Faraday’s First Law of Electrolysis — Electrochemical Reaction and Mass Deposition Explained
Faraday’s First Law of Electrolysis stands as one of the most foundational principles in electrochemistry, forming a bridge between electrical energy and chemical change. Although often represented in vector illustrations as wires, electrodes, ions, and directional arrows showing the flow of charge and deposition of matter, the deeper understanding of this law emerges through a rich, descriptive exploration of how electricity interacts with chemical substances to transform them at the molecular level. Faraday’s First Law does not simply state a relationship between electric charge and deposited mass; it reveals the intimate connection between electrons and ions, showing how movement of charge across an electrolyte produces tangible, measurable changes in material form. Examining this law without formulas allows its meaning to emerge through concepts, processes, and physical behavior, offering a complete narrative of how electrolysis operates and why certain quantities of material appear at electrodes during electrochemical reactions.
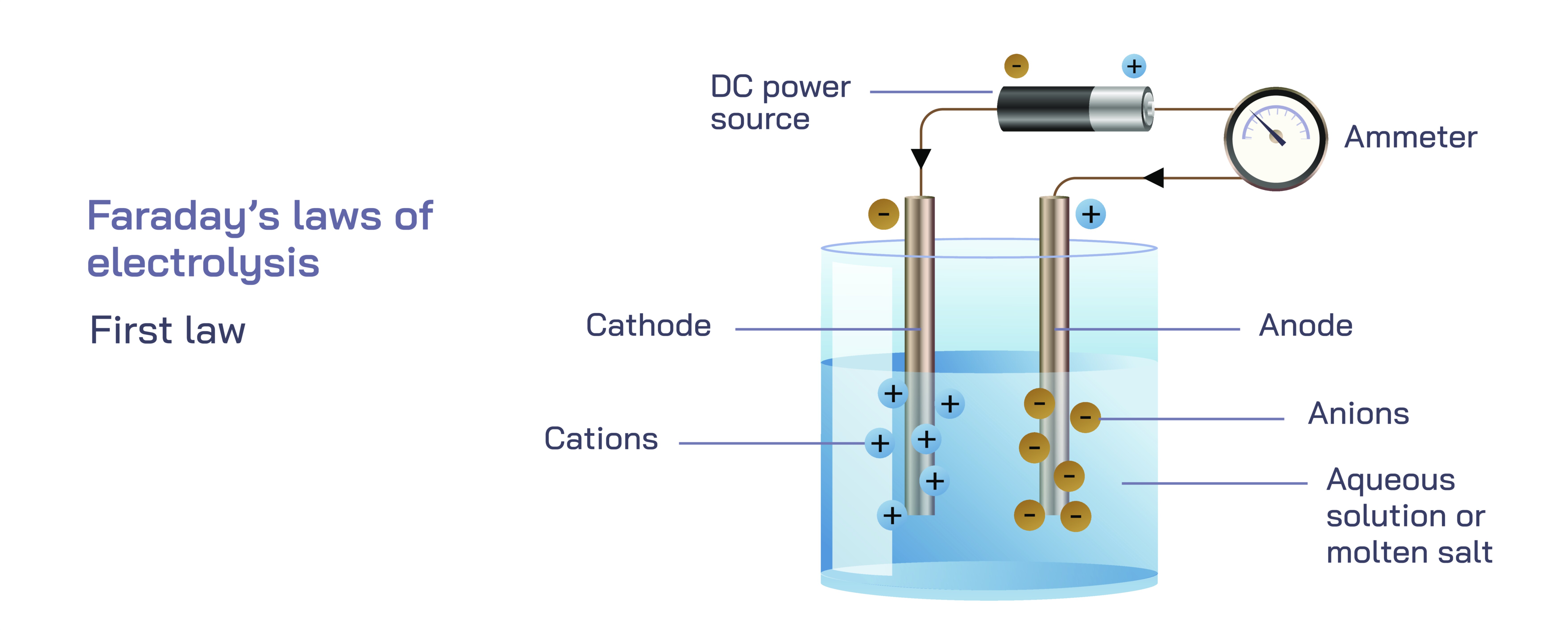
At the heart of electrolysis is the movement of electric current through an electrolyte solution or molten ionic compound. This current is not just a flow of electrons in a wire; it involves ions drifting through the liquid medium under the influence of an applied electric field. When a voltage is applied across two electrodes immersed in an electrolyte, charged species begin moving: positively charged ions migrate toward the negative electrode, while negatively charged ions travel toward the positive electrode. This migration establishes a dynamic process in which chemical species gain or lose electrons at the surface of the electrodes. The negative electrode, called the cathode, supplies electrons to positive ions, causing them to be reduced. This reduction often leads to deposition of a metal or release of a substance. The positive electrode, called the anode, removes electrons from negative ions or stimulates the oxidation of neutral molecules, releasing different species into the environment. Through these transformations, electrolysis becomes a controlled method of driving chemical reactions that would not occur spontaneously.
