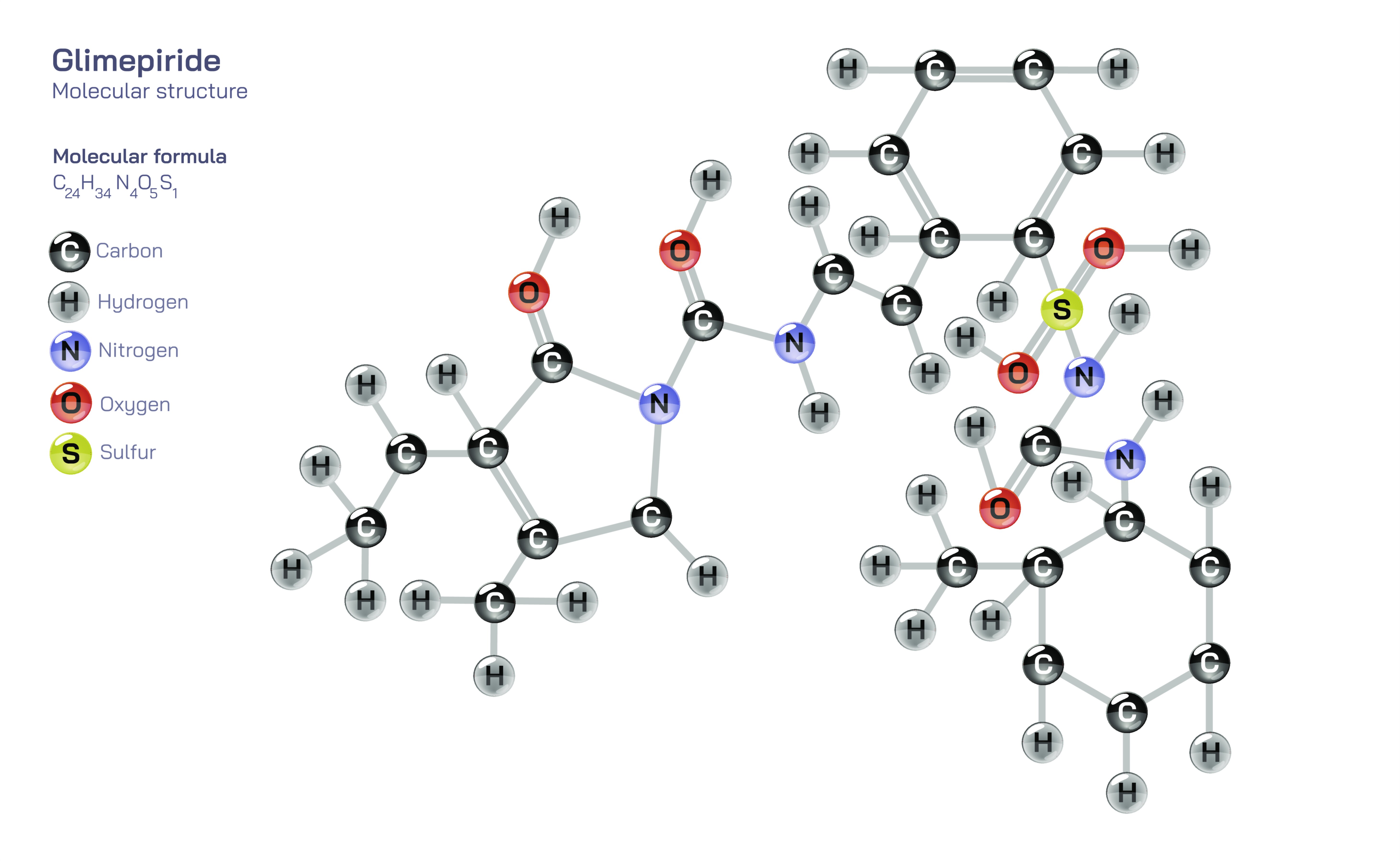
Glimepiride — Chemical Structure, Antidiabetic Action, and Molecular Composition Explained
Glimepiride is a widely prescribed oral antidiabetic medication belonging to the sulfonylurea class, and although medical references often portray it using structural diagrams showing aromatic rings, sulfur-containing groups, and functional substituents, its full significance becomes clear only through a deep conceptual explanation that connects its molecular architecture to its biological mechanism and therapeutic purpose. Glimepiride is used primarily in the treatment of type 2 diabetes mellitus, a condition characterized by insulin resistance and impaired insulin secretion, and it works not by replacing insulin directly but by stimulating the body’s own ability to release insulin from pancreatic β-cells. To understand how glimepiride functions, one must explore its chemical composition, its structural features, and the way these features enable it to interact with specific cellular receptors that control insulin release.
At the molecular level, glimepiride is composed of several linked structural groups that work together to give the compound its characteristic behavior. A large portion of the molecule consists of a stable aromatic ring system, which provides rigidity and allows the molecule to maintain a defined three-dimensional orientation as it interacts with biological targets. Attached to this ring system is a sulfonylurea backbone, a structural motif shared by all drugs of this family and responsible for their unique glucose-lowering effects. This section of the molecule contains sulfur, nitrogen, carbon, oxygen, and hydrogen atoms arranged in such a way that the molecule can bind selectively to receptors on pancreatic β-cells. Additional chemical substituents—including a cyclohexyl group and a heterocyclic portion—enhance the molecule’s lipophilicity and enable it to cross cell membranes efficiently. Each part of the molecule contributes to its potency, duration of action, and ability to reach its intended site within the pancreas.
