Kelvin–Planck Statement of the Second Law of Thermodynamics — Impossibility of Complete Heat-to-Work Conversion and the Fundamental Limits of Heat Engines
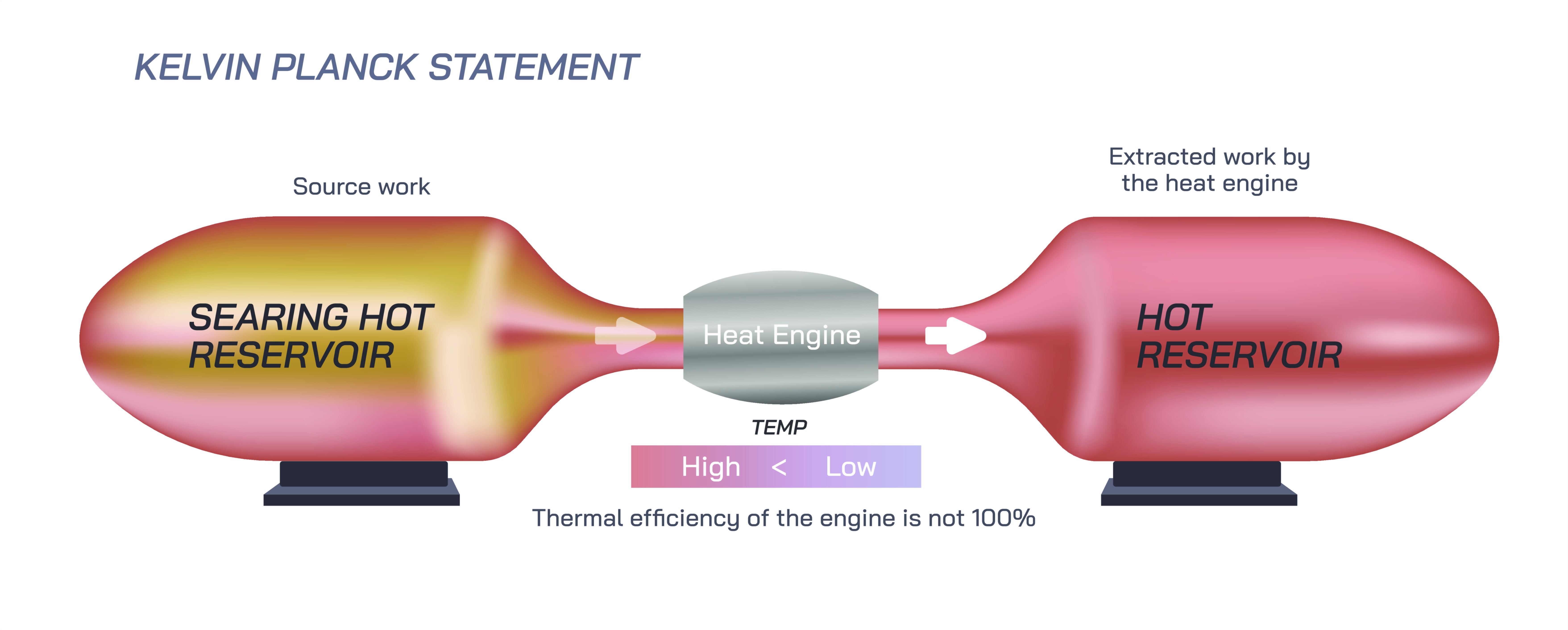
The Kelvin–Planck statement of the second law of thermodynamics is one of the most profound principles in physics because it establishes that no heat engine can ever convert all the heat it receives into useful work. In simple terms, it declares that it is impossible to build a cyclic heat engine whose only effect on the surroundings is to absorb heat from a single thermal reservoir and completely transform that heat into mechanical work. There must always be some heat rejected to a colder region, meaning that perfect efficiency is unattainable. This statement defines the limits of energy conversion and explains why real machines—from automobile engines to power plants to steam turbines—can never operate with 100% efficiency. When visualized through a vector illustration, the Kelvin–Planck statement becomes easier to understand, because the diagram shows heat entering a system from a hot reservoir, part of the energy converting into useful work, and the remaining energy expelled into a cold reservoir that is essential for completing the cycle.
The need to discharge heat into a cold reservoir is not a flaw in engineering but a fundamental property of nature. Heat flows spontaneously only from higher temperature to lower temperature, and only when that temperature difference exists can a heat engine extract usable work. Once work is produced from the available thermal gradient, the remaining energy becomes too degraded—too disordered—to perform further useful work. The cold reservoir provides a thermodynamic sink into which this degraded energy is released, allowing the cycle to continue. This inevitability means that a device drawing heat solely from a high-temperature source and producing nothing but work would violate the way energy distributes itself in the universe. The Kelvin–Planck statement therefore defines a limit on what technology can do, not because of mechanical constraints but because of the natural evolution of energy toward increased disorder and equilibrium.
