Types of Radiation — Ionizing and Non-Ionizing Energy
Radiation represents one of the most fundamental expressions of energy transfer in the universe, and understanding its types reveals a vast landscape of physical principles, biological interactions, technological applications, and natural phenomena. When we speak of radiation in the context of ionizing and non-ionizing energy, we refer to the broad spectrum of electromagnetic and particle-based energies that travel through space, interact with matter, and influence everything from the smallest atoms to the largest cosmic structures. Radiation is not inherently harmful or beneficial; rather, its effects depend on its energy, frequency, and ability to interact with the atomic and molecular structure of the materials it encounters. The division into ionizing and non-ionizing radiation reflects whether the energy carried is strong enough to remove electrons from atoms, a process known as ionization. This distinction forms the foundation of fields such as medical imaging, astronomy, telecommunications, nuclear physics, environmental health, and the fundamental study of electromagnetic waves. By exploring both categories in detail, we gain a deeper appreciation of how radiation shapes technology, supports scientific understanding, influences biological processes, and permeates the natural world.
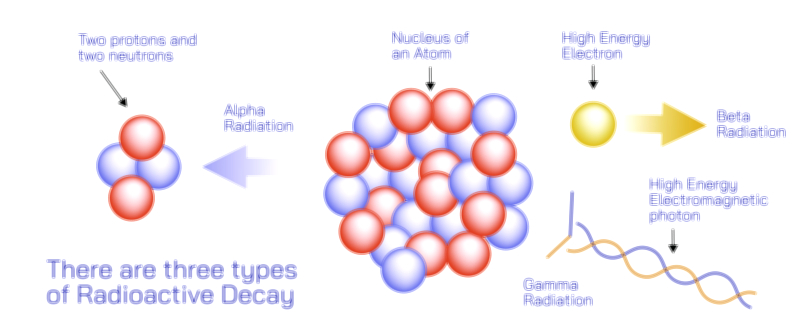
Ionizing radiation represents the category of radiation with enough energy to remove tightly bound electrons from atoms, creating ions. This process fundamentally alters the structure of matter and can cause significant changes in biological tissues, chemical structures, and physical materials. Ionizing radiation includes forms such as ultraviolet radiation of the highest frequencies, X-rays, gamma rays, and particle radiation like alpha particles, beta particles, and high-energy neutrons. These radiation types carry energies that interact with the electron clouds surrounding atoms, giving them sufficient force to disrupt chemical bonds. This property is what makes ionizing radiation essential for medical imaging, cancer treatment, sterilization, and scientific analysis, yet also why it must be handled with strict safety measures to prevent harmful exposure. Ionization can damage DNA, disrupt cellular replication, and initiate mutational changes, making careful control necessary in all settings where it is used. The ability of ionizing radiation to penetrate materials varies widely depending on the type: gamma rays can pass through thick layers of concrete or lead, while alpha particles may be stopped by a piece of paper or even the outer layer of human skin. These penetrating differences create unique applications and distinct safety profiles for each kind of ionizing radiation.
Gamma rays represent the highest-frequency form of electromagnetic radiation and are produced by nuclear reactions, radioactive decay, and cosmic phenomena. Their extremely high energy allows them to pass through dense materials and interact deeply with matter. In medicine, gamma rays are used in imaging and radiotherapy to diagnose and treat disease, particularly in targeting cancer cells. In astrophysics, the detection of gamma-ray bursts reveals information about supernova explosions and extreme cosmic events. X-rays occupy slightly lower frequencies and are widely known for their use in medical diagnostics, where they provide internal images of bones and organs. Their ability to pass through soft tissues but be absorbed by denser materials makes them invaluable for noninvasive imaging. High-frequency ultraviolet light, though just below the threshold of X-rays, still has enough energy to ionize molecules and damage biological tissues, causing sunburn and contributing to DNA mutation. Meanwhile, particle radiation such as alpha and beta particles arises from the decay of unstable atomic nuclei. Alpha particles consist of two protons and two neutrons, making them relatively massive and easily stopped, yet highly damaging if inhaled or ingested. Beta particles, which are high-speed electrons or positrons, carry less mass but greater penetration ability. Neutrons, which are uncharged, penetrate deeply and induce secondary radiation by interacting with atomic nuclei. Each type of ionizing radiation demonstrates how energy interacts at the subatomic level with profound physical and biological consequences.
In contrast, non-ionizing radiation encompasses the portion of the electromagnetic spectrum whose energy is insufficient to remove electrons from atoms. Instead of ionizing matter, non-ionizing radiation typically causes atoms and molecules to vibrate, rotate, or experience thermal effects. This category includes visible light, infrared radiation, microwaves, radio waves, and extremely low-frequency electromagnetic fields. Although non-ionizing radiation does not induce direct ionization, it plays highly significant roles in heating, communication systems, vision, sensing technologies, and biological processes. Visible light, the narrow band perceived by the human eye, is essential for photosynthesis, enabling plants to convert radiant energy into chemical energy that sustains nearly all life on Earth. Infrared radiation is associated with heat; every warm object emits infrared waves, and these waves are used in thermal imaging, remote control devices, and heating systems. Microwaves are used in communication technologies, radar systems, and microwave ovens, leveraging their ability to excite water molecules and generate heat. Radio waves carry telecommunications signals across the globe, allowing radios, cell phones, broadcast systems, and wireless networks to function. Even extremely low-frequency electromagnetic fields, though weak, are continuously present in environments due to power lines, electrical appliances, and natural sources.
