Ammonium Phosphate — Structure, Properties, and Fertilizer Applications
Understanding the Chemical Structure of Ammonium Phosphate
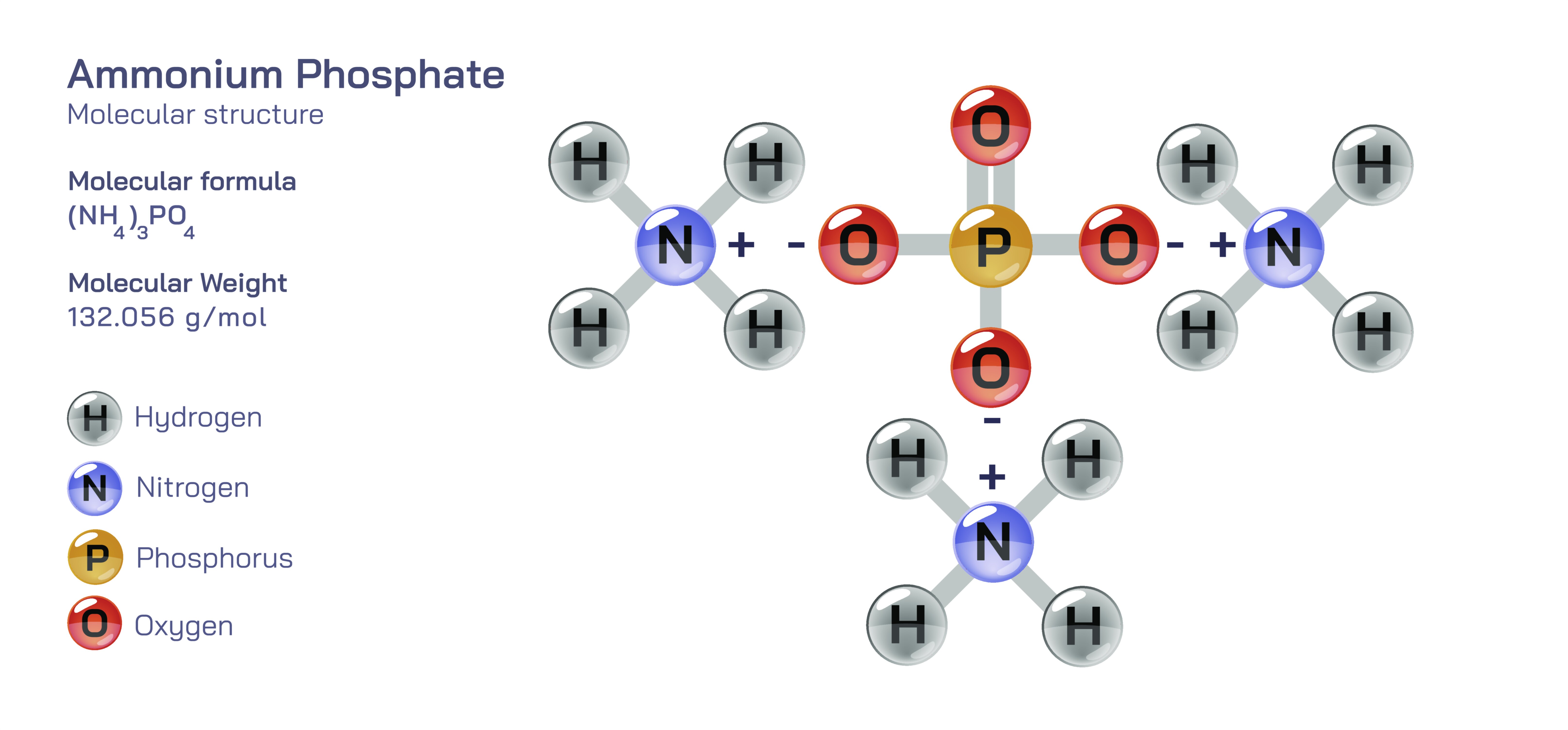
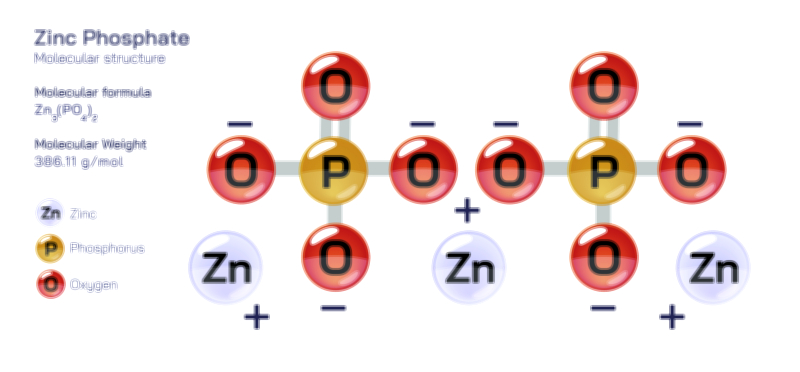
Ammonium phosphate refers to a family of inorganic salts formed by the reaction between ammonia and phosphoric acid, generally represented by formulas such as monoammonium phosphate (MAP, NH₄H₂PO₄) and diammonium phosphate (DAP, (NH₄)₂HPO₄). These compounds consist of ammonium ions bonded ionically to phosphate groups, forming stable crystalline structures that dissolve readily in water. The phosphate ion forms the structural core, while the ammonium ion binds through electrostatic attraction, creating a solid material that is easy to handle, transport, and store. In vector illustrations, the molecule is usually shown by highlighting the tetrahedral geometry of the phosphate group, surrounded by ammonium ions that balance the negative charge. This molecular representation helps viewers understand how each component of the compound contributes to its function: the ammonium provides nitrogen, while the phosphate group supplies phosphorus—two essential macronutrients required by plants for growth and reproduction. By visualizing how these ions interact, one gains a clearer sense of why ammonium phosphate behaves predictably in soil and solution, and why it is valued so highly in agriculture.
Physical and Chemical Properties That Influence Its Behavior
Ammonium phosphate salts display several important properties that determine their performance as fertilizers and industrial chemicals. They are highly soluble in water, allowing them to release nutrients quickly once applied to soil. Their crystalline nature makes them easy to granulate and blend with other fertilizers, forming uniform, free-flowing pellets suited for large-scale agricultural distribution. MAP has an acidic reaction when dissolved, while DAP has a mildly