Zinc Chloride — Structure, Properties, and Industrial Applications
Fundamental Identity and Structural Characteristics of Zinc Chloride
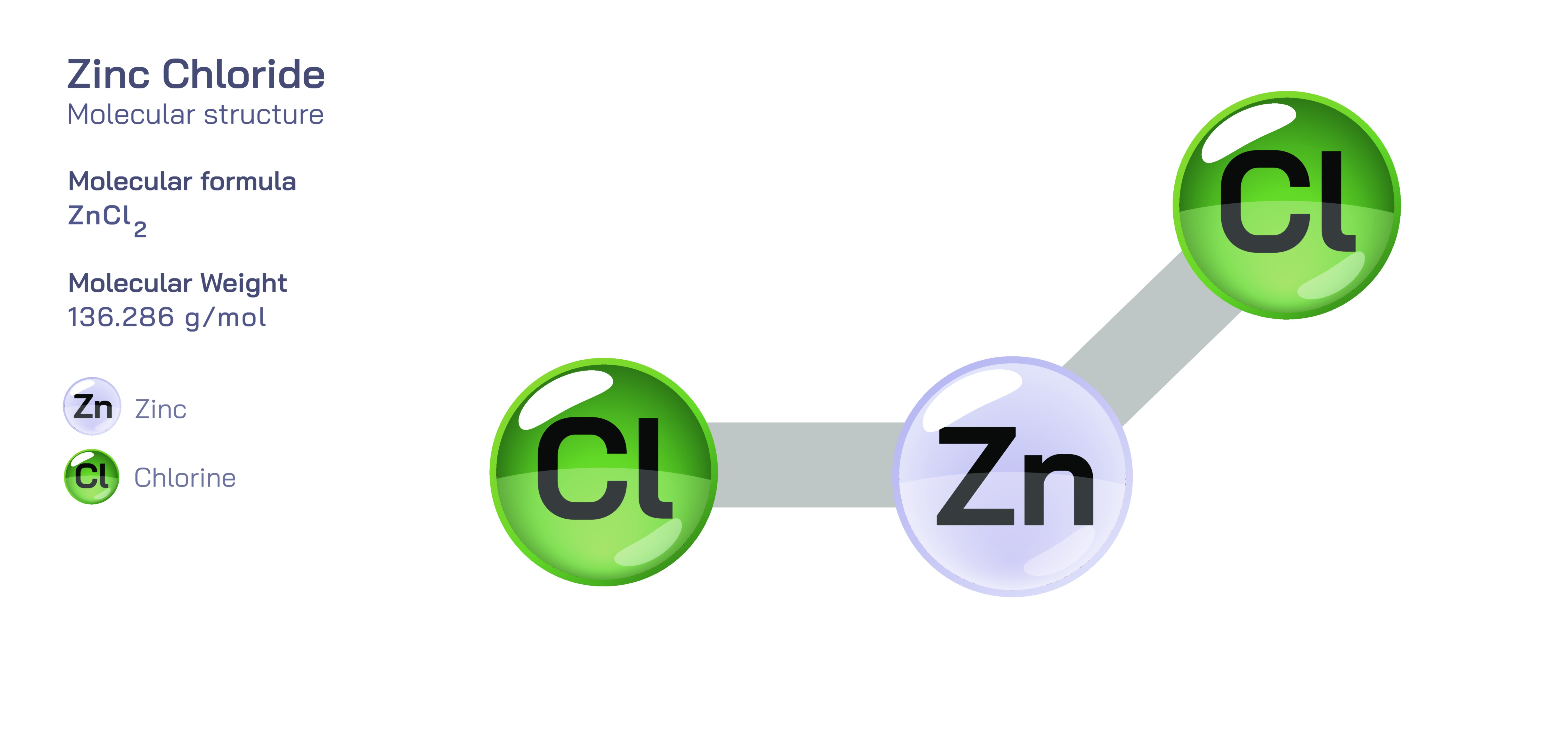
Zinc chloride, commonly represented by the formula ZnCl₂, is an important inorganic compound known for its strong Lewis acidity, high solubility, and versatility across chemical and industrial applications. Although it appears simple as a combination of zinc and chloride ions, the compound forms a variety of structural arrangements depending on whether it exists in solid, molten, or dissolved form. In the solid state, zinc chloride exhibits complex polymeric structures in which zinc ions typically coordinate with four chloride ions in a tetrahedral environment. These tetrahedra connect in extended chains or networks, giving the crystal lattice a highly organized but flexible geometry influenced by bonding strength and ionic polarization. Unlike purely ionic salts such as sodium chloride, ZnCl₂ displays a pronounced covalent character due to the relatively small size and high charge density of the Zn²⁺ ion combined with the polarizable nature of chloride ions. This covalent influence affects melting point behavior, solvation patterns, and its ability to form complex species. Because of this structural adaptability, zinc chloride appears in several polymorphic forms and readily transitions into highly conductive molten or hydrated phases, each offering specialized functions for scientific and industrial use.
