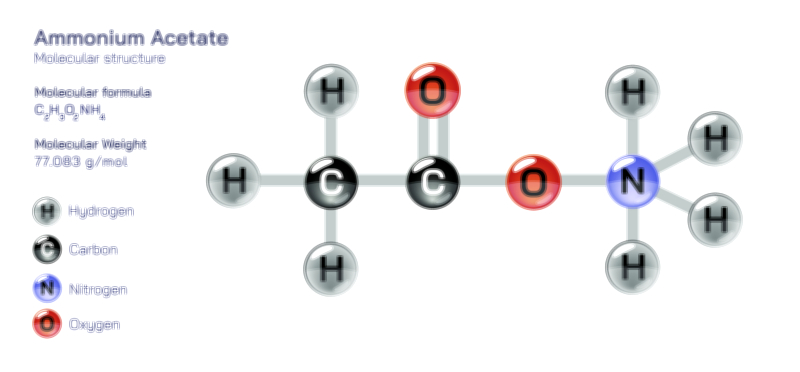
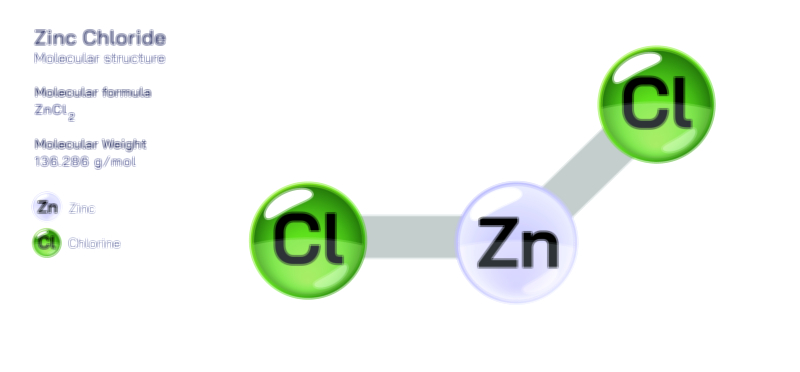
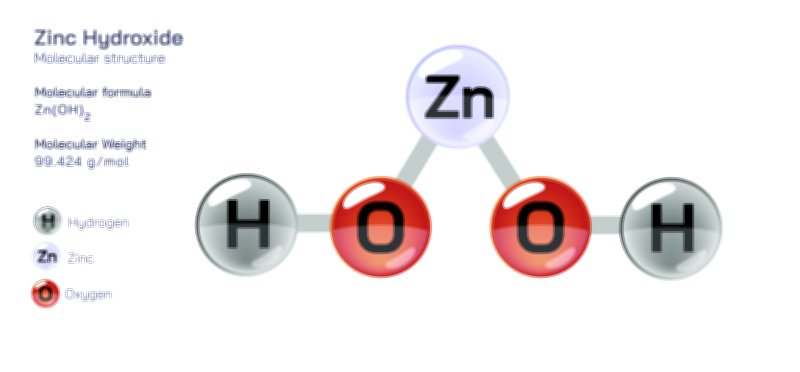
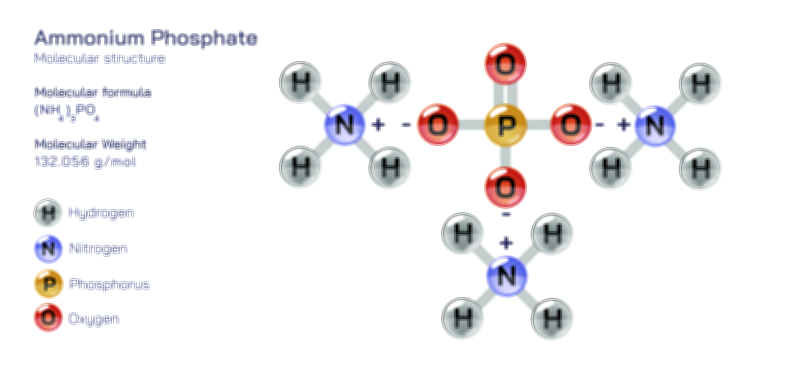
Zinc Acetate — Structure, Properties, and Medical Applications
Fundamental Identity and Structural Characteristics of Zinc Acetate
Zinc acetate, typically written as Zn(CH₃COO)₂ or Zn(OAc)₂, is an inorganic–organic salt formed through the interaction of zinc ions with acetate ions derived from acetic acid. It commonly appears in two forms: anhydrous zinc acetate and zinc acetate dihydrate (Zn(CH₃COO)₂·2H₂O), the latter being more prevalent in pharmaceutical and laboratory settings due to its stability and ease of handling. Structurally, the zinc ion exists in a coordination environment where it binds to oxygen atoms of acetate groups, forming arrangements that may be tetrahedral, octahedral, or polymeric depending on hydration, concentration, and phase conditions. In the dihydrate form, zinc often coordinates with four oxygen atoms from acetate groups and two water molecules, yielding an octahedral geometry with bridging interactions that extend throughout the crystal lattice. The acetate ion itself features a central carbonyl group (C=O) and a hydroxyl-derived oxygen atom, arranged in a planar configuration that stabilizes the resonance structure of the acetate group. This combination of ionic and covalent bonding generates a compound that is both soluble and structurally adaptable, allowing zinc acetate to participate readily in biological systems, organic synthesis, and industrial chemical processes.