Faraday’s First Law states that the mass of a substance deposited or liberated at an electrode during electrolysis is directly proportional to the amount of electric charge that passes through the electrolyte. In conceptual terms, this means that the more electricity delivered into the system, the more material accumulates at the electrodes. Every electron that flows through the external circuit corresponds to a specific number of ions being transformed at the electrode surfaces. Because ions undergo discrete changes—gaining or losing exact quantities of electrons—the amount of matter produced is not arbitrary but follows a strict proportional relationship with the total charge transferred.
Understanding this law requires appreciating the precise, stepwise nature of electron exchange at the atomic level. When an ion approaches an electrode, it interacts with electrons on the surface in a quantized manner. A copper ion, for example, needs a specific number of electrons to convert into metallic copper, and only when those electrons are supplied does the deposition occur. Similarly, other ions require their exact electron count to undergo transformation. Thus, the quantity of substance formed is fundamentally governed by how many electrons arrive or depart, which is controlled by the electric current supplied. Current becomes a measure of electron flow, and electron flow directly dictates how much matter can be chemically altered.
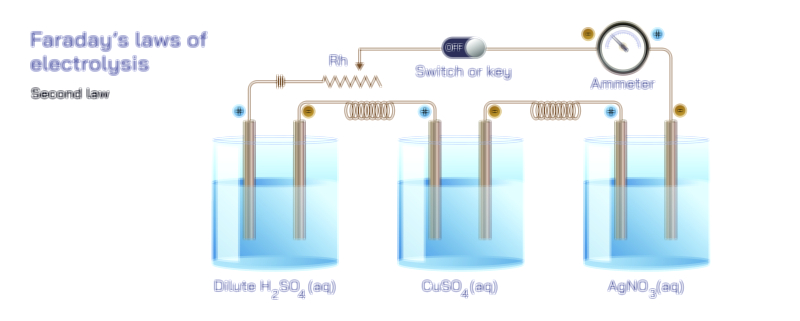
During electrolysis, the chemical changes occurring at the electrodes are not random; they depend entirely on the charge requirements of the ions. Some ions need only one electron to be reduced, while others require two or more. These variations mean that the mass of material deposited is tied to the electrical charge through the intrinsic characteristics of the substance itself. Faraday’s First Law emphasizes that the mass of material deposited is not solely about current flowing; it is equally about the identity of the substance being deposited. Different materials respond to electric charge differently because their ions carry distinct charges and undergo unique transformations. The law therefore explains not only that matter is deposited in proportion to electricity, but also that different substances will appear in different amounts under identical electrical conditions.
To visualize this concept, imagine an electrolytic cell where copper ions in solution move toward the cathode. As current flows, electrons accumulate on the cathode surface. Each incoming copper ion arrives with a positive charge and accepts a precise number of electrons, transforming from an ion in solution into solid copper metal that adheres to the electrode. If more current is applied, more electrons become available, and more copper ions undergo reduction. The longer the current is applied, the more material accumulates. Faraday’s First Law reveals that this process is tightly controlled: supplying twice as much electric charge results in exactly twice as much copper being deposited. The predictability of this behavior is what makes electrolysis a powerful tool in industrial processes such as electroplating, metal refining, and chemical synthesis.
This direct proportionality between deposited mass and electric charge allows electrolysis to be used as a measuring tool. Before the advent of sensitive analytical instruments, scientists relied on electrolysis to determine atomic weights, charge ratios, and chemical identities. The method was straightforward: pass a known amount of electric charge through an electrolyte and measure how much material was deposited. From this observation, they could deduce information about the properties of ions. Faraday’s pioneering work revealed how fundamental this relationship is, showing that it arises from the discrete nature of electric charge and the consistent behavior of ions under electrochemical conditions.
Exploring Faraday’s First Law further requires understanding what “electric charge” means in the context of electrolysis. Electricity in a circuit moves as electrons through wires, but when it enters an electrolyte, it takes on a different form. Inside the electrolyte, the charge carriers are ions—atoms or molecules carrying positive or negative charge. At the electrodes, electrons from the wire convert into ionic charge or vice versa. Thus, charge moves through different mediums in different ways while maintaining continuity throughout the system. Faraday’s insight was to bridge the movement of electrons in the external circuit with the movement of ions in the electrolyte, showing that they are connected by precise and predictable conversion rules. Each electron traveling through the wire corresponds to one electron gained or lost by an ion at the electrode, linking mass deposited with the flow of current.
