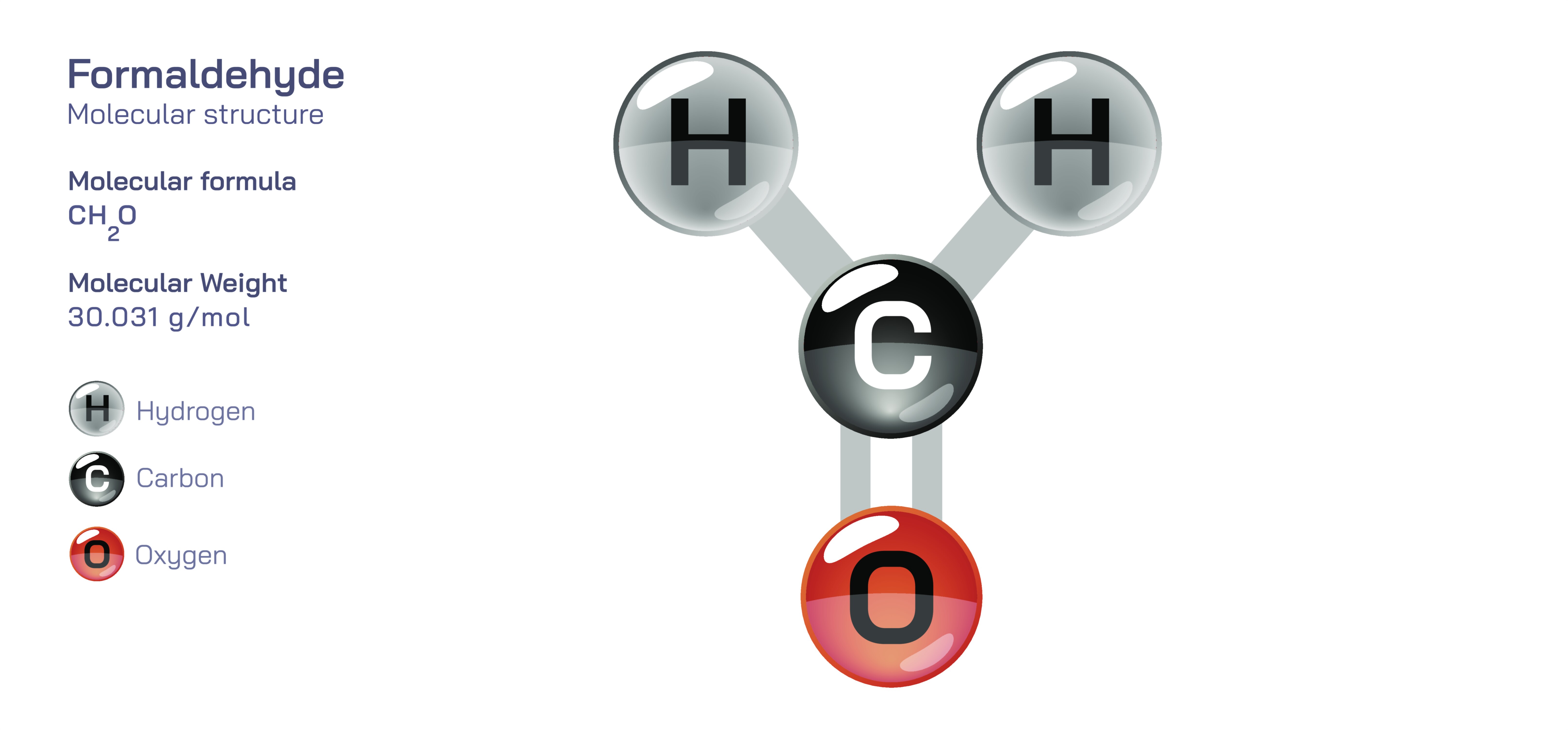
Formaldehyde — Chemical Structure, Properties, and Behavior Explained
Formaldehyde is one of the simplest and most significant organic compounds containing a carbonyl functional group, and although it is often represented in compact structural diagrams that show a central carbon double-bonded to oxygen and single-bonded to two hydrogen atoms, the true meaning of its structure becomes far clearer when explored through detailed conceptual explanation. Understanding formaldehyde begins with its molecular arrangement, but extends into its reactivity, its role in chemical synthesis, its presence in biological systems, and its influence on environmental and industrial processes. Through a richly described narrative, the structural simplicity of formaldehyde transforms into a gateway to broader concepts in organic chemistry and molecular interactions.