Bonding Behavior, Lewis Acidity, and Solution Chemistry
The behavior of zinc chloride is governed by a combination of ionic attraction and covalent bonding, with Zn²⁺ acting as a strong Lewis acid capable of accepting electron pairs from various donors. This acidity becomes evident in solution, where ZnCl₂ forms a range of complex ions, including [ZnCl₄]²⁻, [ZnCl₃]⁻, and mixed hydroxy complexes when water participates in coordination. In aqueous environments, zinc chloride dissolves readily, producing acidic solutions due to hydrolysis of Zn²⁺ ions, which generate hydronium ions. This acidity enables ZnCl₂ to catalyze dehydration reactions, activate alcohols in organic synthesis, and participate in chlorination pathways when combined with other reagents. In molten form, zinc chloride behaves as an ionic liquid with high conductivity and the ability to dissolve organic compounds, cellulose, and even certain metal oxides. This combination of Lewis acidity and structural flexibility makes ZnCl₂ a valuable reagent in chemical laboratories, where it serves as a catalyst, dehydrating agent, and component in specialized reaction media. Its interaction with ammonia, organic solvents, and ligands also forms coordination complexes that reinforce its role as a multipurpose inorganic compound.
Physical Properties and Their Functional Implications
Zinc chloride is a white, crystalline solid that is highly soluble in water, alcohols, and many polar organic solvents. Its solubility is among the highest of all zinc salts, allowing it to form viscous, concentrated solutions used in industrial formulations. The anhydrous form has a high melting point and is deliquescent, meaning it readily absorbs moisture from the air until it dissolves. This hygroscopic behavior is tied to its strong affinity for water, making ZnCl₂ an effective drying agent for organic solvents and gases. When heated, zinc chloride transitions into a molten salt that remains stable across a broad temperature range and exhibits excellent ionic conductivity. These molten salts find use in electrochemical applications and metal fluxes. The compound’s aggressive interaction with water and organic materials gives it powerful dehydrating capabilities, which can break down cellulose fibers or promote catalysis in organic transformations. Its physical characteristics—especially solubility, hygroscopicity, and melt behavior—support diverse applications from metallurgy to chemical synthesis, reflecting the inherent versatility of zinc chloride’s structure.