When glimepiride enters the bloodstream after oral administration, it travels to the pancreas, where it specifically targets β-cells located in the islets of Langerhans. On the surface of these β-cells are ATP-sensitive potassium channels, which act as regulators of insulin secretion. In the absence of stimulation, these potassium channels allow potassium ions to move out of the cell, maintaining an electrical balance that keeps insulin granules stored inside. Glimepiride binds to a regulatory subunit of these potassium channels and causes them to close. The closure of potassium channels alters the electrical charge across the β-cell membrane, triggering the opening of nearby calcium channels. As calcium ions rush into the cell, they initiate a cascade of events that ultimately results in the fusion of insulin-containing vesicles with the cell membrane and the release of insulin into the bloodstream.
This process creates a direct connection between glimepiride’s chemical structure and its physiological effect. The drug is not itself a source of insulin but rather a molecular key that unlocks the β-cell’s stored insulin supply by altering ion flow. Because of this mechanism, glimepiride is effective only when β-cells are still capable of producing insulin, which is why it is prescribed for type 2 diabetes rather than type 1 diabetes, where insulin production is absent. The drug amplifies the body’s natural response to glucose by enhancing insulin secretion at times when blood sugar levels rise. This controlled stimulation helps maintain stable glucose levels throughout the day, particularly after meals.
The molecular composition of glimepiride also influences its pharmacokinetics—that is, how the body absorbs, distributes, metabolizes, and eliminates the drug. Its lipophilic characteristics help it bind to plasma proteins and penetrate cell membranes, giving it a prolonged duration of action. Even after the drug concentration decreases in the bloodstream, its tight binding to the β-cell receptor maintains a therapeutic effect. The liver metabolizes glimepiride into active and inactive compounds, and these are eventually excreted through the kidneys. This balance of absorption, distribution, and metabolism contributes to its once-daily dosing schedule, making it convenient for long-term treatment.
Although glimepiride stimulates insulin release, its effects extend beyond the pancreas. Research suggests that it can also enhance glucose uptake in muscle tissue and improve insulin sensitivity through secondary pathways. These additional metabolic actions complement its primary effect and help improve overall glucose control. Unlike some earlier sulfonylurea drugs, glimepiride has been engineered to be potent even at low doses and to maintain its activity throughout the day without causing excessive stimulation that could lead to rapid drops in blood sugar. Its structural refinements compared with earlier generation sulfonylureas illustrate how small changes in molecular composition can significantly affect clinical outcomes.
Because glimepiride works by increasing insulin secretion, one of its most important clinical considerations is the balance between glucose lowering and the risk of hypoglycemia. If too much insulin is released relative to blood glucose levels, the individual may experience symptoms of low blood sugar. The drug’s dosage therefore must be carefully tailored to each patient’s lifestyle, diet, physical activity, and individual insulin dynamics. The fundamental mechanism—closing potassium channels and opening calcium channels—remains the same regardless of dose, but the degree of stimulation varies with the amount of drug present. This precision underscores how a controlled molecular interaction shapes a physical health outcome.
The therapeutic value of glimepiride becomes more evident when viewed against the broader context of metabolic regulation. In type 2 diabetes, high blood sugar develops not only because tissues do not respond efficiently to insulin but also because insulin secretion becomes delayed or insufficient after meals. Glimepiride helps restore this timing and adequacy of insulin release, enabling glucose to be taken up by cells when needed and preventing chronic hyperglycemia that can lead to long-term complications such as neuropathy, kidney disease, vision loss, and cardiovascular problems. The drug therefore acts at a pivotal point in glucose homeostasis, reinforcing the β-cell’s role as a regulator of metabolic balance.
Beyond its molecular mechanism and medical function, glimepiride illustrates how pharmaceutical chemistry links structure to function. Changing one ring or substituent in the compound could change its binding affinity, its ability to pass through membranes, its metabolic stability, or its duration of action. The sulfonylurea backbone is the essential core for receptor binding, but the surrounding chemical groups fine-tune potency and pharmacological behavior. Drug design in this class demonstrates a wider principle in medicinal chemistry: small structural adjustments can lead to major therapeutic improvements, longer duration, fewer side effects, or improved safety.
The story of glimepiride highlights the interconnectedness of chemistry, biology, and medicine. Its molecular architecture determines how it interacts with β-cell receptors; that interaction determines how insulin is released; insulin then determines how cells receive energy from glucose; and glucose control determines long-term metabolic stability. Through this chain of cause and effect, a microscopic arrangement of atoms influences whole-body health. Taking this conceptual view offers a deeper appreciation for how a single molecule can help people with diabetes maintain functional metabolic regulation and reduce health risks.
Thus, glimepiride is not merely a chemical formula or a medicinal label—it is a molecular tool engineered to interact with the body’s own insulin-producing cells in a precise and targeted way. Its structure creates its biological identity, its receptor binding triggers its therapeutic action, and its effects ripple outward through the body’s metabolic pathways to support glucose homeostasis. Understanding glimepiride through a detailed conceptual lens shows how chemistry becomes biology and how targeted molecular interactions become life-changing medical treatments.