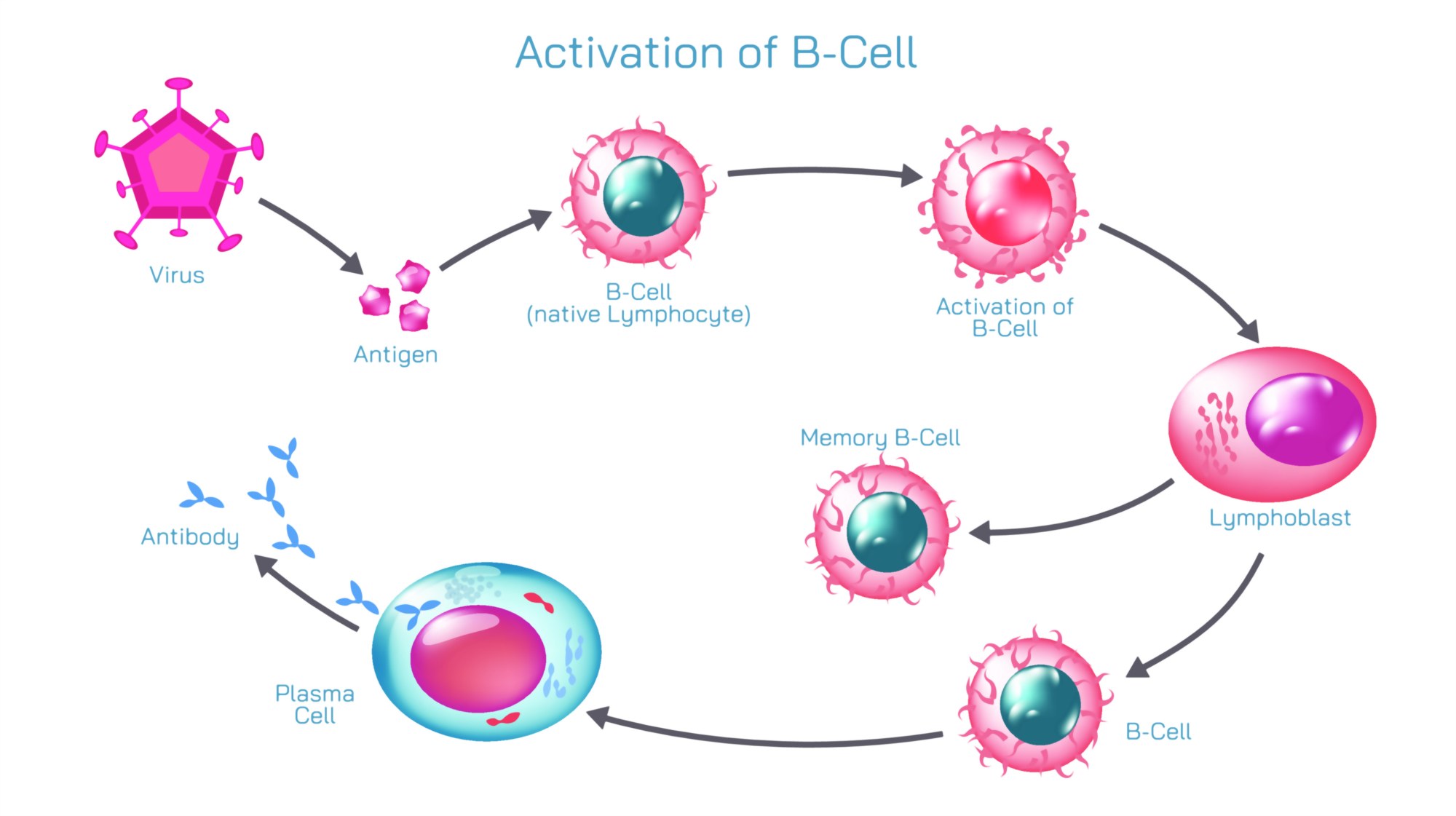
The process begins when a pathogen such as a virus, bacterium, or toxin enters the body and encounters antigen-presenting cells of the innate immune system. These cells degrade the pathogen into fragments known as antigens. Meanwhile, millions of B cells circulate throughout the lymph nodes, spleen, tonsils, and bloodstream, each expressing a unique B cell receptor (BCR) on its surface that recognizes one specific antigen shape. B cells are born in the bone marrow with random antigen-recognition receptors generated by genetic recombination. This genetic mechanism ensures that the body creates an enormous library of B cells, each capable of binding to a different molecular pattern, despite never having encountered that pathogen previously. When a B cell finally meets an antigen that matches its receptor, the BCR binds to it with high specificity, marking the beginning of the activation process. This is a rare event because only a tiny fraction of circulating B cells are born with the right receptor to match any given pathogen, which is why the immune system must maintain such diversity.