Chemical Reactivity, Stability, and Transformation Pathways
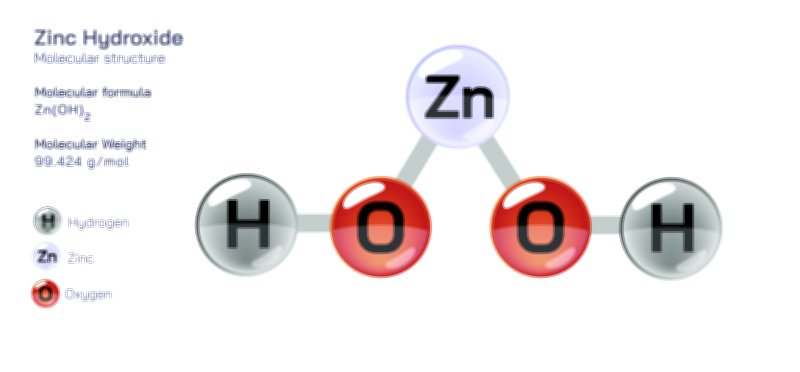
Chemically, zinc chloride is reactive yet predictable, participating in a wide range of transformations due to its Lewis acidity and ability to coordinate with electron-rich species. It reacts with bases to form zinc hydroxide, which can further transform into zinc oxide upon heating. In the presence of metal oxides or salts, molten ZnCl₂ can act as a flux, dissolving oxide layers and facilitating metal joining or purification. Its reaction with ammonia produces complex zinc-ammonia species, while interaction with phosphates or carbonates yields insoluble zinc salts that precipitate out of solution. In organic chemistry, ZnCl₂ is valued for promoting Friedel–Crafts alkylation and acylation reactions, aiding in the formation of carbon–carbon bonds by polarizing electrophiles. It also catalyzes dehydration of alcohols, conversion of amides to nitriles, and preparation of alkyl chlorides from alcohols when used in conjunction with hydrochloric acid. These reaction pathways demonstrate the adaptability of zinc chloride, with its Lewis acidic center enabling diverse chemical transformations that are essential in both laboratory and industrial settings.
Industrial Applications Across Manufacturing, Construction, and Technology
Zinc chloride holds a central place in numerous industries due to its reactivity, solubility, and functional reliability. One major application lies in the production of fluxes used for soldering and galvanizing. Zinc chloride-based fluxes remove oxide layers from metal surfaces, ensuring clean, strong joints during soldering and coating processes. In the fabrication of batteries—especially dry cell batteries—zinc chloride serves as an electrolyte that enhances performance and prevents corrosion, replacing or supplementing ammonium chloride in modern formulations.
In the textile and paper industries, concentrated ZnCl₂ solutions dissolve cellulose fibers, allowing the production of specialty fabrics, rayon, and vulcanized fiber materials. Wood preservation treatments historically used zinc chloride to inhibit microbial growth and protect against decay, though more environmentally friendly alternatives have replaced many such applications. In water treatment systems, ZnCl₂ assists in coagulation processes and helps remove impurities when used with other chemical agents.
Its role in organic synthesis includes catalysis, dehydrating functions, and activation of key organic intermediates. Additionally, zinc chloride is used in petroleum refining, dye manufacturing, and adhesive production, where its chemical responsiveness supports crucial industrial reactions. Even in modern materials science, zinc chloride is employed to prepare activated carbon by facilitating pore formation during carbonization. This activated carbon is used in filtration, purification, and environmental remediation applications.
Safety, Environmental Considerations, and Handling Precautions
Despite its usefulness, zinc chloride must be handled with care due to its corrosive nature, strong acidity in solution, and potential environmental impact. It can cause severe skin and eye irritation and must be stored in airtight containers to prevent moisture absorption. In industrial settings, proper ventilation and protective equipment are essential. Environmental concerns stem from zinc’s role as a heavy metal; excessive release into natural waters can harm aquatic ecosystems. Waste solutions containing ZnCl₂ require controlled neutralization or recovery before disposal, often through precipitation as zinc hydroxide followed by filtration. Responsible handling ensures that the compound’s benefits are realized while minimizing risks to health and the environment.
