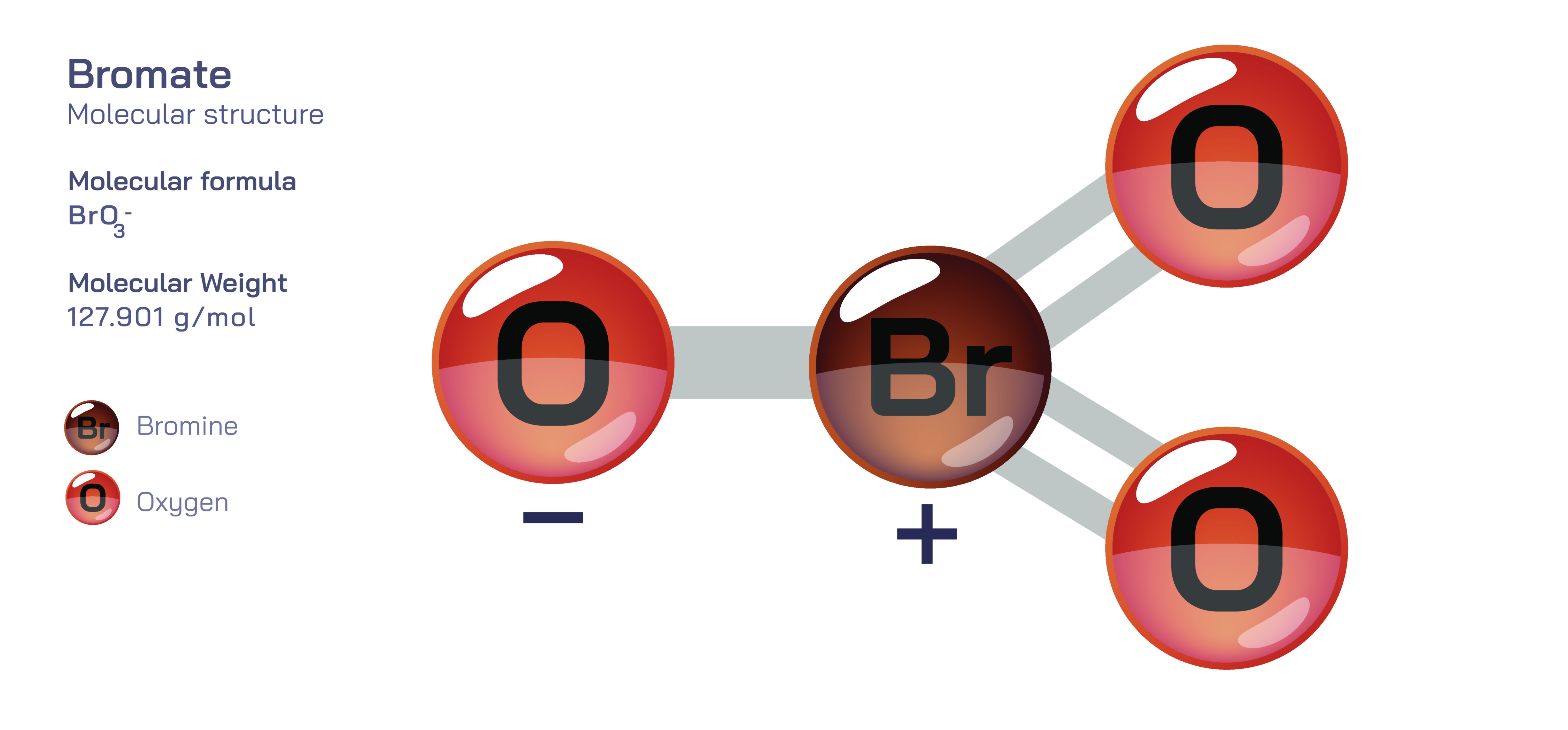
Bromate — Inorganic Oxoanion Compound
Bromate is an inorganic oxoanion that occupies an important position within the broader chemistry of halogens, particularly due to its oxidizing nature and the complex reactions it undergoes in various environmental, industrial, and biological contexts. Understanding bromate requires exploring its behavior as an oxygen-containing anion formed from bromine, a halogen known for its versatile and reactive chemistry. Even without the use of mathematical formulas or symbolic structures, the essence of bromate can be understood through the interactions it forms with surrounding molecules, the transformations it undergoes as part of the bromine redox cycle, and the conditions under which it appears in nature and in human-engineered systems. Its formation often involves processes where bromide—its reduced counterpart—undergoes oxidation in the presence of strong oxidants such as ozone, chlorine dioxide, or other high-energy chemical conditions. Because of this, bromate serves as both a diagnostic indicator of oxidative processes and a species with regulatory importance, especially in drinking-water systems where its presence needs to be carefully monitored.
