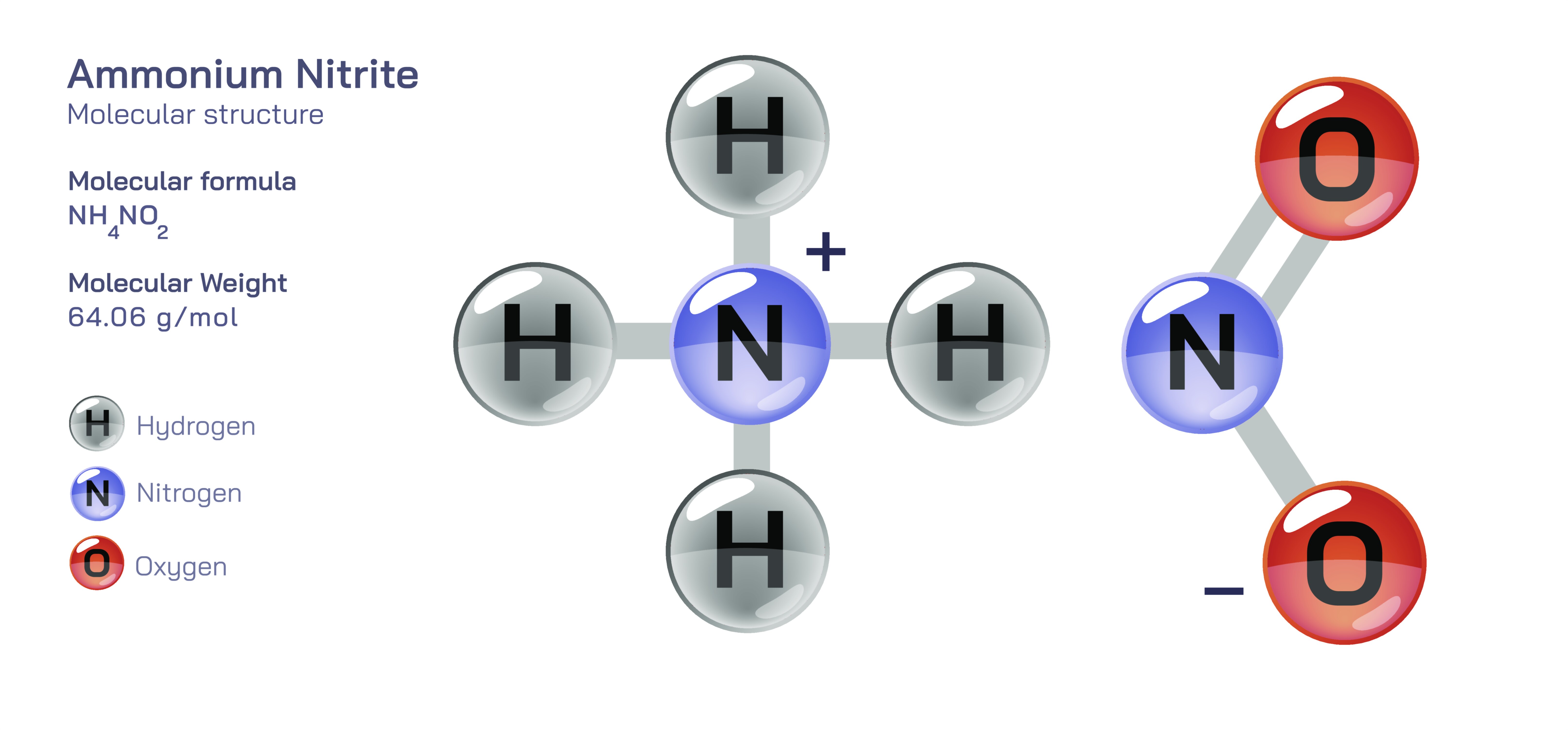
Ammonium Nitrite — Molecular Structure, Properties, and Chemical Behavior
Understanding the Nature of Ammonium Nitrite
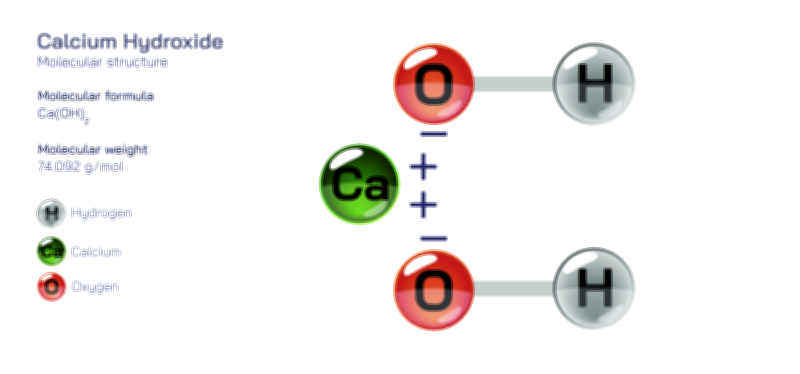
Ammonium nitrite is a chemical compound composed of ammonium and nitrite ions, forming a substance that is structurally simple yet extremely unstable. Although it can exist briefly under very controlled conditions, it is prone to rapid decomposition and therefore does not appear commonly in pure form outside laboratory studies. In essence, it is a salt formed from a positively charged ammonium group and a negatively charged nitrite group, but the combination of these two reactive species creates a material that tends to break down almost as soon as it forms. Because of this instability, ammonium nitrite is known more for its behavior and reactions than for practical uses. When depicted in a vector illustration, the molecule is usually shown with its two main components connected through ionic attraction, creating a conceptual image that highlights its basic composition even though the compound itself is difficult to isolate in the real world.
