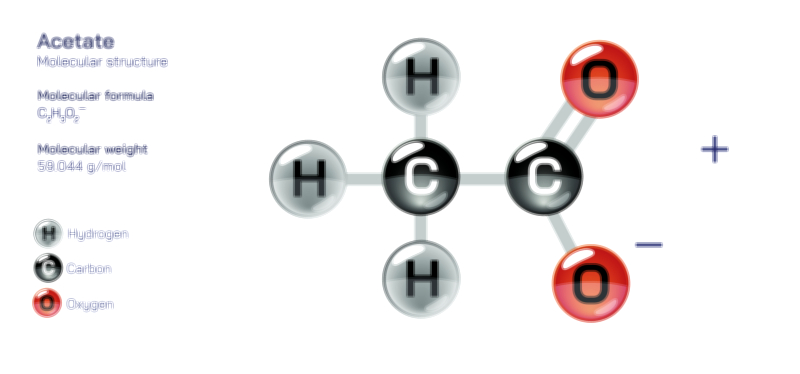
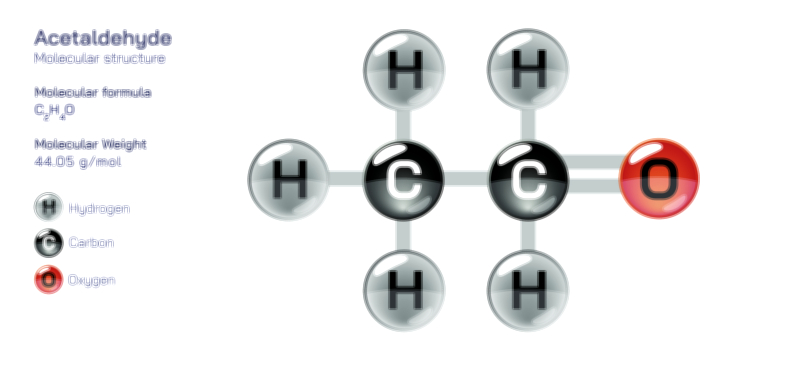
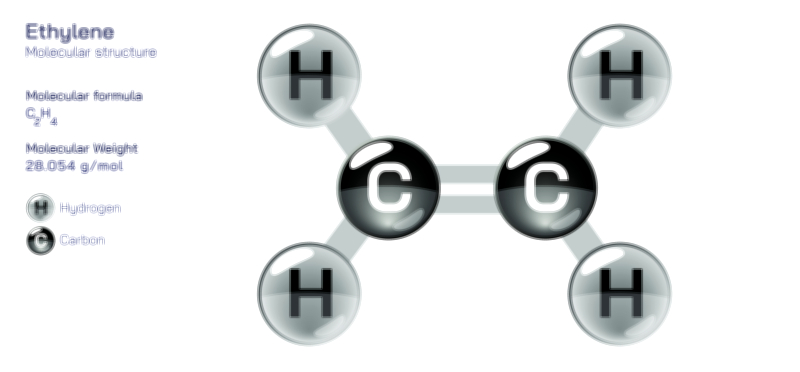
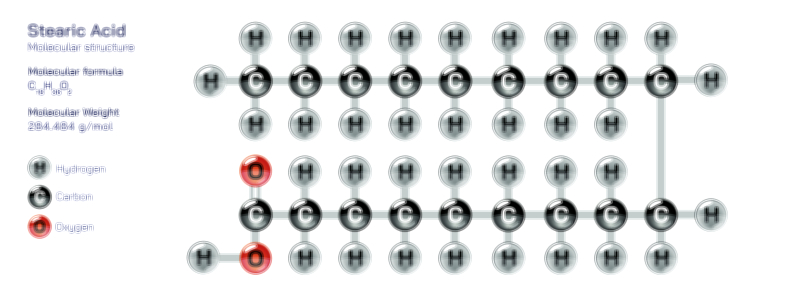
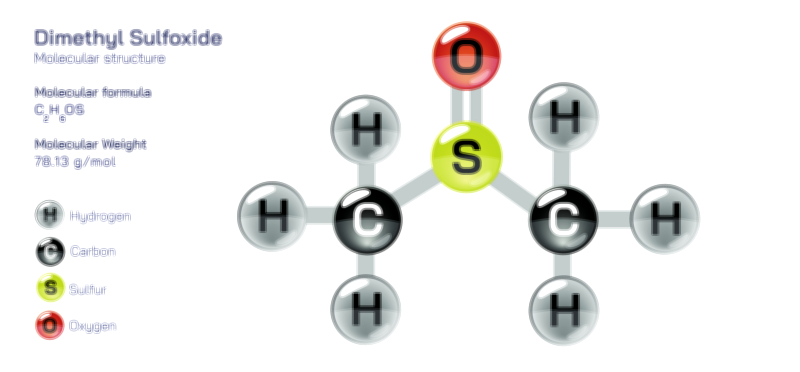
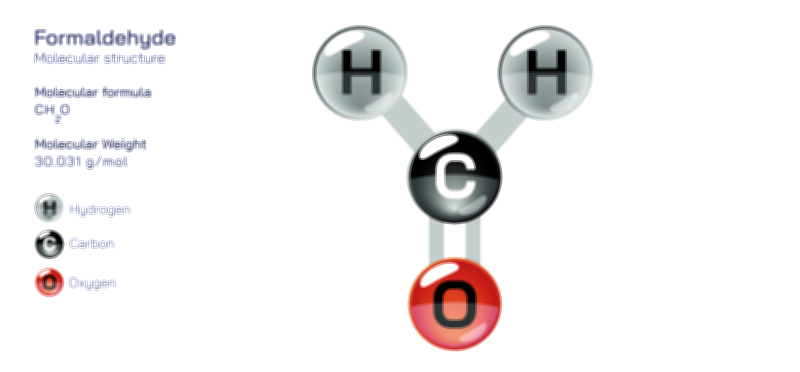
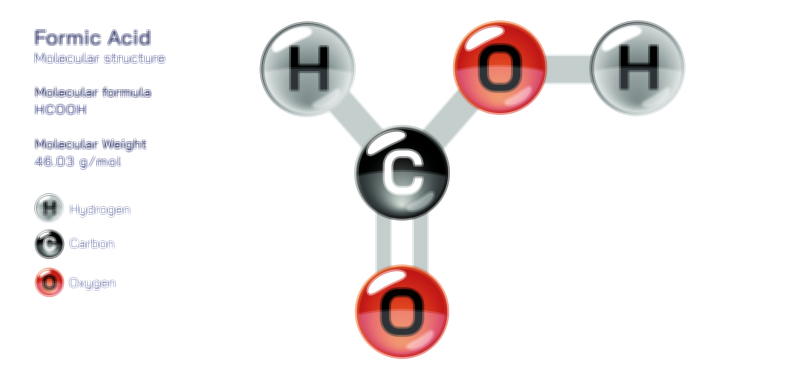
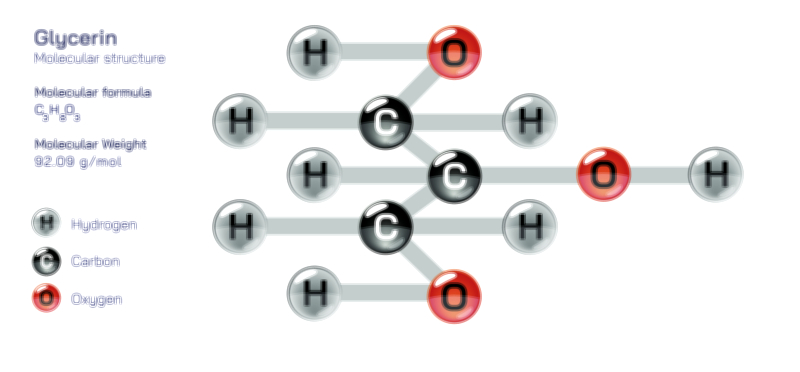
Achiral vs Chiral Vector Illustration Showing Molecular Symmetry, Mirror Image Difference, and Stereochemistry Concept
In stereochemistry—the branch of chemistry that explores the three-dimensional arrangement of atoms in molecules—the distinction between achiral and chiral structures is one of the most fundamental concepts. A vector illustration comparing achiral and chiral molecules side by side helps make a difficult topic instantly intuitive by visually contrasting molecular symmetry and mirror image behavior. While textbook explanations describe chirality in terms of symmetry and superimposability, illustrations allow learners to see what these words mean: chiral molecules are not identical to their mirror images, while achiral molecules are.