When examining the physical nature of deposition, one sees that materials accumulate atom by atom, forming layers or clusters depending on the metal and the conditions. This deposition process is influenced by factors such as electrode shape, ion concentration, temperature, current density, and the presence of impurities. However, regardless of these variations, the total mass of deposited material still follows the proportional rule set forth by Faraday’s First Law. Even in systems where deposition occurs unevenly—forming crystalline patterns, branching structures, or rough metallic textures—the total mass is still controlled by the amount of charge passing through the cell. This consistency makes electrolysis an extremely reliable process for coating metals, purifying materials, and synthesizing compounds.
The law also applies to electrolysis of nonmetallic substances. For example, when water undergoes electrolysis, hydrogen bubbles form at the cathode and oxygen bubbles appear at the anode. Here too, the amount of gas produced is directly proportional to the electric charge delivered. Although gas evolution looks different from metal deposition, the underlying principle remains the same: the ions being transformed require specific numbers of electrons, and the supply of those electrons dictates how much product forms. In industrial processes such as chlorine production, aluminum extraction, and production of hydrogen gas, Faraday’s First Law allows precise prediction of how much product will be obtained when a given electric current is applied for a specific duration.
Faraday’s First Law also brings attention to the idea of efficiency in electrochemical systems. In ideal cases, all the current contributes to the desired chemical reaction, meaning the deposition or liberation of matter follows the theoretical relationship perfectly. In practical systems, however, side reactions can occur. Unintended reactions may consume some portion of the current, reducing the efficiency of deposition. For example, in certain solutions, unwanted reactions such as hydrogen evolution may occur at the cathode. Even in these cases, the fundamental proportionality remains valid: the mass of desired product depends on the effective charge that actually participates in the primary reaction. Understanding efficiency becomes an essential part of applying Faraday’s First Law to real systems, especially in industrial electrolysis where optimizing performance and reducing waste are crucial.
The significance of Faraday’s First Law extends far beyond laboratory electrochemical cells. It forms the foundation of technologies such as battery science, electrodeposition manufacturing, metal purification, corrosion protection, and electrochemical sensing. In rechargeable batteries, ions move between electrodes during charging and discharging, following principles that mirror Faraday’s foundational insights. In electroplating, the precise control of deposition allows manufacturers to coat materials with layers of metal for protection, conductivity, or decoration. In metallurgy, electrolysis enables extraction of pure metals from ores. All these technologies depend in some way on the predictable relationship between electric charge and chemical transformation.
Faraday’s First Law also underscores a deeper philosophical idea: that nature operates with consistent rules linking electricity and matter. Before Faraday’s discoveries, the connection between electrical phenomena and chemical processes was mysterious. His work provided clarity, showing that electric currents do not merely induce or accompany chemical reactions—they quantitatively govern them. Electricity, therefore, is not just a force that can spark reactions; it is a measure of how much material will change. This profound link between electrons and physical matter reflects a unity in natural processes that spans physics and chemistry.
In biological systems, electrochemical processes governed by principles akin to Faraday’s Law play essential roles. Neurons transmit signals through ion movements, and metabolic pathways involve electron transfer reactions that sustain life. Although biological electrochemistry is far more complex than simple electrolytic cells, the same underlying rule applies: the transfer of electrons correlates with specific chemical transformations. Faraday’s conceptual insight helps illuminate why electron-transfer reactions form the backbone of countless physiological phenomena.
When represented through vector illustrations, Faraday’s First Law often appears as a schematic showing ions moving toward electrodes, electrons flowing through external circuits, and deposited matter accumulating. These diagrams capture the visual essence of electrolysis, but the law itself reaches deeper, describing a quantitative truth about the relationship between energy and matter. It reveals how electrical energy translates directly into physical change, how ions use electrons to transform, and how chemical substances respond predictably to current.
Ultimately, Faraday’s First Law of Electrolysis transforms our understanding of chemical change by tying it directly to electrical charge. It provides a conceptual framework for predicting how much matter will be deposited or liberated during electrolysis, illustrating how electrons and ions exchange matter and energy in precise proportions. Through richly detailed understanding of this relationship, electrolysis becomes not merely a laboratory technique but a window into the fundamental interplay between electricity and the material world.