James Chadwick — Discovery of the Neutron and Its Revolutionary Impact on Atomic Science
James Chadwick’s discovery of the neutron in 1932 stands as one of the most transformative breakthroughs in the history of physics, fundamentally reshaping the understanding of atomic structure and paving the way for the development of nuclear science, particle physics, and modern technological innovations. Before Chadwick’s work, scientists believed that atoms consisted only of positively charged protons and negatively charged electrons, and they assumed that the mass of the nucleus could be explained solely by protons paired with electrons inside the nucleus. However, this model left unexplained inconsistencies, because the mass number of many elements did not correspond to the number of their protons. Something invisible, neutral in charge, and nearly equal in mass to the proton was missing — and its absence kept atomic theory incomplete. Chadwick’s research proved the existence of this missing particle, the neutron, and the neutron immediately became a cornerstone of nuclear science, clarifying how atoms are built, why isotopes exist, and how atomic reactions unfold.
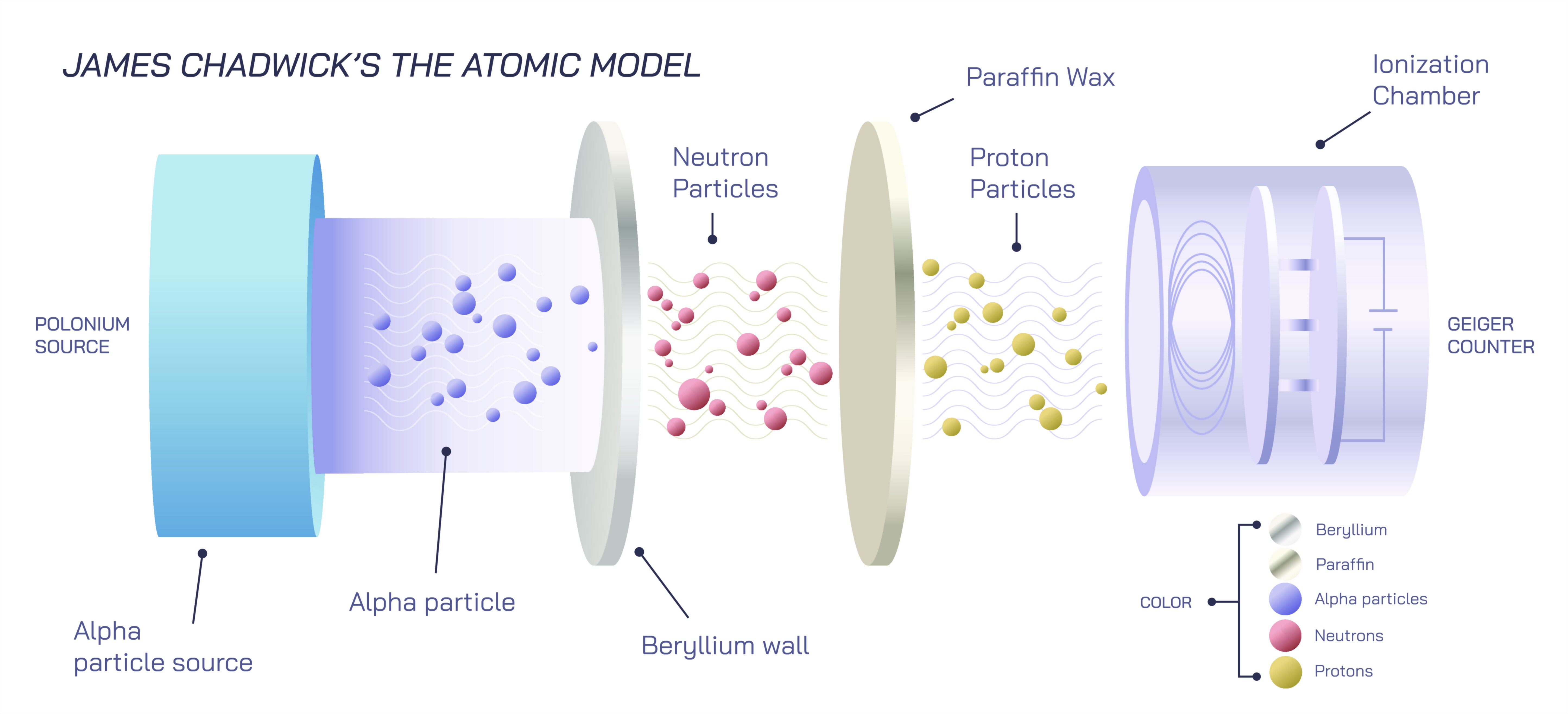
The breakthrough occurred when Chadwick analyzed experimental results coming from bombarding beryllium with alpha particles. Earlier researchers observed that the collision released a mysterious high-energy radiation capable of knocking protons out of hydrogen-containing substances, yet they misinterpreted the radiation as gamma rays. Chadwick saw contradictions in that explanation: if the radiation were gamma rays, the observed recoil energy would be far too high. He realized that the evidence pointed instead to a new type of particle with a mass similar to that of a proton but carrying no electric charge. Through a series of meticulous measurements and mathematical reasoning, he demonstrated that the particle responsible for the phenomenon must be electrically neutral and massive — a neutron. In 1932 he announced his discovery, marking the moment when the model of the atom expanded from a two-particle nucleus to a dual-particle nucleus composed of both protons and neutrons.