Non-ionizing radiation interacts with matter in fundamentally different ways than ionizing radiation. Instead of altering atomic structures, it induces oscillation and motion, often resulting in gentle warming or changes in molecular alignment. This makes non-ionizing radiation essential in technologies that require safe and reliable communication across distances without damaging biological tissue. The safety considerations for non-ionizing radiation focus mainly on avoiding excessive heating effects. For example, microwaves must be enclosed in shielding to prevent accidental heating of surrounding materials, while infrared lasers require caution to avoid thermal burns. Yet, under normal usage conditions, non-ionizing radiation poses far fewer risks to living organisms compared to ionizing radiation. At the same time, non-ionizing radiation contributes enormously to technological innovation and daily life. Wireless communication, optical fiber networks, medical imaging techniques like MRI, and even simple lighting all depend on the properties of non-ionizing radiation.
The boundary between ionizing and non-ionizing radiation is not entirely sharp but rather defined by the amount of energy photons carry. Ionizing radiation begins roughly at the ultraviolet range where wavelengths become short enough and frequencies high enough to eject electrons. Below this, photons do not have sufficient energy to cause ionization, even though they may excite atomic and molecular movement. This distinction reveals how radiation transitions from benign heating or illumination effects to interactions that alter atomic structure. The electromagnetic spectrum itself is a continuous range of wavelengths and frequencies stretching from extremely low-frequency waves with kilometer-long wavelengths to ultra-high-energy gamma rays with wavelengths smaller than atomic nuclei. This spectrum demonstrates the unity of electromagnetic radiation: the same fundamental phenomenon manifests in radically different ways depending on scale, energy, and frequency.
Understanding the difference between ionizing and non-ionizing radiation also reveals how the natural world continuously bathes the Earth in radiation from cosmic, solar, geological, and biological sources. The Sun emits a broad spectrum of radiation that includes both non-ionizing components—infrared and visible light—and ionizing ultraviolet radiation. Earth’s atmosphere protects us by absorbing harmful high-energy radiation while allowing beneficial wavelengths to reach the surface. Cosmic rays, which include ionizing particles and gamma radiation, constantly interact with the atmosphere, creating secondary particles that reach ground level in small but measurable quantities. Within the Earth, radioactive decay of elements such as uranium and thorium produces ionizing radiation that contributes to geothermal heat. Even the human body contains trace amounts of radioactive isotopes such as potassium-40, contributing to internal ionizing radiation exposure. These natural background radiation sources show that radiation is an inherent part of the environment and essential to the planet’s balance.
In human society, the applications of radiation extend across fields ranging from medicine and scientific research to communications and industrial processes. Ionizing radiation enables medical imaging through X-rays, CT scans, and nuclear medicine techniques, while radiation therapy uses targeted ionization to destroy cancer cells. Industrial radiography uses radiation to inspect welded joints or detect internal flaws in materials. Scientific instrumentation relies on ionizing radiation to study atomic structures and nuclear reactions. At the same time, non-ionizing radiation supports telecommunications, heating, lighting, manufacturing, and sensor technologies. Fiber-optic systems transmit information using light; microwave towers carry internet and phone signals; lasers enable precision cutting and measurement; and infrared systems monitor environmental changes. Without both forms of radiation, modern civilization would lose many of its essential capabilities.
Despite the enormous benefits of radiation, understanding its types is crucial for managing associated hazards. Ionizing radiation requires strict regulatory guidelines due to its ability to damage tissue at the cellular level. Medical professionals, nuclear workers, and researchers use shielding, controlled exposure limits, and monitoring to ensure safety. Non-ionizing radiation also requires awareness, particularly with respect to powerful laser systems, microwave sources, and equipment that can produce excessive heating. Yet the risks associated with non-ionizing radiation under everyday conditions are generally far lower. The key lies in matching radiation type with appropriate safety measures and technological design.
Ultimately, the study of ionizing and non-ionizing radiation reveals how energy interacts with matter in ways that shape life, technology, and the universe at every scale. Radiation is not simply a scientific concept but a phenomenon that touches every aspect of existence, from the sunlight that warms the planet to the gamma rays released by distant stars. It illuminates, communicates, diagnoses, heals, heats, and transforms. Understanding these types of radiation deepens our recognition of how energy flows through the cosmos, how matter responds to these energies, and how humans harness, control, and coexist with the invisible waves that surround us.