The very first step of activation is antigen recognition, but recognition alone is not enough to produce a full immune response. Once the BCR binds to its matching antigen, the B cell internalizes the antigen through receptor-mediated endocytosis. Inside the cell, the antigen is digested into smaller peptides and loaded onto major histocompatibility complex class II (MHC-II) molecules. These antigen–MHC-II complexes are then transported to the surface of the B cell, effectively transforming it into an antigen-presenting cell. This step is essential because it facilitates communication between the adaptive immune system’s two major arms — B cells and T helper cells. Without antigen presentation, the immune system risks accidental activation or overreaction. Presentation functions as a verification mechanism, ensuring that only threats, not harmless molecules, trigger the powerful downstream processes of antibody production.
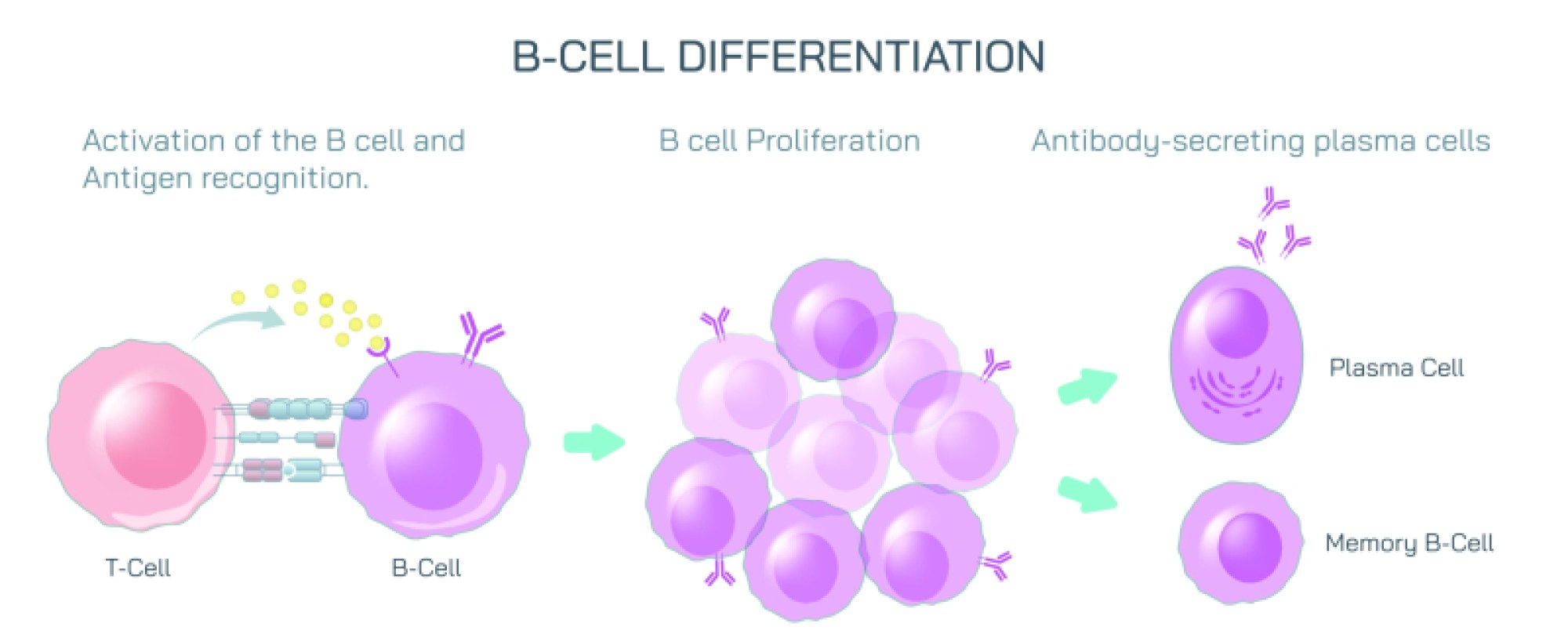
The next stage of B cell activation relies on collaboration with CD4+ T helper cells, a partnership that dramatically enhances the quality and strength of the immune response. When a T helper cell encounters a B cell carrying an antigen–MHC-II complex that matches its own T cell receptor (TCR), the two cells form a highly organized synapse. This interaction allows the T cell to confirm that the antigen is indeed foreign and dangerous. Only then does the T cell release critical cytokines and express the CD40 ligand, which binds to CD40 on the B cell surface. This signal provides the second activation checkpoint, without which the B cell would remain inactive or undergo apoptosis. The need for T cell involvement acts as a powerful safeguard against autoimmunity, because most self-molecules are not presented in contexts that activate T cells. When T cell help is received, the B cell undergoes major internal changes, including the activation of transcription programs, metabolic acceleration, and preparation for rapid proliferation. At this point, the B cell is fully activated and ready to expand and differentiate.
