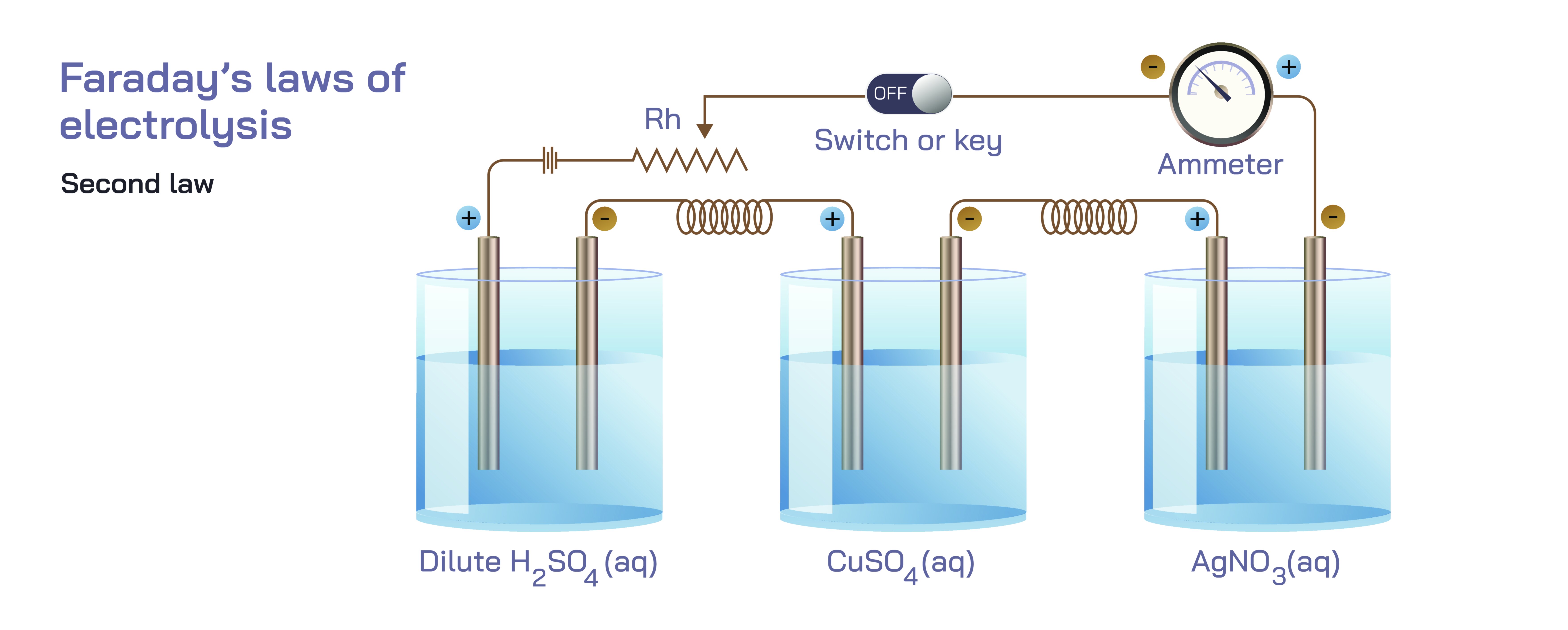
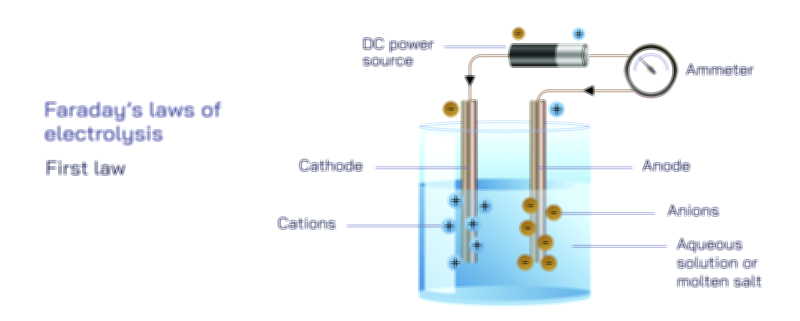
Faraday’s Second Law of Electrolysis — Proportional Mass Deposition in Electrochemical Reactions
Faraday’s Second Law of Electrolysis provides one of the most profound conceptual insights into how electrical energy translates into chemical change, revealing that electrolysis is not merely an electrical process but a quantifiable, predictable transformation governed by the intrinsic nature of chemical substances. While Faraday’s First Law explains that the mass of a substance deposited or liberated at an electrode is directly proportional to the quantity of electric charge that passes through an electrolyte, the Second Law takes this understanding a step deeper by comparing different substances undergoing electrochemical change under identical electrical conditions. In essence, the Second Law states that if the same amount of electric charge passes through multiple electrolytic cells arranged in series, the masses of different substances deposited at their respective electrodes are proportional to their chemical equivalent weights. This principle, though often represented in simplified vector illustrations showing ions moving toward electrodes and different masses forming under equal charge flow, becomes far more meaningful when explored through a detailed conceptual narrative. It reveals why different materials respond differently to the same amount of electrical input, how intrinsic atomic properties influence electrochemical behavior, and how these ideas reshape our understanding of electrochemical processes in chemistry, industry, and natural systems.