To grasp the significance of bromate, it helps to appreciate its position within halogen chemistry, where each element can exist in multiple oxidation states depending on environmental conditions. Bromine, in particular, transitions through states ranging from bromide, which is stable and relatively nonreactive, to bromine intermediates, and ultimately to higher oxidation-state species like bromate. These shifts are influenced by factors such as pH, temperature, available oxidants, and the presence of other ions. Bromate is among the more oxidized forms, meaning it has a strong tendency to pull electrons away from other molecules. This oxidizing capability gives it both functional usefulness and potential hazards, depending on the setting in which it appears. In industrial environments, oxidizing agents like bromate can play useful roles in manufacturing and analytical chemistry. But in natural and biological systems, high oxidation-state halogens can interfere with cellular processes, prompting concerns about safety and environmental persistence.
The formation of bromate as a byproduct in water treatment has become one of the most widely studied aspects of its chemistry. When ozone is used to disinfect water—an increasingly common choice due to its efficiency in eliminating pathogens—it can inadvertently convert trace bromide naturally present in source water into bromate. This transformation illustrates how a beneficial process can have unintended consequences when reactive species are involved. The conversion depends on factors such as the concentration of bromide, the amount of ozone used, the contact time, the water’s alkalinity, and the presence of organic matter. Because bromate is far more reactive and potentially harmful than bromide, regulatory bodies and water-treatment engineers devote significant attention to controlling its formation through optimized process design. Strategies include adjusting pH, modifying ozone dosage, or using alternative disinfection combinations to minimize the oxidation pathway that leads to bromate. The understanding of these pathways stems from extensive research into the behavior of halogen oxoanions and how they respond under various chemical conditions.
Beyond water treatment, bromate plays a notable role in analytical and industrial processes due to its strong oxidizing properties. In laboratories, bromate can serve as a standardized oxidizing agent in redox titrations because its behavior is predictable and stable under controlled conditions. This makes it useful for determining the concentration of reducing agents in solutions, allowing chemists to carry out precise quantitative analyses. In some manufacturing processes, bromate-containing compounds were historically used in food-related applications, particularly in flour treatment, because the oxidizing effect could strengthen dough and improve its handling properties. However, growing awareness of the risks associated with bromate exposure led many regions around the world to restrict or eliminate its use in food contexts. This shift highlights the evolving relationship between industrial practices and the scientific understanding of chemical impacts on human health.
In environmental chemistry, bromate represents a reactive species that interacts with natural waters, sediments, and organic matter in complex ways. Though it is relatively stable compared to less oxidized intermediates, bromate can undergo slow reduction under the right conditions, forming bromide or other bromine-containing species. These transformations depend on factors such as microbial activity, the presence of natural reductants, and the chemical composition of the environment. Because its persistence varies, understanding bromate’s environmental behavior is important for assessing how long it remains in aquatic systems and how far it may travel before undergoing transformation. Such knowledge informs environmental monitoring, ecological risk assessment, and strategies for remediation in cases where bromate contamination arises.
Bromate also carries importance in the study of reaction kinetics, especially in nonlinear chemical systems where oscillating reactions, pattern-forming processes, and complex dynamic behaviors emerge. One of the most famous examples is the Belousov–Zhabotinsky reaction, a system in which bromate plays a key role as an oxidizing agent driving rhythmic color changes and spatial patterns. Although this reaction is studied primarily for its theoretical and mathematical interest, it demonstrates how bromate participates in intricate chemical cycles, interacting with organic molecules and catalysts in ways that produce mesmerizing and scientifically valuable dynamic behavior. These oscillating reactions reveal the deep interconnectedness of reaction rates, redox cycles, and chemical feedback loops, providing insights that extend into biological modeling, materials science, and the study of self-organizing systems.
From a toxicological perspective, bromate’s position as a strong oxidant means it has the potential to damage biological tissues when exposure occurs. Its interaction with cellular components can disrupt normal biochemical pathways, particularly those involving proteins, enzymes, and DNA. Because of these concerns, bromate is subject to strict regulation in drinking water and food. Regulatory limits differ across countries, but the underlying principle remains consistent: bromate must be kept at levels low enough to prevent long-term health risks. Continued scientific research focuses on better understanding how bromate forms, how it behaves in the body, and how environmental or industrial systems might generate or eliminate it. These insights guide public policy, engineering practices, and public-health strategies aimed at protecting consumers.
What makes bromate especially fascinating is how it connects multiple scientific domains. In environmental chemistry, it represents a potential contaminant that must be controlled. In industrial chemistry, it serves as a useful but carefully managed oxidizing species. In analytical chemistry, it provides a reliable component for standardized measurements. In physical chemistry and theoretical studies, it contributes to the understanding of dynamic systems and non-linear reaction processes. This wide-ranging relevance underscores how a single anion can influence diverse fields through its chemical properties, reactivity, and behavior under different conditions.
Ultimately, bromate serves as a reminder of the importance of understanding not just chemicals themselves but the pathways through which they are formed, transformed, and transported. It highlights the delicate balance required in technological and environmental systems, where beneficial processes like water disinfection must be carefully optimized to avoid generating unintended byproducts. Through ongoing research, regulation, and scientific advancement, societies continue to refine their approach to managing bromate and related compounds. This ensures that the powerful chemistry of halogens can be harnessed safely and effectively while minimizing risks to human health and the natural world.