The neutron’s discovery solved key scientific mysteries instantly. It explained why isotopes exist: atoms of the same element contain the same number of protons but vary in the number of neutrons, resulting in different mass numbers without changing chemical identity. It also clarified why atomic nuclei do not fly apart despite containing positively charged protons that should repel each other. The presence of neutrons provides strong nuclear forces that bind the nucleus together, stabilizing the atom. Without neutrons, the existence of matter in the form we know today would be impossible. Chadwick’s contribution, therefore, was not only a discovery of a subatomic particle but an unlocking of the deeper principles of nuclear stability, binding force, and the internal architecture of atoms.
The worldwide significance of the neutron became evident very quickly. Because neutrons carry no electric charge, they can penetrate atomic nuclei easily without being repelled by positive charge. This ability made neutron bombardment one of the most powerful tools in nuclear reactions. Within only a few years of Chadwick’s discovery, scientists recognized that neutron-induced reactions could lead to controlled nuclear energy and, tragically, also to destructive nuclear weapons. Neutron-based fission became the foundation of nuclear reactors used for electricity generation and the core driver behind the atomic bomb during World War II. Although Chadwick did not discover the neutron with destructive intent, the implications of his discovery shaped world history and global power dynamics. His scientific achievement held extraordinary potential for both creation and destruction, reminding humanity that the advancement of knowledge always carries responsibility.
Beyond military and energy implications, neutron science opened new chapters in medical imaging, radiotherapy, crystallography, material science, and particle physics. Neutron scattering and diffraction techniques give researchers the ability to study molecular and crystal structures in ways that X-rays cannot achieve, especially for soft matter and magnetic materials. In medicine, neutron beams play an important role in cancer treatment approaches such as boron-neutron capture therapy. In industry, controlled neutron sources are used to inspect welds, examine turbine blades, and test aircraft components without damaging them. Every one of these advancements is connected to Chadwick’s original discovery and represents the expanding ripple effect of identifying a single neutral particle that resides inside every atomic nucleus.
Chadwick himself received the Nobel Prize in Physics in 1935, only three years after announcing the neutron’s existence — one of the fastest recognitions in Nobel history. His discovery completed the basic picture of the atomic nucleus and opened the door to quantum nuclear physics, neutron science, and the global transformation of technology in the twentieth century. Educational illustrations of Chadwick’s discovery typically show alpha particles striking beryllium, the emission of neutral radiation, and the collision of the newly discovered neutron with hydrogen nuclei to prove its mass. In such vector diagrams, highlighting the lack of electric charge visually distinguishes the neutron from the proton and electron, helping learners understand why it behaves differently in nuclear reactions.
More importantly, these visual representations help students recognize that the discovery of the neutron was not an isolated event but a turning point that clarified atomic structure, enabled nuclear science, and reshaped industries, medicine, and geopolitics. Scientific progress is built layer upon layer, and the neutron became one of the foundational building blocks that anchored modern physics. Chadwick’s insight emphasized the importance of questioning assumptions and re-examining experimental results rather than accepting accepted explanations. It demonstrated that scientific breakthroughs often come not from observing something new but from interpreting familiar results with a deeper understanding.
The story of James Chadwick and the neutron stands today as an enduring lesson: the building blocks of matter can change the building blocks of civilization. Matter, energy, technology, and society are profoundly connected, and uncovering the structure of the smallest components of the universe can reshape the largest systems of human life. Chadwick’s discovery continues to be taught not only as a historical milestone in physics but as a symbol of how curiosity, persistence, and scientific reasoning have the power to expand the limits of human knowledge and change the world.