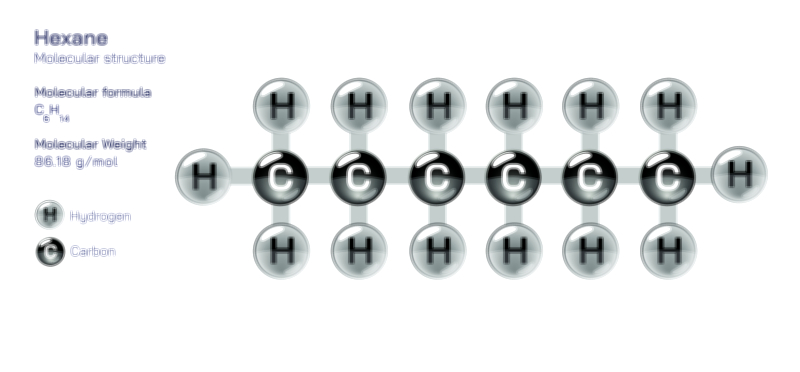
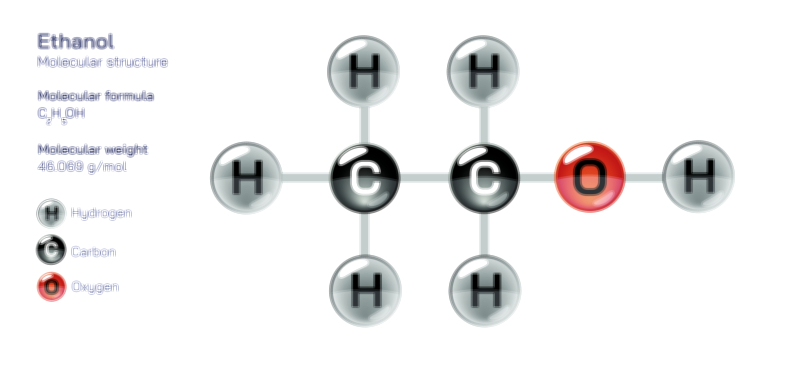
An achiral molecule is one that can be superimposed on its mirror image, meaning both the molecule and its reflection occupy exactly the same spatial arrangement when aligned. This property arises from symmetry, such as a plane of symmetry or a center of symmetry within the structure. For example, methane (CH₄) or 2-propanol contain atomic arrangements that look identical even when mirrored, because the positions of atoms around the central carbon are equivalent. In a vector illustration, the achiral example is typically drawn with its mirror image placed beside it, and arrows or overlays show that rotating one structure results in perfect alignment with the other. The visual symmetry becomes obvious: no unique left or right version exists, and no stereoisomerism is generated.
