Hund’s Rule Vector Illustration Showing Electron Arrangement, Orbital Filling, and Spin Configuration in Atomic Structure
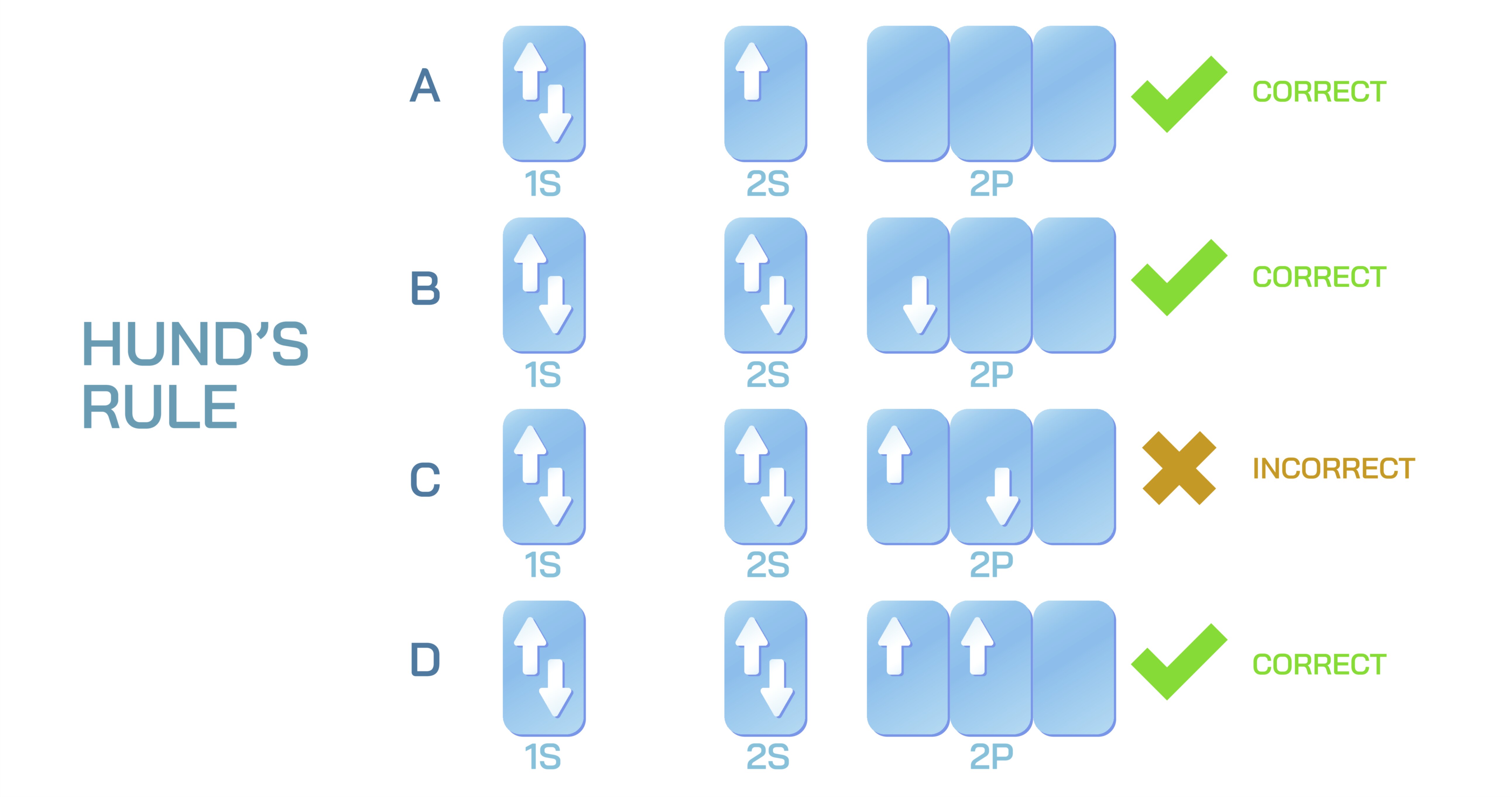
Hund’s rule is a fundamental principle in atomic theory and quantum chemistry, stating that electrons will occupy degenerate orbitals singly with parallel spins before pairing up. This rule governs the electron configuration of atoms, influencing their chemical properties, magnetism, and reactivity. A vector illustration depicting Hund’s rule typically integrates atomic orbitals, electron placement, spin orientation, and orbital energy levels, providing a visually intuitive representation of electron behavior in atoms. By combining labeled orbitals, spin arrows, and stepwise filling sequences, such diagrams allow learners to grasp both the conceptual and structural basis of Hund’s rule in atomic electron configuration.