Chemical Behavior, Dissociation, and Reactivity Patterns
When dissolved in water, zinc acetate readily dissociates into Zn²⁺ cations and acetate anions, a process facilitated by the strong hydration tendencies of zinc and the resonance stability of the acetate ion. The resulting aqueous solution is mildly acidic, reflecting the interaction of zinc ions with water molecules to form partially hydrolyzed complexes. Zinc acetate participates in a wide range of reactions owing to the Lewis acidity of Zn²⁺ and the nucleophilic nature of acetate ions. In acidic conditions, the acetate groups become protonated, yielding acetic acid while freeing zinc ions to form alternative salts. In alkaline environments, zinc acetate reacts with hydroxide ions to produce zinc hydroxide, which can subsequently convert into zinc oxide upon heating. The compound also participates in ligand-exchange reactions, forming complexes with ammonia, amino acids, proteins, organic acids, and other biological molecules. These interactions form the chemical basis for many of zinc acetate’s biological and medical functions, where the zinc ion must be delivered in a form that is both stable and bioavailable. Additionally, in laboratory synthesis, zinc acetate often serves as a precursor for zinc oxide nanoparticles, metal–organic frameworks, and coordination polymers, demonstrating its versatility and reactivity in diverse chemical pathways.
Physical Properties and Their Functional Significance
Zinc acetate appears as a white, crystalline solid with a characteristic slightly acetic odor due to residual acetic acid or partial decomposition under humid conditions. The compound dissolves readily in water, ethanol, and other polar solvents, forming clear solutions ideal for pharmaceutical preparation and chemical formulation. The dihydrate form melts at relatively low temperatures and loses water molecules progressively upon heating, transitioning into the anhydrous salt. Both forms decompose before reaching full boiling, making thermal behavior a key factor in industrial and scientific handling. The hygroscopic nature of zinc acetate, particularly the anhydrous variety, requires careful storage to prevent moisture uptake and subsequent alteration of physical properties. These features—high solubility, moderate volatility of acetic components, and hydration-dependent stability—support its widespread use in medical, nutritional, biochemical, and materials science contexts, where controlled zinc release or reactivity is essential.
Medical and Therapeutic Applications of Zinc Acetate
Zinc acetate is best known for its prominent role in medical applications, especially as a zinc supplement and therapeutic agent. One of its most recognized uses is in the treatment of Wilson’s disease, a genetic disorder in which copper accumulates abnormally in the body. Zinc acetate acts as a copper-chelating therapy by inducing metallothionein synthesis in intestinal cells. Metallothionein binds dietary copper and prevents its absorption into the bloodstream; instead, the copper-bound cells are shed naturally through normal gastrointestinal turnover, thereby reducing systemic copper levels. Zinc acetate is often preferred for long-term copper management because it is effective, well tolerated, and suitable for chronic treatment without the toxicity risks associated with stronger chelating agents.
In general nutrition, zinc acetate appears in lozenges designed to shorten the duration and severity of the common cold. Zinc is thought to inhibit viral replication and stabilize cell membranes, although the effectiveness of zinc lozenges depends on formulation specifics, including zinc ion availability and the absence of chelating additives that would suppress activity. In dermatology, zinc acetate is included in topical formulations to reduce inflammation, soothe irritation, and support wound healing. Its mild antimicrobial properties also contribute to its use in treating acne, diaper rash, and minor skin infections. Because zinc is an essential trace element necessary for enzyme function, immune activity, DNA synthesis, and cellular repair, zinc acetate provides a highly bioavailable form that supports multiple physiological processes.
Industrial, Chemical, and Material Science Uses
Beyond its medical relevance, zinc acetate plays a notable role in industrial and chemical applications. It is widely used as a catalyst in polyester production, where it accelerates esterification and transesterification reactions. In dyeing and textile processing, zinc acetate serves as a mordant—an agent that binds dyes to fabric fibers, improving color fastness. In the production of wood preservatives, latex products, and chemical coatings, zinc acetate acts as a stabilizer or activator, influencing the polymerization or setting characteristics of the final product.
In materials science, zinc acetate is a primary precursor for synthesizing zinc oxide nanoparticles through sol–gel, hydrothermal, and combustion-based methods. These nanoparticles hold value in electronics, cosmetics, photonics, catalysts, and antibacterial coatings. Zinc acetate is also used to manufacture specialized zinc-containing materials such as zinc-based metal–organic frameworks (MOFs), which are valued for gas storage, catalysis, and environmental purification. Its ability to coordinate with organic ligands makes it a versatile starting material for designing functional materials with unique structural and chemical properties.
Biochemical Significance and Environmental Considerations
In biochemical contexts, zinc acetate is used to precipitate proteins and nucleic acids under controlled conditions, enabling researchers to purify biological macromolecules or study their structural behavior. The acetate ion functions as a mild buffer component, maintaining solution stability in enzymatic or cellular assays. Environmentally, zinc acetate is considered moderately safe when used responsibly, though excessive zinc discharge can disrupt aquatic ecosystems. Proper waste management and controlled dosing are necessary to minimize ecological impact.
Illustrating the Structure and Functional Behavior of Zinc Acetate
A vector illustration of zinc acetate typically depicts the Zn²⁺ ion at the center, coordinated by oxygen atoms from surrounding acetate groups. The acetate ion is drawn with its characteristic carbonyl and hydroxyl-derived oxygen atoms in a planar, resonance-stabilized configuration. Illustrations may show octahedral coordination in hydrated forms or polymeric linkages extending through the lattice. Medical diagrams often highlight zinc acetate’s action in copper-binding pathways, while industrial illustrations show its decomposition into zinc oxide or its use in catalytic polymerization processes. These visuals reinforce how the compound’s structural and chemical attributes directly influence its practical utility across medicine, science, and industry.