At the center of formaldehyde’s molecular identity lies its carbonyl group—an arrangement where a carbon atom forms a double bond with an oxygen atom. This carbonyl group is one of the most important functional groups in all of organic chemistry, and what makes formaldehyde unique is that it is the smallest molecule in which this group appears. The carbon atom in formaldehyde is sp² hybridized, meaning that it forms three sigma bonds in a trigonal planar arrangement. Two of these sigma bonds connect the carbon atom to two hydrogen atoms, while the third sigma bond connects it to the oxygen atom. The double bond contains both a sigma and a pi bond, with the pi bond forming from the sideways overlap of p orbitals on carbon and oxygen. This double bond gives formaldehyde both its planar geometry and its high reactivity, as the carbonyl carbon is left electron-poor due to the strong electronegativity of oxygen.
The geometry of formaldehyde ensures that its entire structure lies in a single plane. The bond angles around the carbon are approximately one hundred twenty degrees, reflecting the symmetrical trigonal planar arrangement. The oxygen atom holds two lone pairs of electrons, contributing to its electron-rich character and strong pull on the shared electrons of the carbon–oxygen bond. This unequal distribution of electrons creates a polarized system: the carbon becomes partially positive, while the oxygen becomes partially negative. This polarity underlies nearly all of formaldehyde’s chemical interactions, making the molecule highly susceptible to reactions involving nucleophiles—species that seek out electron-deficient centers.
Because formaldehyde is so small, its carbonyl carbon is particularly exposed and accessible to other molecules, increasing its reactivity compared with larger aldehydes. Molecules with greater substituent bulk may experience steric effects that block access to the carbonyl carbon, but formaldehyde’s minimal size presents no such hindrance. As a result, nucleophilic addition reactions proceed readily. Water, alcohols, amines, and many other nucleophiles can add to the carbonyl carbon, forming products such as hydrates, acetals, and imines. These reactions form the foundation for much of formaldehyde’s industrial and biological significance, because the molecule readily participates in polymerization and condensation reactions that build larger structures.
One of the most well-known outcomes of formaldehyde’s reactivity is the formation of formalin, a solution of formaldehyde dissolved in water. When formaldehyde mixes with water, the two react to form a hydrate called methanediol. This reaction reflects the molecule’s inherent drive to stabilize its polar carbonyl group by bonding with nucleophilic oxygen atoms from water. Industrial formalin solutions often contain stabilizers to prevent polymerization, because formaldehyde molecules naturally have a tendency to join together, forming long chains or three-dimensional networks. These polymers, which include urea–formaldehyde resins and phenol–formaldehyde resins, are widely used in adhesives, coatings, insulation materials, and composite products. The ability of formaldehyde to form extended polymers arises directly from its structural features, particularly the electron-deficient carbonyl carbon and its responsiveness to nucleophiles.
In living organisms, formaldehyde appears as an intermediate in various metabolic pathways. Although it is toxic at elevated concentrations, trace amounts arise naturally during the metabolism of amino acids and methylated compounds. Because of its high reactivity, biological systems typically convert it rapidly into less reactive substances. Enzymes such as formaldehyde dehydrogenase help detoxify the molecule, converting it into formate, which can then be integrated into cellular metabolic cycles. These detoxification mechanisms reflect the biochemical sensitivity to formaldehyde’s strong electrophilic nature, which, if left unregulated, could disrupt cellular components by attacking nucleophilic sites in proteins, nucleic acids, or membrane structures.