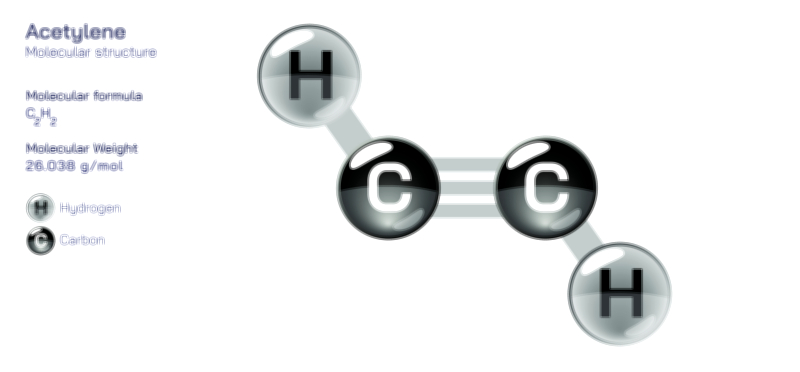
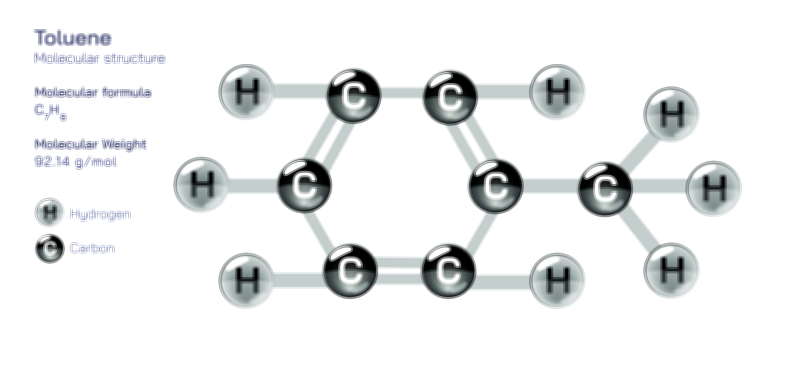
A chiral molecule, however, is defined by the absence of superimposability. Its mirror image cannot be aligned perfectly with the original structure, even after rotation. This lack of symmetry produces two distinct, non-superimposable mirror images, called enantiomers. These enantiomers are like right and left hands: mirror reflections that are not interchangeable. A vector illustration usually depicts a chiral molecule and its mirror image separated by a vertical mirror plane. Even after rotating one structure, certain atoms or functional groups remain mismatched, visually demonstrating why the two forms are fundamentally different. In many organic molecules, this property arises from a chiral carbon—a carbon atom bonded to four different substituents. Highlighting the central stereocenter with color coding or labels reinforces that chirality stems from unique spatial arrangements rather than from differences in atom identity.
Including mirror plane graphics makes the stereochemical concept even clearer. For the achiral molecule, a plane of symmetry divides the structure so that one side is a reflection of the other. For the chiral molecule, no such plane exists. This symmetrical versus asymmetrical property is a powerful way to help students grasp why some molecules form stereoisomers while others do not. In educational vector illustrations, the absence of symmetry in the chiral structure is often emphasized by marking specific substituents that cannot exchange positions without breaking bonds—symbolizing fixed three-dimensional priorities.
Because chirality plays an essential role in chemistry and biology, the illustration may also show the consequences of stereochemistry. Enantiomers often share identical formulas and connectivity yet differ dramatically in odor, biochemical activity, drug interaction, and optical behavior. In polarimetry, for example, chiral enantiomers rotate plane-polarized light in opposite directions, whereas achiral molecules show no rotation. Although the illustration focuses on spatial structure rather than optical testing, references to left-handed vs right-handed stereochemistry visually reinforce the idea that small changes in orientation generate distinct chemical personalities.
For fuller educational impact, vector diagrams of achiral vs chiral molecules typically include:
• 3D projections, such as wedge-dash drawings, to emphasize three-dimensional arrangement
• Mirror planes showing symmetry vs asymmetry
• Flipping or rotation overlays demonstrating superimposability vs non-superimposability
• Color coding of substituents to mark differences in spatial priority
A comparison becomes especially intuitive when the illustration uses hands as a conceptual analogy: left and right hands mirror each other but are not superimposable, just as chiral molecules exist in two distinct mirror-image forms.
Ultimately, a vector illustration of achiral vs chiral molecules showing molecular symmetry, mirror image difference, and stereochemistry concept does far more than compare two structures—it builds the foundation for understanding stereochemistry across organic chemistry, pharmaceutical science, biochemistry, molecular recognition, and sensory biology. Without chirality, drug design, amino acid recognition, fragrance chemistry, catalysis, and enzyme specificity would be impossible to explain. By transforming abstract definitions into intuitive visual geometry, the illustration provides learners, researchers, and educators with an immediate and lasting understanding of how the simplest question—“Can the molecule be superimposed on its mirror image?”—leads to some of the most important distinctions in molecular science.