Osmosis – Biological Process of Water Movement, Cellular Balance, Membrane Transport, Homeostasis, and Its Essential Role in Living Organisms
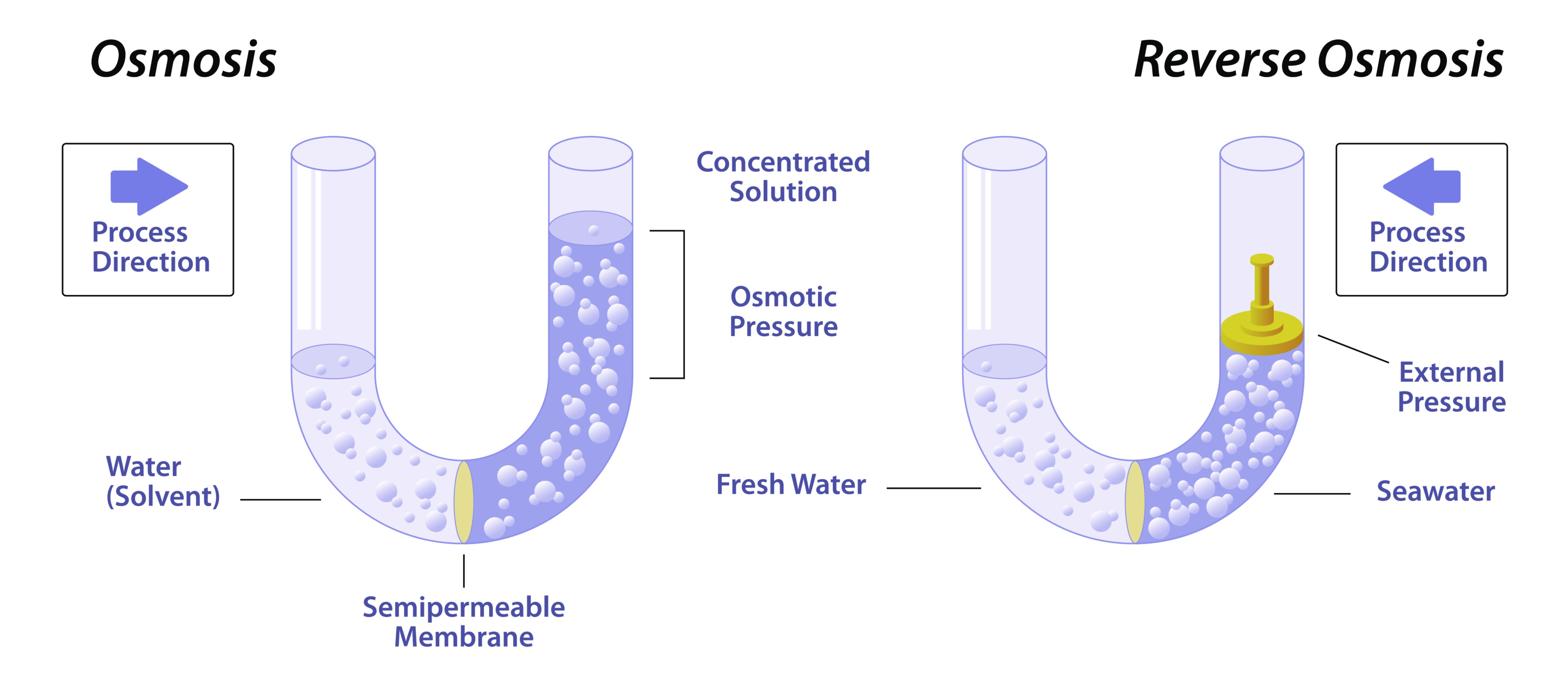
Osmosis is one of the most fundamental biological processes that supports life at the cellular level, a natural movement of water that occurs without energy expenditure yet has immeasurable influence on the structure, stability, and function of living organisms. It is defined as the passive movement of water molecules from a region of lower solute concentration to a region of higher solute concentration across a selectively permeable membrane. Rather than occurring haphazardly, osmosis follows a predictable biological pattern driven by the principle of equilibrium: water moves toward the area containing more solutes in an attempt to equalize concentrations. The membrane involved in this process is not just a barrier but a regulatory gatekeeper that allows the passage of water while restricting most solutes. All living cells—from bacteria and fungi to plants and animals—depend on osmosis to maintain internal fluid balance, exchange nutrients, transport waste, stabilize shape, and protect structural integrity. Without osmosis, the chemistry of life would be impossible because cells would not be able to regulate the delicate interactions between internal and external environments that sustain metabolism and prevent structural damage.
The mechanics of osmosis reveal deeper biological meaning beyond the movement of water. Inside and outside a cell, fluids contain varying amounts of dissolved substances such as ions, sugars, proteins, and salts. Water does not move randomly; it moves based on concentration differences, and these differences create conditions described as hypotonic, hypertonic, and isotonic. A hypotonic solution has a lower solute concentration compared to the inside of the cell, causing water to flow inward. If the inward movement becomes excessive, animal cells risk swelling to the point of bursting because they lack a rigid outer wall, whereas plant cells counter this pressure using their strong cell walls, creating the healthy turgid state essential for plant structure. A hypertonic solution has a higher solute concentration outside the cell, causing water to move out and leading to cell shrinkage. In animal cells, excessive water loss causes shrinking called crenation, while in plant cells it causes plasmolysis, where the cell membrane pulls away from the cell wall. An isotonic environment has equal solute concentration inside and outside, allowing water to move in and out at the same rate, maintaining volume and shape. These three osmotic conditions demonstrate that water movement is not random but biologically purposeful, defining the physical state of cells according to the chemical environment around them.
