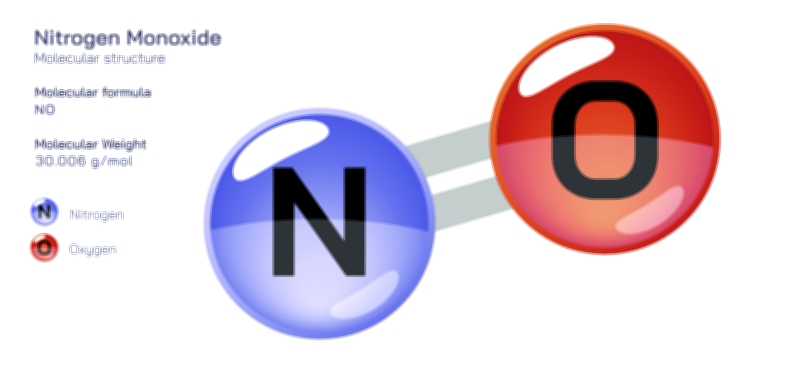
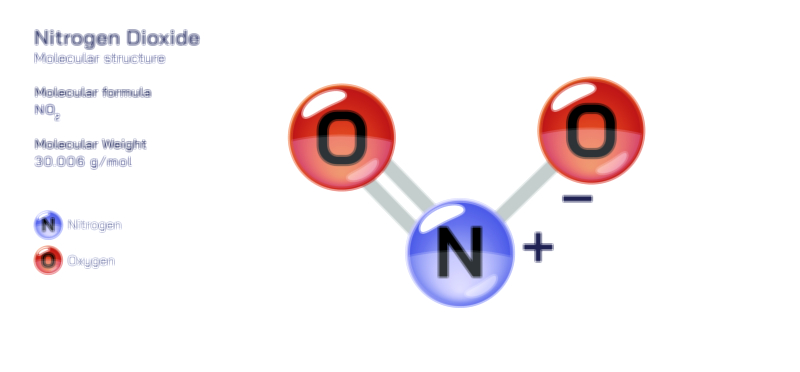
Nitrite — Structure, Properties, and Chemical Applications
Nitrite is a small yet influential inorganic ion whose structural identity and chemical behavior have made it essential in environmental chemistry, industrial processes, biological systems, food technology, and analytical science. Represented by the formula NO₂⁻, the nitrite ion consists of one nitrogen atom bonded to two oxygen atoms in a bent or angular geometry rather than a straight line. In a vector-style structural illustration, the ion typically appears with nitrogen at the center, connected to each oxygen by one double bond and one single bond — but resonance arrows are drawn to show that the true structure is a hybrid in which both N–O bonds share partial double-bond character. This resonance is the defining reason behind nitrite’s chemical reactivity: electron density is delocalized across the oxygen atoms, giving the ion the ability to act as both a nucleophile and a weak oxidizing or reducing agent depending on the system it participates in. The negative charge is distributed rather than confined to one atom, contributing to nitrite’s solubility in water and its role in ionic equilibria. Although simple in appearance, the structural arrangement of nitrogen, oxygen, and delocalized charge situates nitrite at the center of complex and highly consequential chemical pathways in nature and technology.
One of the most visible applications of nitrite is in food preservation, particularly in curing meats such as ham, bacon, sausages, and corned beef. In this context, sodium nitrite inhibits the growth of Clostridium botulinum, the bacterium responsible for botulism, making it an important preventive measure in food safety. Nitrite also reacts with myoglobin in muscle tissue to form nitrosomyoglobin, a stable pink-red pigment that gives cured meats their characteristic color. Beyond coloration, nitrite influences flavor development and reduces lipid oxidation, helping preserve texture and sensory quality during cooking, processing, and storage. At the molecular level, nitrite’s action arises from its ability to convert into nitric oxide (NO) under mildly acidic and reducing conditions. This nitric oxide binds to the iron atom in myoglobin, altering the protein structure in a way that both stabilizes meat color and protects against microbial development. Although excessive nitrite ingestion and the formation of nitrosamines in overprocessed or overheated foods have raised understandable health concerns, strict regulations and modern formulation technologies allow nitrite to remain an indispensable component of safe curing practices while minimizing unintended by-products.
