Fick’s Laws of Diffusion — Molecular Transport and the Diffusion Process Explained
Diffusion is one of the most fundamental and widespread natural processes, responsible for the movement of molecules in gases, liquids, and even solids. It shapes everything from the way scents spread through air to how nutrients move inside cells, how ink disperses in water, how lungs absorb oxygen, and how materials exchange particles across membranes. Fick’s Laws of Diffusion, though often shown in vector illustrations as arrows representing molecular motion, concentration gradients, and directional flux, express a deep conceptual understanding of how and why molecules spread from regions of higher concentration to regions of lower concentration. These laws describe not only the rate at which diffusion occurs but also the mechanisms behind it, offering profound insight into chemical processes, biological systems, environmental behavior, and engineering applications. When explored through a detailed conceptual narrative—without formulas or mathematical expressions—the meaning of Fick’s Laws becomes both clear and deeply connected to real-world phenomena.
At its essence, diffusion arises from the random motion of particles. Molecules in any fluid system, whether gas or liquid, are in constant thermal motion. They collide, bounce, drift, shuffle, and move in unpredictable ways due to their internal kinetic energy. This random motion causes particles to spread out over time, gradually occupying available space in a more uniform manner. Fick’s Laws provide a structured conceptual explanation for this otherwise chaotic movement by showing how concentration differences drive directional behavior in a statistical sense. While individual molecules move randomly, the overall movement of a population of molecules has a definite direction if a concentration gradient exists. This direction is always toward lower concentration, reflecting nature’s tendency toward uniformity and equilibrium.
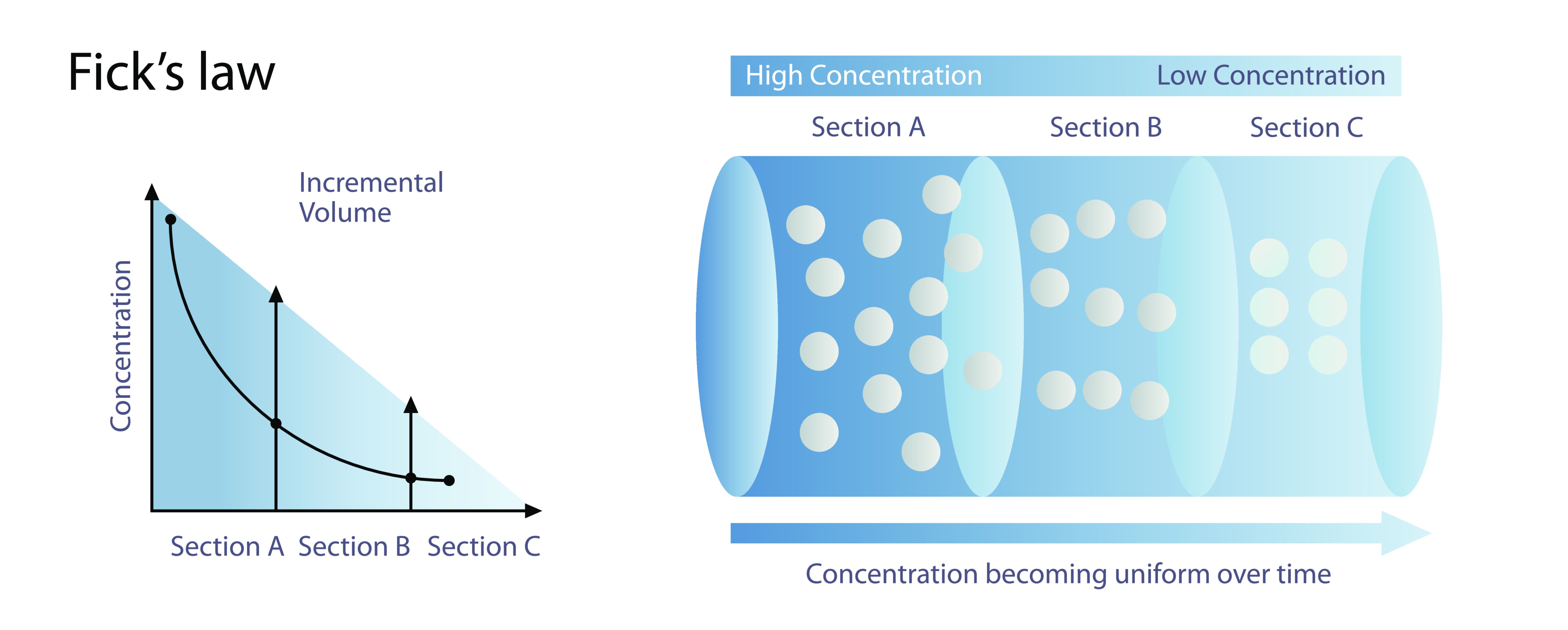
Fick’s First Law of Diffusion captures the idea that diffusion occurs because molecules naturally flow from areas of high concentration to areas of low concentration. This law describes a steady-state situation—one in which concentrations do not change over time at each location, even though diffusion continues to occur. The law states conceptually that the rate at which molecules move through a surface (commonly called the flux) is proportional to the steepness of the concentration gradient. A steep gradient means that concentration changes rapidly over a short distance; a shallow gradient means that concentration changes slowly across distance. Fick recognized that when the gradient is steeper, molecules have a stronger statistical “push” toward the region of lower concentration, leading to greater motion in that direction. This does not imply any conscious behavior; molecules do not seek equilibrium. Instead, it is the natural result of random motion: where more molecules are present, their random motion creates a net drift toward regions with fewer molecules simply because collisions and movement are more frequent in the densely populated region.