Osmosis is also crucial in the context of homeostasis, the stable internal environment necessary for life. Every cell must regulate its water content with precision, and organisms devote extensive physiological mechanisms to ensuring that extracellular fluids stay within ranges that prevent osmotic damage. In humans, this balance is maintained through processes such as thirst regulation, kidney filtration, hormonal control via antidiuretic hormone (ADH), and salt balance maintenance through aldosterone. These regulatory pathways demonstrate that osmosis is not simply a basic property of water but a central pillar of physiological survival. The kidneys in particular illustrate osmosis at its finest complexity: in the nephron, water is reabsorbed through osmotic gradients created by dissolved ions in the surrounding tissues. Without osmotic reabsorption, the body would lose enormous amounts of water through urine, leading rapidly to dehydration and death. The same osmotic principles that determine the movement of water across a single cell membrane scale upward to influence the hydration state of an entire organism.
The role of osmosis in plant biology is equally indispensable. Plants rely heavily on osmotic pressure to absorb water from the soil through root cells. Root hair cells develop osmotic gradients that draw water inward, and once water enters, it travels through tissues via osmosis and cohesion-tension forces to reach stems and leaves. In leaves, osmotic pressure supports the turgidity that keeps leaves expanded for capturing sunlight, allowing photosynthesis to occur at maximum efficiency. During drought, soil becomes hypertonic relative to root cells, preventing water entry and causing wilting—a visible sign of disrupted osmotic balance. Even the opening and closing of stomata, which regulate gas exchange and water loss, depend on osmotic changes within guard cells. When water enters these guard cells through osmosis, they swell and open stomatal pores; when water exits, the pores close. Thus, osmosis links environmental water availability to plant metabolism, growth, and survival, integrating cellular processes with ecosystem conditions.
At the microscopic level, osmosis affects metabolic processes through its impact on molecular diffusion, cellular respiration, waste removal, and nutrient uptake. When water moves into a cell, it alters internal concentrations of solutes, which influences biochemical reaction rates because enzymes operate most efficiently in water-based environments with specific solute ranges. Osmosis indirectly controls cell pH, ion concentration, and electrolyte balance, which in turn regulate electrical signaling in neurons and muscle contraction. During cellular respiration, metabolic byproducts dissolved in the cytoplasm must be removed through transport systems that require water as a solvent, making osmotic balance essential for maintaining optimal intracellular chemical composition. Osmosis ensures that water continuously enters and leaves cells in a rhythm that supports sustained biochemical life rather than allowing toxic metabolic buildup or dilution.
Osmosis also plays a major role in real-world medical applications. Intravenous (IV) fluids must be carefully formulated to match osmotic conditions inside human cells. Administering a hypotonic or hypertonic fluid accidentally can cause dangerous swelling or shrinking of red blood cells, leading to circulation problems or tissue damage. For this reason, medical IV solutions are usually isotonic to protect cellular integrity. Oral rehydration therapy—a life-saving treatment for dehydration—works specifically by leveraging osmotic principles to draw water rapidly into the bloodstream by pairing salts and glucose in optimal proportions. Dialysis machines for kidney failure patients rely on controlled osmosis and diffusion to remove waste from the bloodstream across a semi-permeable membrane. These medical applications show that understanding osmosis is not simply academic; it is foundational for designing life-saving treatments.
Even at the organismal level, diseases and disorders can arise when osmotic balance fails. Dehydration reduces the water available for osmotic reactions and causes cells to shrink, leading to confusion, fatigue, organ dysfunction, and eventually severe medical crises. Overhydration dilutes solutes in the blood, creating dangerously hypotonic conditions and causing cells—especially brain cells—to swell. In diabetes, high blood glucose increases osmotic pressure in the bloodstream, forcing more water out of cells and increasing urinary water loss, explaining why frequent urination and thirst are hallmark symptoms. These clinical manifestations demonstrate that living organisms exist in constant osmotic negotiation with their environment, and disruptions to this balance immediately threaten survival.
Osmosis holds educational significance because it teaches students that water movement is not passive but deeply purposeful. It underscores how chemical gradients govern biological structure and shows how cells avoid randomness by using physical laws to maintain order. It teaches that life sustains itself not only through chemical reactions but also through regulated transport that protects those reactions from destabilizing conditions. Osmosis connects molecular motion to structural integrity and transforms a simple principle—water moves toward higher solute concentration—into a powerful explanation for countless biological phenomena. Understanding osmosis enables learners to conceptualize how tiny invisible particles create large-scale biological outcomes, bridging scales from membrane physiology to whole-body function.
Ultimately, osmosis is one of the most elegant demonstrations of biological design: without consuming energy, cells utilize natural fluid movement to sustain shape, transport necessary substances, prevent toxic accumulation, and maintain internal stability in a constantly changing world. It is a reminder that life depends not only on what substances exist inside cells, but on the dynamic balance between inner and outer environments. Osmosis shows that homeostasis is not a static state but a continuous process, and through this lens, students begin to appreciate that biological systems are governed by beautifully simple principles operating with extraordinary impact.