In environmental chemistry and ecological cycles, nitrite serves as a key intermediate in the nitrogen cycle — the global process through which nitrogen moves between atmosphere, soil, water, and living organisms. Nitrite is produced during nitrification, when ammonia-oxidizing bacteria convert ammonia into nitrite, and during denitrification, when nitrate-reducing microorganisms convert nitrate into nitrite on the path toward nitrogen gas formation. Because nitrite is an intermediate between reduced and oxidized nitrogen species, it provides a critical checkpoint that determines whether nitrogen remains available to plants or is lost to the atmosphere. High nitrite concentrations in soil or water can signal an imbalance in microbial activity, insufficient oxygen, or pollution from agricultural runoff or wastewater discharge. In aquatic systems, elevated nitrite is toxic to fish because it interferes with oxygen transport in the bloodstream, disrupting hemoglobin function. For this reason, nitrite testing is one of the most important analytical measurements used to evaluate water quality in aquaculture, aquariums, treatment plants, and environmental monitoring programs. The structural ability of nitrite to transition chemically into nitrate, nitric oxide, or ammonia depending on redox conditions is what makes it such a sensitive marker of ecological balance.
Nitrite is also widely used in industrial and laboratory chemistry as a reagent and precursor for numerous controlled reactions. In diazotization reactions, nitrite ions react with aromatic amines in acidic solution to form diazonium salts — versatile intermediates that enable the synthesis of azo dyes, polymers, pharmaceutical compounds, and surface functionalization agents. The success of this transformation rests on the ability of nitrite to generate nitrosyl cations and nitric oxide species under the correct conditions, which is a direct consequence of the resonance stabilization and redox flexibility of the nitrite ion. In metal chemistry, nitrite forms coordination complexes with transition metals, influencing oxidation states and ligand geometry. Some complexes of iron, cobalt, and ruthenium display dramatic color changes as nitrite switches from N-bonded (nitro) to O-bonded (nitrito) attachment, a phenomenon that serves as a vivid demonstration of ligand isomerism in inorganic chemistry. In synthetic reactions, nitrite participates in selective oxidation and reduction pathways and can serve as either a mild oxidizing agent or reducing agent because of its capacity to exchange electrons depending on reaction conditions.
Nitrite also plays a role in corrosion inhibition and industrial cooling systems, where nitrite salts help protect steel and other ferrous metals from oxidative degradation. In these systems, nitrite participates in the formation of a stable oxide film on the metal surface, slowing reaction with dissolved oxygen and extending the service life of pipes, heat exchangers, and machinery. The protective effect arises from nitrite’s oxidizing ability, which nudges iron toward forming a tightly adherent oxide layer instead of flaking rust. This application—although chemically different from food preservation—again depends on nitrite’s ability to generate nitric oxide or related species under controlled conditions.
In biological systems, nitrite is both a signaling molecule and a reservoir for nitric oxide, especially in contexts where tissues experience low oxygen levels. While nitric oxide acts as a vasodilator and cellular messenger, the body cannot rely solely on enzymes that produce NO from L-arginine when oxygen availability is low. In those conditions, nitrite becomes a biochemical backup system capable of releasing nitric oxide to regulate blood flow, protect tissues from ischemic damage, and support cardiovascular function. This reversible relationship between nitrite and nitric oxide has reshaped scientific understanding of how the body controls vascular tone, metabolic efficiency, and oxygen delivery — an insight now guiding research in medical fields such as cardiovascular health, metabolic disease, and mitochondrial biology.
Analytical chemistry also relies on nitrite because its reactions with aromatic amines produce strongly colored azo dyes, enabling highly sensitive spectrophotometric detection. Nitrite assays remain essential for environmental studies, industrial waste evaluation, and food-quality inspection. In these measurements, the formation of a stable color complex serves as a quantitative signal of nitrite concentration, showing how molecular reactivity becomes a practical tool for quality control and ecological monitoring.
Nitrite’s properties — solubility, resonance stabilization, redox versatility, and ability to interconvert with nitrate and nitric oxide — allow it to adapt to an enormous range of chemical environments. Whether it is forming a pigment in a cured ham, serving as a diagnostic indicator of water quality, enabling the synthesis of pharmaceutical intermediates, regulating blood flow in low-oxygen tissues, or preventing corrosion in industrial cooling loops, its behavior arises from the same underlying structural identity. The bent geometry of NO₂⁻ and the delocalization of electrons across nitrogen and oxygen atoms give the ion its capacity to act as a nucleophile, ligand, oxidant, reductant, and reservoir of reactive nitrogen species.
A vector illustration of nitrite may show nothing more than a nitrogen atom connected to two oxygens with resonance arrows — but that picture represents an ion embedded in agriculture, medicine, metallurgy, food sciences, environmental chemistry, and biological signaling. Nitrite demonstrates clearly that even the simplest molecular structures can become linchpins of complex chemical, industrial, and ecological systems when their intrinsic properties align with the needs of the world around them.