alkaline initial effect that transitions to slight acidity as ammonium converts to nitrate. Both variants contain high concentrations of nutrients: MAP provides a balanced ratio with slightly more phosphorus, while DAP offers a higher nitrogen content along with substantial phosphorus. These compounds remain stable under normal environmental conditions and resist decomposition unless exposed to extreme heat. Their predictable dissolution rate ensures that plants can access nutrients promptly during early growth stages. Vector illustrations often show ammonium phosphate dissolving into ammonium and phosphate ions in water, visually representing how the compound becomes immediately available for plant absorption.
Role of Ammonium Phosphate as a Fertilizer
Ammonium phosphate is one of the most widely used fertilizers in global agriculture due to its dual supply of nitrogen and phosphorus—macronutrients that are critical for plant development. Nitrogen promotes vigorous leaf growth, protein formation, and chlorophyll production, while phosphorus supports root development, flowering, energy transfer, and cellular metabolism. Because ammonium phosphate delivers both nutrients in a single compound, farmers can apply it efficiently to stimulate early growth in crops such as wheat, maize, rice, vegetables, fruit trees, and legumes. The phosphorus content, being in highly soluble form, is immediately available to young roots, which is essential during the early stages of plant establishment. The ammonium form of nitrogen helps minimize leaching losses because it binds to soil particles until converted by microbes into nitrate. This behavior makes ammonium phosphate particularly effective in soils that are prone to nitrogen losses. In vector illustrations, ammonium phosphate granules are often shown breaking down into ions near plant roots, emphasizing how the nutrient release aligns with plant needs during critical growth phases.