The Structure of the Ammonium Ion in This Compound
At the heart of ammonium nitrite is the ammonium ion, a stable, positively charged grouping made of one nitrogen bonded to four hydrogen atoms in a compact, symmetrical arrangement. This form appears commonly in many safe and stable ammonium salts, and in diagrams it is often represented as a central nitrogen surrounded evenly by four hydrogens to emphasize its balanced shape. In the context of ammonium nitrite, the ammonium ion behaves exactly as it does in other compounds: it acts as the cation that binds to the nitrite anion through electrostatic attraction. In a vector diagram, this relationship is usually indicated by placing the ammonium group near the nitrite group, often with subtle charge indicators to show how they associate. Even though the ammonium component is stable on its own, its presence in ammonium nitrite contributes to a delicate balance that can easily collapse due to the reactive nature of the nitrite partner.
Characteristics of the Nitrite Component and Its Role in Instability
The nitrite ion brings a completely different personality to the compound. It is reactive, sensitive to heat, and prone to participating in transformations that release gaseous products. In ammonium nitrite, this reactivity creates a situation where the two ion types work against the stability of the compound. The nitrite ion tends to promote decomposition because it can rearrange itself under mild conditions, producing nitrogen-based gases as it breaks apart. This decomposition is not slow or gentle but often sharp and rapid, which is why ammonium nitrite must be handled with extreme caution if it is prepared in the laboratory. In a vector illustration, the nitrite ion is often shown as a compact cluster of oxygen and nitrogen arranged in a bent shape that suggests its reactive nature, providing a visual reminder of why the compound is so short-lived.
Behavior and Decomposition Tendencies in Real Conditions
Ammonium nitrite decomposes very easily, even at room temperature or when left undisturbed for a short period. When it breaks down, it forms nitrogen gas and water, releasing the gas in a way that can be sudden and sometimes dangerous if the material accumulates. This tendency to collapse into simpler products means that ammonium nitrite rarely exists for long and is generally prepared only moments before it is needed in laboratory experiments. Its decomposition is clean and leaves no solid residue, which makes it unique among unstable salts. The process is so spontaneous that the compound is never found in storage or transport scenarios and must always be managed with immediate use in mind. When illustrated, vector diagrams often use directional arrows or fading structures to indicate the fleeting nature of the compound and the way it transitions rapidly into gases.
How It Is Used and Why Its Applications Are Limited
Due to its instability, ammonium nitrite has very limited direct use. It is sometimes produced in laboratory settings for controlled demonstrations of decomposition or as an intermediate in specific chemical processes that require rapid generation of nitrogen gas. However, its hazardous behavior prevents it from being applied in large-scale industrial settings or in products requiring chemical stability. Its primary value lies in academic and theoretical chemistry, where it helps demonstrate concepts related to ionic pairing, decomposition reactions, and the limits of compound stability. The compound also serves as an educational example of how two ions that appear perfectly balanced on paper can create a highly unstable material when combined in reality.
Representing Ammonium Nitrite in Vector Illustration
A vector illustration of ammonium nitrite usually highlights its simple ionic structure while subtly hinting at its fragile nature. The ammonium ion is depicted with its characteristic symmetrical arrangement, while the nitrite group appears with its bent, reactive structure. The two parts are shown near one another to represent their ionic association, often with slight graphical cues that point to the compound’s tendency to break apart. These visuals help convey the idea that ammonium nitrite is meaningful as a structural concept even though it resists practical existence. The illustration becomes not just a depiction of a molecule, but a representation of a chemical relationship that is inherently unstable and short-lived.