When engineers design heat engines, they strive to increase efficiency by maximizing the conversion of heat to work and minimizing waste heat. However, the Kelvin–Planck principle sets a ceiling that no engine can exceed. Even the idealized Carnot engine—an abstract, perfectly reversible engine with no friction or losses—still requires both a hot reservoir and a cold reservoir. Because of this, real engines always fall short of the theoretical ideal. Piston engines in cars convert only a portion of fuel heat into motion; internal combustion engines, jet engines and steam turbines all reject significant heat through exhaust, coolant, or condenser systems. This is not due to poor engineering but because thermal processes are inherently irreversible. No matter how a machine is optimized, some fraction of heat becomes unavailable for work and must be expelled to complete the cycle.
Understanding the Kelvin–Planck statement also brings clarity to why perpetual motion machines of the second kind are impossible. Many historical inventors attempted to design devices that would operate indefinitely by continuously converting environmental heat directly into mechanical work. If such a machine were possible, it would violate the Kelvin–Planck definition by requiring no cold reservoir and allowing 100% conversion of heat to work. The statement rules out the possibility of such a device not because of mechanical friction or material limitations but because it defies the underlying direction of energy flow dictated by thermodynamic laws. This is why thermodynamics is often called a science of impossibility: instead of telling us what machines can do, it first tells us what they cannot do.
Vector illustrations of the Kelvin–Planck statement typically depict a heat engine in a closed cycle interacting with three key elements: a hot reservoir supplying energy, a working system producing work, and a cold reservoir rejecting heat. Arrows show the transfer of thermal energy from the hot reservoir to the working substance, the extraction of mechanical work, and the transfer of remaining heat to the cold reservoir. Such a graphic—often labeled "Qᵢn" for incoming heat, "W" for work output, and "Qᵒut" for rejected heat—makes the concept intuitive even for students unfamiliar with advanced thermodynamics. It visually communicates that the cold reservoir is not an optional component, but a mandatory one for cyclic operation.
The Kelvin–Planck formulation also reveals the deeper philosophical meaning of the second law: energy quality declines through natural processes. Energy is never destroyed, but high-quality, ordered energy capable of doing work gradually becomes lower-quality energy that spreads out and becomes unavailable. The cold reservoir in the heat-engine illustration symbolizes this transformation. While total energy remains constant, the energy left after work extraction becomes dispersed, contributing to entropy—the measure of disorder—and no longer retaining the potential to power mechanical motion. In this sense, the Kelvin–Planck statement is one of the clearest ways to understand entropy as an unavoidable consequence of energy conversion.
The statement applies not only to engines that burn fuel; it applies to every thermal system that tries to produce work cyclically. Solar-thermal stations, geothermal systems, nuclear power plants, gas turbines, steam engines, refrigeration cycles, and even biological systems follow its limitations. A stove or heater cannot fully transform the chemical energy of fuel into useful heat without unavoidable thermal losses. A power station cannot turn all boiler heat into electricity without releasing heat to a cooling tower or river. Even the human body must reject heat through the skin and breath while converting food energy into motion. The second law applies universally—from the largest industrial installations to living organisms and nanoscale devices.
Although the Kelvin–Planck statement is sometimes expressed in highly abstract mathematical terms, its essence is deeply practical. It sets realistic expectations for engineers and scientists, forcing them to design systems that respect the fundamental direction of heat flow. It inspires innovation not by promising unlimited efficiency but by encouraging smart energy use—reducing waste, recovering rejected heat, improving insulation, optimizing thermodynamic cycles and choosing working fluids that maximize temperature differences. The statement continues to guide modern research in renewable energy and sustainable engineering because every system intended to convert heat to work must respect thermodynamic limitations.
The profound importance of the Kelvin–Planck statement lies in how it defines the boundaries of what is physically possible. It reminds us that heat and work are not interchangeable without loss, and every attempt to convert thermal energy into mechanical energy leaves behind some unavoidable waste. In doing so, it reveals the hidden logic shaping technology, economics and even ecology: no energy transformation is perfect, systems must be designed with losses in mind, and the natural direction of energy flow determines the rules of the physical world. A vector illustration of this principle captures not only the structure of a heat engine but the fundamental truth that governs engines, energy systems and the universe itself — we can extract useful work from heat, but never all of it, and understanding that limit is what makes thermodynamics both humbling and profoundly insightful.