At the center of the illustration is an energy-level diagram (orbital diagram), showing degenerate orbitals such as the three p-orbitals (px, py, pz), five d-orbitals (dxy, dyz, dzx, dx²−y², dz²), or f-orbitals. Each orbital is represented as a horizontal or vertical box, and labels indicate the specific orbital type. Color-coded boxes or shading can highlight sets of degenerate orbitals, emphasizing that these orbitals have the same energy before electron pairing occurs. Arrows representing electrons are placed inside each orbital to indicate spin orientation, with up and down arrows showing parallel and antiparallel spins, respectively.
The illustration emphasizes the stepwise filling of electrons according to Hund’s rule. In a vector diagram of a p-orbital with three electrons, each orbital receives one electron with the same spin before any orbital is doubly occupied. Arrows or animated highlights indicate the order of filling and the rationale behind maximizing unpaired electrons. Labels may note “parallel spin” or “single occupancy first,” reinforcing that this configuration minimizes electron repulsion and stabilizes the atom.
Spin configuration is clearly represented using up and down arrows. In single-occupancy stages, all arrows point in the same direction (parallel spins), while paired electrons have opposite arrows (antiparallel spins). Insets may magnify individual orbitals to show Pauli exclusion principle compliance, demonstrating that no two electrons in the same orbital can have the same set of quantum numbers, particularly spin. Color-coding may distinguish unpaired electrons from paired electrons, enhancing visual understanding of orbital filling patterns.
Vector diagrams often include comparative orbital diagrams for different elements. For example, carbon with two electrons in the p-orbitals shows each orbital singly occupied with parallel spins, while nitrogen with three p-electrons shows full single occupancy across all three degenerate orbitals. Arrows indicate electron addition sequence, demonstrating consistency with Aufbau principle and Hund’s rule, and labels highlight the number of unpaired electrons, connecting orbital structure to magnetic properties.
To enhance educational clarity, the illustration may incorporate stepwise electron filling animations. Arrows move sequentially from empty orbitals to partially filled ones, emphasizing that electrons occupy each degenerate orbital singly before pairing. Additional callouts may indicate how Hund’s rule contributes to stability, magnetic moments, and chemical reactivity, linking electron configuration to observable atomic behavior.
Vector diagrams may also show energy-level ordering, integrating s, p, d, and f orbitals in a comprehensive layout. Arrows indicate that within a given subshell, electrons fill orbitals singly first before pairing. Insets may demonstrate exceptions in transition metals, where electron repulsion and energy considerations modify the idealized filling sequence, providing learners with a more nuanced understanding of orbital occupancy and spin configuration.
By combining labeled orbitals, spin arrows, electron arrangement, and filling sequence annotations, a vector illustration of Hund’s rule provides a clear, educational depiction of atomic structure. Color coding, magnified insets, directional arrows, and stepwise placement of electrons allow learners to understand both the conceptual rationale and practical application of Hund’s rule in predicting electron configurations.
Ultimately, a vector illustration of Hund’s rule demonstrates the relationship between orbital degeneracy, electron spin, and stability of atoms, linking microscopic quantum principles to macroscopic chemical properties. Through labeled orbitals, spin configurations, sequential electron filling, and comparative examples, the diagram transforms abstract quantum rules into an intuitive, visually engaging, and educational tool for students of chemistry, physics, and material science.