This principle shows up in countless everyday experiences. Consider a drop of perfume released in one corner of a room. The perfume molecules, initially concentrated in a small region, rapidly disperse outward due to random molecular motion. Because molecules are far more crowded near the perfume source, their random motion statistically favors movement outward, leading to a net flow in that direction. Over time, the entire room becomes permeated with the scent because diffusion continues until the concentration becomes uniform. The same basic principle governs the spread of ink in water, the removal of waste gases from the lungs, and even the exchange of substances through the microscopic pores of cell membranes. Fick’s First Law provides the conceptual foundation for explaining these processes, emphasizing that diffusion rate depends on concentration differences and the intrinsic ability of molecules to move through a medium.
Fick’s Second Law of Diffusion goes deeper by describing how concentrations change over time during non-steady-state situations. While the first law deals with cases where concentrations remain constant at each point, the second law explains what happens when concentrations evolve as diffusion proceeds. In real-world systems, diffusion rarely begins in a steady state. When a new substance enters a region—such as dye introduced to water, or an odor released into air—concentrations shift continuously until equilibrium is achieved. Fick’s Second Law describes this dynamic evolution. Conceptually, it states that the rate of concentration change at any location depends on the curvature or shape of the concentration gradient around that point. If concentrations vary unevenly from one point to another, diffusion acts to smooth out these differences, reducing steep gradients and spreading the substance more uniformly over time.
This dynamic behavior can be imagined by looking at how heat spreads across a metal rod or how salt disperses in water. When a spoonful of salt is added to one side of a container, the concentration there is high, while other regions are salt-free. As diffusion begins, salt molecules move outward from the concentrated region, initially creating a steep concentration gradient. But as time passes, the gradient becomes smoother because salt spreads farther. The second law describes this smoothing process, showing that the strongest diffusion occurs at the steepest gradients, and as gradients weaken, diffusion slows. This explains why the initial spread of a substance can be relatively fast but gradually slows down as the system moves closer to uniformity.
Together, Fick’s two laws provide a complete conceptual framework for understanding molecular transport. The first law explains what happens when diffusion reaches a steady state; the second law explains how systems evolve toward such steady states. These laws reveal that diffusion is not driven by external forces but by internal, natural tendencies arising from random molecular motion and statistical distributions. They provide insight into how molecules distribute themselves, how substances mix, and how gradients diminish over time. These principles lie at the heart of numerous scientific disciplines, including chemistry, physics, biology, medicine, and materials science.
In biological systems, Fick’s Laws play central roles. Human respiration, for example, depends critically on diffusion. When air enters the lungs, oxygen concentration in the alveoli is much higher than in the blood arriving from the body. This difference creates a concentration gradient that drives oxygen diffusion across the thin membranes of the alveoli into the bloodstream. Simultaneously, carbon dioxide concentration is higher in the blood than in the alveolar air, causing carbon dioxide to diffuse in the opposite direction. Fick’s First Law describes the net rate of gas transfer at any moment, while the Second Law explains how concentrations equilibrate as breathing continues. Every inhalation and exhalation renews concentration gradients, ensuring a continuous supply of oxygen and removal of carbon dioxide. Without diffusion, life as we know it would not be possible.