To understand Faraday’s Second Law, begin with the realization that not all ions require the same number of electrons to undergo reduction or oxidation. Each ion has a specific charge, meaning it needs a defined number of electrons to transition from its ionic state to a neutral or deposited state. In electrolysis, ions move through a solution toward electrodes where they either gain or lose electrons, but the number of electrons they require is determined by their valence or oxidation state. Some ions need only one electron to be reduced, while others need two, three, or more. This intrinsic requirement creates differences in how materials respond to identical electrical conditions. When a fixed quantity of electric charge passes through an electrolytic system, only a certain number of electrons are available. Because each ion has a fixed electron demand, the number of ions that can undergo transformation is limited by how much charge each of them requires. Therefore, under identical electrical input, the amount of material deposited depends not merely on the number of ions but on the electron requirement per ion, which is intimately tied to the substance’s chemical equivalent weight.
Chemical equivalent weight is a concept that reflects how much of a substance participates in electrochemical reactions per unit charge. It is derived from the molar mass of the substance divided by the valence of the ion participating in the reaction. This means substances with higher valences require more electrons per atom or ion, and therefore fewer atoms will be deposited per unit charge compared with substances of lower valence. Conversely, substances with lower valence charges require fewer electrons per atom, allowing more atoms to be deposited for the same electrical input. Faraday’s Second Law captures this relationship with remarkable clarity, showing that the mass of each substance deposited at an electrode is proportional to its chemical equivalent, meaning that the masses of different materials deposited under the same charge ratio reflect their inherent chemical characteristics rather than external experimental factors.
Consider the example of depositing silver and copper using the same electric current passing through two separate electrolytic cells. A silver ion typically requires one electron to become metallic silver, whereas a copper ion may require two electrons to reduce from Cu²⁺ to Cu. If one electron corresponds to the deposition of one atom of silver, then two electrons correspond to the deposition of one atom of copper. This means that under the same electric charge, fewer copper atoms will be deposited than silver atoms because copper requires more electron input per atom. However, when comparing mass rather than number of atoms, additional differences arise due to atomic weights: silver atoms are heavier than copper atoms. Faraday’s Second Law reveals that the ratio of deposited masses, once adjusted for charge requirements, reflects a deeper proportionality tied to atomic and ionic characteristics. Thus, equal charges produce different masses not by accident but due to the chemical structure and charge requirements of each substance involved.
Faraday’s Second Law also clarifies why electrochemical reactions are fundamentally quantized processes. Ions do not partially undergo electron transfer; they must receive or lose exact whole numbers of electrons. This quantized nature of electron exchange creates strict proportionality between electric charge and chemical deposition. When comparing different materials, the quantized nature of their electron requirements shapes the ratio in which they transform under electrolysis. This proportionality emphasizes the profound connection between atomic structure and electrical behavior, linking electrochemistry directly to the internal logic of atoms and ions. Through this lens, electrolysis becomes a window into the atom’s inner architecture, showing how valence and atomic mass govern measurable, macroscopic changes in deposited material.