Following T cell help, the activated B cell migrates into specialized micro-environments of lymph nodes or the spleen called germinal centers, where the next transformative stage — clonal expansion — takes place. Through rapid cell division, the single B cell that recognized the antigen multiplies into thousands to millions of genetically identical daughter cells, forming a clone. This expansion ensures that the immune system does not rely on a single cell to fight infection but creates a large army capable of producing substantial quantities of antibodies. However, rather than remaining identical copies forever, B cells undergo an evolutionary-like refinement process known as somatic hypermutation and affinity maturation. During these processes, mutations occur in the variable region of the BCR gene, generating slight differences in antigen-binding strength between daughter cells. Those B cells whose receptors bind the antigen most strongly receive survival signals and continue to divide, while weaker binders die out. As a result, over time the pool of B cells becomes increasingly specialized to bind the pathogen more tightly. This Darwinian selection within germinal centers is one of the reasons why antibody responses become stronger and more precise over the course of an infection or after a vaccine booster.
Once affinity maturation establishes the most effective antigen-binding cells, the B cell population differentiates into two specialized types: plasma cells and memory B cells. Plasma cells are antibody factories — massive, metabolically active cells that migrate to the bone marrow and begin secreting enormous quantities of antibodies into the bloodstream. Unlike the BCR that remains anchored to the cell membrane, antibodies are soluble versions of the same antigen-specific receptor that travel through body fluids to neutralize pathogens from a distance. These antibodies bind to the antigen with high precision and trigger multiple immune defense mechanisms, including neutralizing viral entry into host cells, marking bacteria for destruction by macrophages, activating complement proteins that rupture microbial membranes, and clustering toxins to prevent them from interfering with host tissue. Plasma cells can generate thousands of antibody molecules per second, providing overwhelming immune pressure that rapidly controls or eliminates infection.
While plasma cells combat the active infection, memory B cells act as the immune system’s long-term strategic force. Rather than participating in the immediate response, memory B cells circulate quietly for years or even decades. If the pathogen returns, memory B cells recognize the antigen instantly, bypass the slow activation process, and rapidly trigger antibody production and clonal expansion far faster than the original response. This rapid response is the foundation of immunity, whether acquired naturally after infection or artificially through vaccination. Memory B cells not only allow a faster reaction to future exposure but also produce antibodies with much higher affinity due to their history of affinity maturation. This is why secondary immune responses are typically stronger and prevent reinfection more effectively than primary responses.
The activation of B cells represents a stunning biological balance between aggression and caution. The immune system must produce powerful weapons that can destroy pathogens, yet it must avoid reacting to harmless environmental molecules or the body’s own tissues. When these checkpoints fail, autoimmune disorders can develop when B cells mistakenly target self-antigens; allergies can arise when harmless substances are misinterpreted as dangerous; hypersensitivities can occur when responses become exaggerated; and immunodeficiencies arise when B cells cannot activate or produce antibodies correctly. Conversely, successful B cell activation ensures that the body is armed with precision-targeted defenses both during infection and long after recovery. Antigen recognition, T cell cooperation, clonal expansion, affinity maturation, plasma cell differentiation, and memory formation together form a powerful adaptive system that evolves rapidly within each person’s lifetime.
Ultimately, the activation of B cells is not just a biochemical process — it is the biological foundation of adaptive immunity, long-term protection, and immunological memory. It demonstrates how the immune system learns from threats and becomes wiser with every encounter. Every fever that resolves, every infection that fails to take hold again, and every vaccine that provides lifelong immunity reflects the extraordinary capacity of B cells to detect, adapt, refine, and remember. Understanding this system reveals the deep intelligence built into human physiology — an intelligence that not only protects life in the moment, but also carries forward the memory of survival for the future.