In cellular processes, diffusion governs nutrient delivery, waste removal, and intracellular signaling. Small molecules such as glucose, oxygen, ions, and signaling chemicals diffuse across membranes and within the cytoplasm. The efficiency of diffusion depends on factors such as membrane thickness, molecular size, and the magnitude of concentration differences. Cells evolved thin membranes and large surface areas to maximize diffusion efficiency, demonstrating how biological systems adapt structurally to physical principles. Even processes such as nerve conduction rely on diffusion of ions to reestablish membrane potentials between action potentials. Thus, Fick’s Laws are woven into the fabric of life at the cellular and molecular levels.
In chemical engineering, Fick’s Laws guide the design of reactors, separation systems, and material fabrication processes. Engineers must understand how chemicals diffuse through porous materials, how gases diffuse in packed beds, and how liquids diffuse across membranes. Without accurate conceptual models of diffusion, systems such as distillation columns, catalytic converters, fuel cells, and polymer membranes could not be designed or optimized. Fick’s Laws help engineers anticipate how substances will move, how concentration profiles will change, and how quickly desired reactions or separations will occur.
In environmental science, diffusion governs the spread of pollutants in air, soil, and water. When contaminants enter groundwater, diffusion helps spread them beyond their initial source, affecting wider regions. In the atmosphere, diffusion helps disperse gases and particles, influencing air quality and pollution patterns. Understanding these diffusion processes allows environmental scientists to predict pollutant behavior, assess risks, and design remediation strategies.
In materials science, diffusion is essential for processes such as alloy formation, crystal growth, sintering, and heat treatment. At high temperatures, atoms in solids become more mobile and diffuse through crystal lattices, changing material properties. These diffusion processes influence the hardness, ductility, grain structure, and strength of metals and ceramics. Fick’s Laws help scientists understand how atoms migrate within materials and how this movement affects mechanical and structural behavior over time.
In pharmacology, diffusion determines how drugs move through tissues, how they cross membranes, and how they reach target sites. Many medications rely on passive diffusion to enter cells or spread within biological fluids. Understanding diffusion helps with designing effective drug delivery systems, controlling release rates, and predicting how drugs distribute within the body.
What makes Fick’s Laws remarkable is their ability to explain a vast range of processes using only conceptual principles grounded in random molecular motion. They show that diffusion is not a mysterious or chaotic process but a predictable, directional response to concentration differences. They bridge microscopic randomness and macroscopic order, revealing how countless tiny movements combine to produce smooth, large-scale gradients and flows.
A key insight from Fick’s Laws is that diffusion ultimately seeks equilibrium. This does not mean that molecules consciously move to achieve balance; rather, the natural randomness of molecular motion combined with statistical distribution leads to equalization of concentration differences over time. Once uniformity is achieved, no net diffusion occurs, although molecules continue moving randomly. This state of dynamic equilibrium reflects the fundamental nature of diffusion: movement continues endlessly, but without directional bias once gradients have vanished.
Fick’s Laws also illuminate why diffusion is slow over long distances. While diffusion is efficient at microscopic scales—such as within cells or across thin membranes—it becomes inefficient over macroscopic distances. Molecules take much longer to travel farther distances through random motion, as movement does not follow a straight path but involves countless collisions and random shifts. This is why biological systems use circulatory systems, convection, and active transport to move substances rapidly over long distances: diffusion alone would be too slow to sustain life at larger scales.
These principles also help explain why diffusion rates vary between gases, liquids, and solids. In gases, molecules move freely and rapidly, allowing diffusion to occur quickly. In liquids, molecular motion is slower due to closer packing, making diffusion more gradual. In solids, atoms are held in fixed lattice positions and can only diffuse at elevated temperatures or over long periods. Fick’s Laws apply conceptually to all states of matter but manifest differently based on the medium’s structure and the mobility of molecules or atoms.
Ultimately, Fick’s Laws of Diffusion provide a rich framework for understanding how molecules spread, how concentration gradients shape motion, and how natural systems evolve toward equilibrium. They reveal the underlying simplicity and beauty of diffusion as a universal process arising from random motion yet producing orderly, predictable outcomes. Through deeply detailed conceptual understanding, one sees how these laws govern the movement of particles in physical, biological, environmental, and technological systems, uniting diverse fields under a common scientific principle.