Soil Interactions and Nutrient Availability
When ammonium phosphate dissolves in soil, the ammonium ions can temporarily lower pH in the surrounding zone as they are converted through nitrification, while the phosphate ions interact with soil particles depending on pH and mineral content. In acidic soils, phosphorus may react with iron and aluminum compounds, and in alkaline soils, it may bind with calcium—both processes can reduce phosphorus availability. However, the high solubility of ammonium phosphate ensures sufficient immediate phosphorus release before strong fixation occurs. This makes it especially useful for broadcasting before planting or banding during seed sowing, where it remains accessible during early root development. The ammonium ion provides a localized acidifying effect that helps keep phosphorus soluble for longer periods in alkaline soils. Vector illustrations often depict these interactions by showing ions spreading through soil layers, with roots absorbing nutrients, helping viewers visualize how soil chemistry influences fertilizer efficiency.
Different Forms: MAP and DAP in Agricultural Practice
Monoammonium phosphate (MAP) is commonly used where crops benefit from a slightly acidic root-zone environment or where soil pH is already high. Its lower nitrogen content compared to DAP allows precise phosphorus application without excess nitrogen. Diammonium phosphate (DAP), on the other hand, is favored where a stronger nitrogen boost is needed alongside phosphorus, especially during early growth. DAP’s initial alkalinity must be considered in very sensitive crops or extremely alkaline soils, but it generally transitions quickly to a soil-acidifying effect. Both forms provide high nutrient density, making them ideal for precision agriculture and bulk-field application. Vector illustrations often show MAP and DAP side by side with nutrient percentages labeled, making it easier to compare their chemical makeup and crop suitability.
Industrial and Non-Agricultural Uses of Ammonium Phosphate
Beyond agriculture, ammonium phosphate is used in multiple industries due to its chemical stability, fire-retardant behavior, and buffering properties. It appears in fire extinguishers, where it helps smother flames and prevent re-ignition by forming a heat-resistant coating upon decomposition. Certain grades of ammonium phosphate are used in food processing as acidity regulators, leavening agents, and nutrient additives. The compound also appears in metal finishing, fermentation processes, and laboratory reagents in biochemical research. In firefighting foams and coatings, ammonium phosphate helps interrupt the chemical reactions that sustain flames, making it valuable for industrial fire suppression. Vector illustrations in industrial contexts often show ammonium phosphate being sprayed as fire retardant or added to chemical reactors, highlighting its versatility beyond plant nutrition.
Environmental Considerations and Best Practices for Use
Although ammonium phosphate is effective and widely used, its environmental impact depends on responsible management. Over-application can lead to phosphorus runoff into waterways, contributing to algal blooms and ecosystem imbalance. Nitrogen from the ammonium component can also contribute to groundwater contamination if poorly managed. Sustainable use requires soil testing, calibrated application rates, and timing that aligns with crop nutrient uptake patterns. Precision agriculture technologies increasingly rely on ammonium phosphate due to its predictable nutrient composition, allowing farmers to tailor fertilization strategies for maximum efficiency. Vector diagrams related to sustainability often show ammonium phosphate within nutrient cycles, emphasizing balanced application and environmental stewardship.
Illustrating Ammonium Phosphate Structure and Applications Through Vector Graphics
A vector illustration of ammonium phosphate often combines molecular and real-world perspectives: tetrahedral phosphate groups and ammonium ions on one side, and agricultural field applications on the other. The visual may include granules dissolving into soil solutions, root uptake pathways, or nutrient transport processes within plants. These clear, stylized graphics help bridge the gap between chemical structure and agricultural function, making complex concepts easier to grasp for students, agronomists, and industry professionals.