Barium Bromide — Understanding the Structure and Nature of an Ionic Chemical Compound
The Fundamental Identity of Barium Bromide as an Ionic Substance
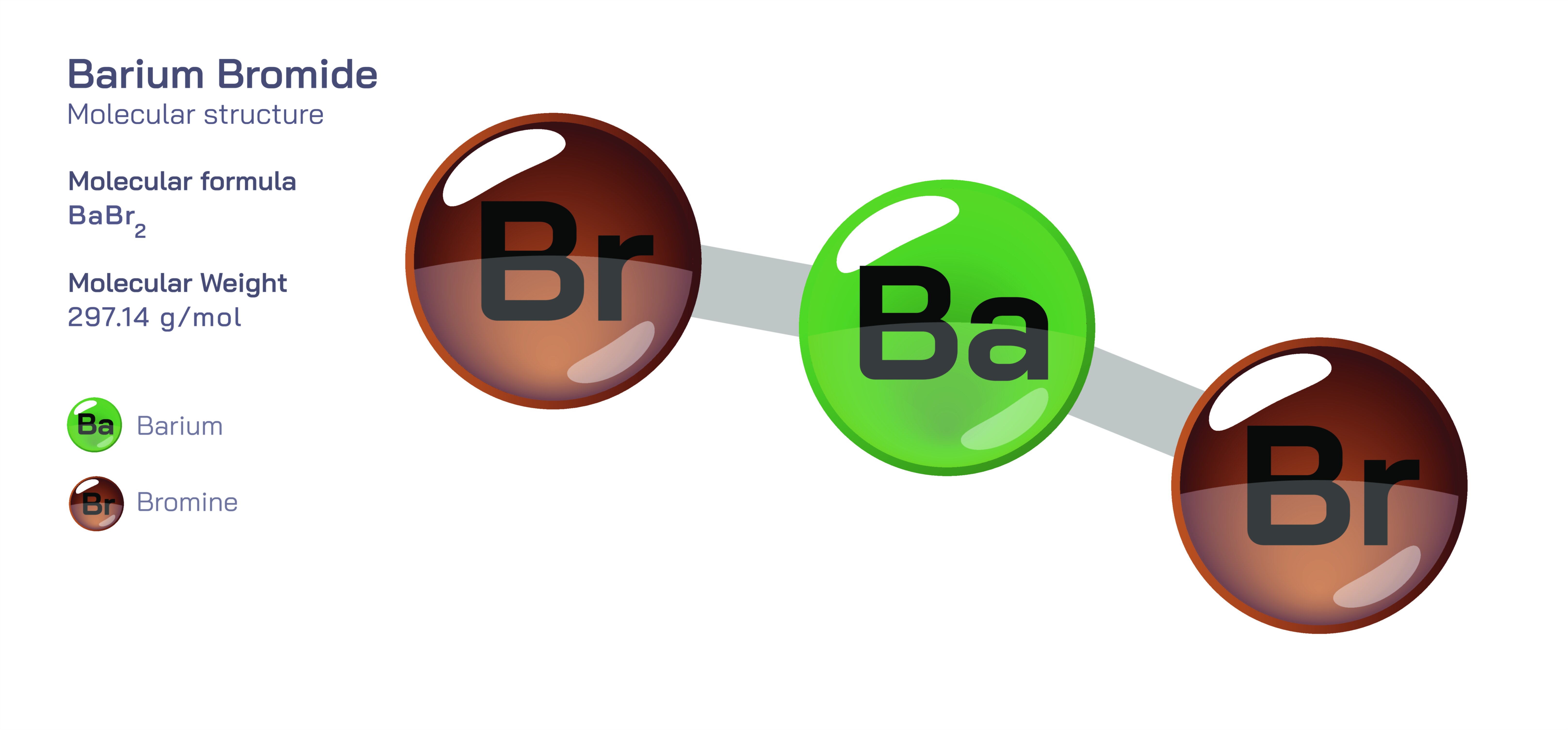
Barium bromide is a classic example of an ionic compound, formed through the strong attraction between positively charged and negatively charged ions. It is created when barium, a reactive metallic element, combines with bromine, a non-metal known for its ability to form stable negatively charged ions. The interaction between these two elements results in a compound with a firm internal structure, where the ions arrange themselves in an orderly pattern that reflects the stability of their bond. Unlike molecules made from shared electron pairs, ionic compounds like barium bromide rely on the complete transfer of charge between their components. This transfer results in ions with opposite charges that lock together through strong electrostatic forces. Understanding this compound provides insight into the behavior of ionic substances and the principles that govern how metals and non-metals interact to form stable chemical structures.