The chemical structure of formaldehyde also allows it to crosslink biological molecules, a property that underlies its use as a preservative and disinfectant. When formaldehyde encounters proteins, it reacts with amino groups to form crosslinks that stabilize the protein structure. This crosslinking process prevents decay and microbial activity, enabling formalin solutions to preserve biological specimens. In the context of disinfection, the same reactivity disrupts microbial function by modifying essential biomolecules, providing formaldehyde broad antimicrobial power. Although effective, this behavior also explains its potential toxicity to humans, since similar reactions may occur in human tissues during high-level exposure.
Understanding formaldehyde’s chemical structure also brings clarity to its behavior in the atmosphere and environment. Due to its high volatility, formaldehyde readily evaporates at room temperature, releasing into the air from various natural and industrial sources. Sunlight and atmospheric oxidants rapidly break it down, forming carbon monoxide, carbon dioxide, and organic radicals. These reactions contribute to the complex mixture of atmospheric chemistry, and they also illustrate the broader principle that highly reactive molecules seldom persist long in natural environments. Even so, indoor environments may contain greater concentrations due to the slow release of small amounts of formaldehyde from manufactured products made with formaldehyde-based resins.
In the realm of synthetic chemistry, formaldehyde serves as a fundamental building block. Its simple structure makes it a common starting material for forming larger, more complex molecules. Many plastics, dyes, solvents, and pharmaceuticals owe part of their molecular frameworks to reactions initiated by formaldehyde. Because the molecule can react at multiple points—especially with compounds that contain amine or phenol groups—it serves as a versatile reagent capable of forming structured polymers, crosslinked networks, or tailored organic intermediates.
Examining formaldehyde’s electron distribution reveals vital insights into why it behaves the way it does. The double bond between carbon and oxygen creates a strong, polarized region in the molecule. Oxygen’s electronegativity draws electrons toward itself, leaving the carbon electron-deficient and ready to accept electron density from incoming nucleophiles. This dynamic sets up a cycle of interactions where bonds form and break with relative ease, making formaldehyde one of the most aggressive electrophiles among simple organic compounds. Its small size further accentuates this tendency: there are no bulky substituents to stabilize the carbon or shield it from reaction, allowing nearly unrestricted access to reactive species.
Even the bonding arrangement in formaldehyde contributes to its odor and sensory detection. The molecule’s volatility and relatively low molecular weight allow it to vaporize readily, enabling human sensory receptors to detect its pungent smell at extremely low concentrations. This sensitivity reflects both formaldehyde’s molecular mobility and its widespread presence in natural and synthetic environments. Wood combustion, forest fires, metabolic pathways, cooking processes, and industrial emissions all produce small amounts of formaldehyde, making it a familiar yet sometimes overlooked component of everyday atmospheric chemistry.
In materials science, the structural behavior of formaldehyde determines the properties of the polymers it helps to create. When combined with urea or melamine, formaldehyde forms highly crosslinked networks that provide hardness, durability, and resistance to heat. These characteristics make such polymers ideal for construction materials, kitchenware, and molded components. The rigidity of these materials arises from the intermolecular crosslinks formed during polymerization—a direct consequence of formaldehyde’s chemical structure and its ability to bond repeatedly with reactive sites on other molecules.
Despite its usefulness, formaldehyde’s reactive nature requires careful handling and regulation. Because it can bind strongly to biological macromolecules, high exposure may affect respiratory systems, vision, and skin. Regulatory agencies around the world place strict limits on allowable exposure levels, particularly in occupational settings and indoor environments. These regulations underscore the delicate balance between formaldehyde’s chemical utility and the need to manage its interactions with biological systems.
Ultimately, the structure of formaldehyde—simple yet highly reactive—serves as a key to understanding its behavior across chemistry, biology, industry, and the environment. The molecule’s trigonal planar geometry, polarized carbonyl group, and exposed carbonyl carbon define a pattern of interactions that make it both a powerful synthetic tool and a compound requiring respectful handling. By exploring formaldehyde through a richly detailed conceptual lens, one gains a deeper appreciation of how its structural features influence its reactions, applications, and presence in the natural and synthetic worlds.