This principle also has significant implications for electrochemical process design. In industrial contexts where multiple electrolysis reactions may occur simultaneously—such as electrorefining of metals, the production of chemical intermediates, or the operation of electrochemical sensors—understanding the proportionality of mass deposition becomes essential for predicting product yields, controlling reaction conditions, and optimizing efficiency. For example, if a set amount of electricity is passed through different solutions to extract metals, Faraday’s Second Law ensures that the relative mass of each metal obtained is predictable as long as the equivalent weights are known. This allows industries to calibrate processes precisely, avoid waste, and maintain consistent quality in metal plating, electrorefining, or electrochemical synthesis.
The law also played a key role historically in helping scientists determine atomic weights and chemical equivalents before advanced analytical instruments existed. By comparing the masses of different metals deposited by the same electric charge, early chemists could calculate relative atomic weights based on the proportionality described by Faraday. These early experimental results contributed significantly to the development of atomic theory, strengthening the idea that atoms and ions behave in consistent, quantifiable ways. Faraday’s electrolytic laws provided experimental support for the view that matter is composed of discrete units that respond predictably to electric charge, helping to shape modern chemistry’s understanding of atomic and molecular structure.
Faraday’s Second Law also sheds light on electrochemical processes that involve more complex ions. Many electrochemical reactions do not involve simple metal ions but polyatomic ions or molecules with multiple possible oxidation states. Even in these cases, the law holds: the number of electrons required for transformation determines the chemical equivalent, and the mass deposited or liberated reflects that requirement. This applies to electrolysis of salts, oxides, hydroxides, and even certain organic compounds. The consistent application of the law across such diverse systems demonstrates its deep universality. It reveals that electrochemical reactions, while varied in their external appearance, follow the same internal logic dictated by electron transfer and ionic charge.
Another important aspect of Faraday’s Second Law is its connection to system efficiency. In practical systems, not all electric charge may contribute to the intended deposition reaction. Side reactions may occur, such as hydrogen evolution, oxygen formation, or competing ion reduction. Even when these side reactions occur, the Second Law still provides a conceptual framework for understanding the effective charge that contributes to deposition. Engineers and chemists use this understanding to calculate faradaic efficiency—the fraction of total charge that actually produces the desired chemical change. This concept is crucial in advanced technologies such as battery systems, electrochemical water splitting, fuel cell operation, and corrosion control. Faraday’s insights guide the design of electrodes, selection of electrolytes, and control of reaction environments to maximize the proportion of charge that participates in the main reaction.
In biological systems, although electrolysis does not occur in the same way as in industrial cells, the underlying principle of electron-driven proportionality still applies. Many biochemical processes rely on electron transfer chains, where specific molecules undergo reduction or oxidation in a strictly proportional manner. These reactions obey the same deep principles that Faraday described: electron transfer defines the stoichiometry of chemical change. In cellular respiration, for example, electron carriers receive and donate electrons in fixed quantities, leading to consistent formation of chemical products. While these processes occur in complex environments, they reflect the same quantized nature of electron exchange that Faraday’s laws describe at a fundamental level.
When represented visually, vector illustrations often depict Faraday’s Second Law with multiple electrolytic cells connected in series, showing ions of different charge states depositing at separate electrodes. These diagrams emphasize that the same electric charge flows through each cell, but the masses deposited differ because the ions have different electron requirements. Although these illustrations help build intuitive understanding, the deeper narrative reveals why the law holds true universally. It is not the wiring arrangement or the diagrammatic representation that creates proportionality—it is the intrinsic atomic characteristics that govern how each ion interacts with electrons.
Faraday’s Second Law also invites a broader reflection on how electrical energy interacts with matter. Electricity is not merely a force that pushes ions around; it is a fundamental part of how matter transforms at the most basic level. When electric charge moves through an electrolyte, it becomes the driving force for rearranging electrons around atoms, shifting ions into new states and generating new materials. The proportionality described by Faraday’s Second Law reflects the harmony between physical charge flow and chemical identity, showing that the transformation of matter is not chaotic but deeply structured and predictable. Through this law, electricity becomes a tool for understanding the nature of substances themselves.
Ultimately, Faraday’s Second Law of Electrolysis unveils the elegant proportionality that connects electric charge to chemical identity. It explains why equal charges produce unequal masses, how ion valence controls deposition, and why chemical equivalent weight governs electrochemical transformation. Through richly detailed understanding of this law, electrolysis becomes not merely a matter of passing current through a solution but a profound exploration of the relationship between electrons, ions, mass, and matter itself.