How the Ions Form and Create a Stable Structure
Barium bromide’s formation begins with the ability of barium atoms to easily release their outer electrons, which allows them to adopt a stable, positively charged form. Bromine atoms, on the other hand, strongly attract additional electrons to achieve a stable configuration. When these two tendencies meet, the electron transferred from barium becomes attached to bromine, transforming the neutral atoms into oppositely charged ions. This creation of a positive barium ion and negative bromide ions is the foundation of the compound’s structure. Once formed, these ions arrange themselves into a repeating pattern that extends through the solid material. The powerful attraction between their opposite charges holds the structure firmly together, making the compound stable under a wide range of conditions.
The Crystal Arrangement and Its Influence on Properties
In its solid state, barium bromide forms a crystal lattice that reflects the organized nature of ionic bonding. The ions are arranged so that each positive ion is surrounded by negative ions, and each negative ion is surrounded by positive ions. This symmetrical pattern distributes the attractive forces evenly across the entire structure, creating both physical strength and internal stability. As a result, barium bromide exhibits the typical characteristics of ionic compounds. It forms hard crystals, remains solid at high temperatures, and dissolves readily in water. These traits arise because the ionic network, while strong, separates in water as the ions become surrounded by water molecules. The crystalline structure also allows the compound to conduct electricity when dissolved or melted, since the ions are free to move. This blend of rigidity and reactivity illustrates the balanced nature of an ionic lattice.
How Barium Bromide Behaves in Water and Solutions
When barium bromide is placed in water, the strong attraction between water molecules and the ions causes the crystal lattice to break apart. Each ion becomes surrounded by water molecules, enabling them to move freely through the solution. This separation of ions is what allows dissolved barium bromide to conduct electricity, since the positive and negative ions can carry electrical charge from one point to another. The ability of the compound to dissolve and form electrically active solutions makes it useful in analytical chemistry and laboratory applications. This behavior also illustrates one of the defining characteristics of ionic compounds: their tendency to separate into fully independent ions once they enter a liquid environment.
The Role of Barium Bromide in Chemical Processes and Applications
Barium bromide is used in several fields due to its predictable ionic behavior and ease of dissolution. In laboratories, it is often employed to prepare other barium compounds or to study reactions involving heavy metal ions. Its role as a source of bromide ions makes it valuable in chemical synthesis and analytical testing. Barium compounds can also be used in specialized industrial processes, including materials testing and certain purification techniques. While barium itself must be handled with caution due to its reactive nature, the ionic form found in barium bromide is more stable, allowing it to be used safely in controlled settings. Each application relies on the compound’s stable ionic structure and its ability to interact readily with water.
The Relationship Between Ionic Bonds and Stability in Barium Bromide
The strength of barium bromide as a compound lies in the nature of its ionic bonds. Because these bonds are formed from the attraction between full positive and full negative charges, they are stronger than many other types of chemical interactions. This strength gives the compound its high melting point and solid crystalline form. It also ensures that barium bromide maintains its structural integrity under conditions that might weaken less robust compounds. Yet the same attraction that creates a firm lattice also makes the compound responsive to water, which can easily disrupt the arrangement and separate the ions. This special balance between solidity in its dry state and flexibility in solution highlights the dual nature of ionic bonding.
Why Understanding Barium Bromide Helps Explain Ionic Chemistry
Studying barium bromide provides a clear example of how metals and non-metals combine to form stable ionic structures. It showcases the principles behind ionic bonding, crystal formation, and solution behavior, offering insight into how many essential compounds behave in nature and in industry. Through its structure and properties, barium bromide illustrates the fundamental concepts that govern ionic chemistry: the formation of charged particles, the organization of ions into symmetrical lattices, and the dynamic interactions that occur when such compounds dissolve. Understanding this compound deepens scientific appreciation for how simple atomic tendencies can produce materials with strong, useful, and predictable characteristics.