Illustrating the Structure and Functional Behavior of Zinc Chloride
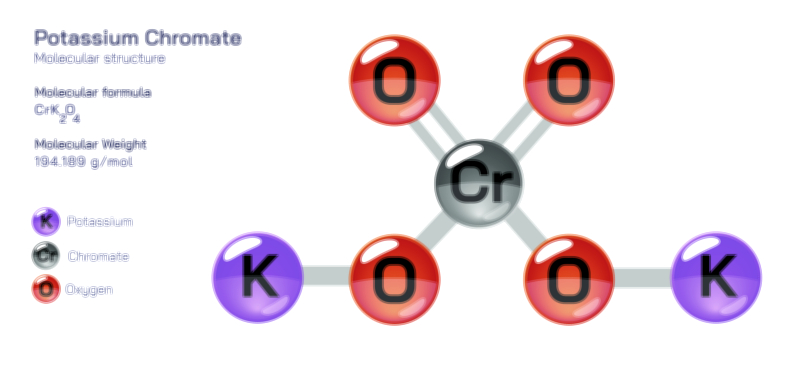
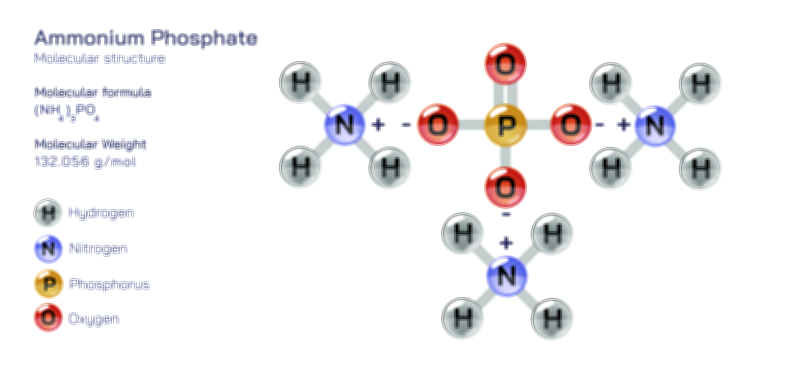
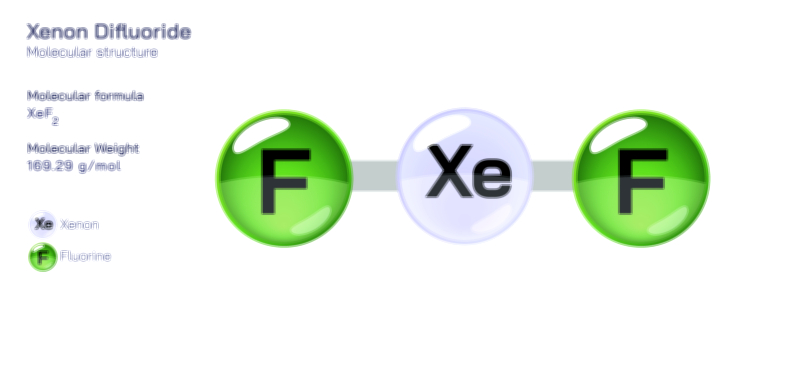
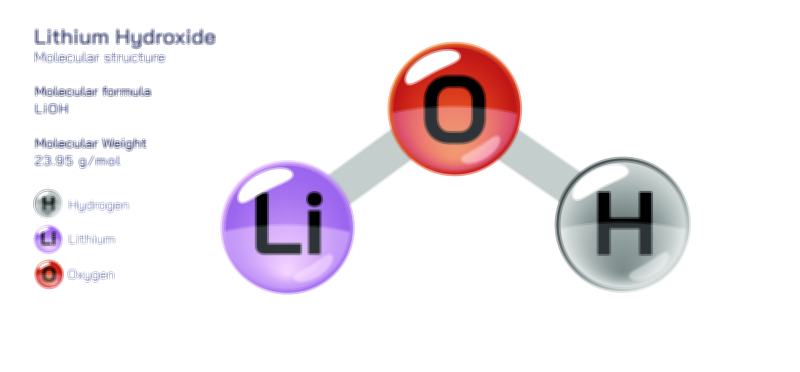
A vector illustration of zinc chloride typically visualizes the tetrahedral ZnCl₄ units in solid or coordinated forms, showing how zinc sits at the center bound to surrounding chloride ions. Diagrams may also contrast the polymeric solid-state structure with the fully dissociated ionic arrangement seen in aqueous solution. Industrial illustrations often depict zinc chloride as part of soldering fluxes, battery electrolytes, cellulose-processing tanks, or reaction vessels where it acts as a catalyst. These visuals clearly connect molecular behavior to functional applications, demonstrating how the compound’s structural flexibility and reactivity shape its diverse roles across chemistry and industry.