Zinc Phosphate — Structure, Formula, and Applications
Fundamental Identity and Structural Nature of Zinc Phosphate
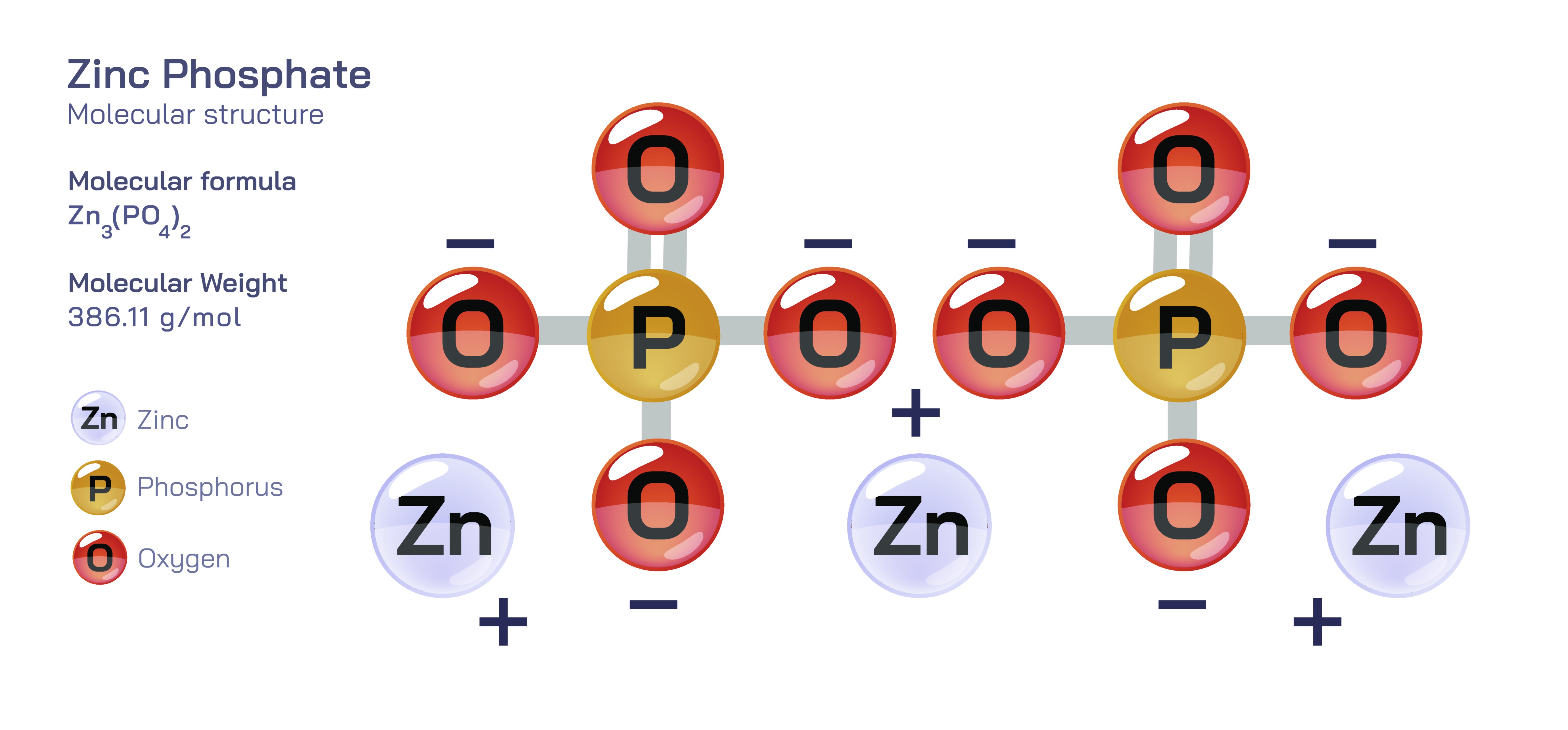
Zinc phosphate is an inorganic compound primarily represented by the formula Zn₃(PO₄)₂, though it often appears in various hydrated forms such as zinc phosphate tetrahydrate (Zn₃(PO₄)₂·4H₂O) or as complex coatings formed during industrial processes. The structure of zinc phosphate reflects the interaction between zinc ions, which carry a +2 charge, and phosphate ions, PO₄³⁻, which possess a tetrahedral geometry. These ions form an extended lattice in which zinc coordinates with oxygen atoms from the phosphate groups, creating a three-dimensional network that is both strong and chemically stable. The phosphate ion, with its central phosphorus atom bonded to four oxygen atoms, interacts with multiple zinc centers, leading to a cross-linked structure that imparts rigidity and low solubility. Because of this intricate structural arrangement, zinc phosphate is recognized for its durability, corrosion resistance, and low reactivity in neutral environments. Its ability to form stable coatings on metals, its biological compatibility, and its environmental safety have made it a crucial material in manufacturing, medicine, and protective surface treatments.
Chemical Structure, Bonding Behavior, and Formula Significance
Chemically, zinc phosphate is characterized by its ionic bonding framework. The Zn²⁺ cations coordinate with partially negative oxygen atoms from the PO₄³⁻ anions, producing a lattice that balances charge through complex connectivity patterns. Although zinc typically exhibits octahedral coordination in hydrated forms, anhydrous zinc phosphate structures often contain less symmetrical configurations depending on the arrangement of phosphate groups. These tetrahedral phosphate units share oxygen atoms with neighboring zinc centers, forming a network that resists dissolution in water. The formula Zn₃(PO₄)₂ reflects the requirement for charge neutrality: three Zn²⁺ ions contribute a total charge of +6, which balances the −6 charge of two PO₄³⁻ ions. Hydrated versions incorporate water molecules that coordinate with zinc or occupy interstitial positions in the crystalline lattice, altering the compound’s solubility and mechanical behavior while preserving its essential ionic framework. The resulting structures allow zinc phosphate to interact effectively with metal surfaces, tooth enamel, and biological tissues, depending on the form and preparation method used.
Physical Properties and Functional Behavior of Zinc Phosphate
Zinc phosphate typically appears as a white crystalline powder or as a tightly adherent coating when formed during chemical conversion processes. It is only sparingly soluble in water, a property that contributes greatly to its usefulness in protective applications. The compound exhibits high thermal stability, maintaining structural integrity under conditions that would degrade many organic materials. Its hardness and chemical inertness make it resistant to weathering, acids, and moisture, though it can react gradually in highly acidic or strongly basic environments. Hydrated forms of zinc phosphate may have slightly different textures and solubilities, but they share the essential characteristic of forming dense, protective films when interacting with metal ions. These films reduce surface porosity, enhance corrosion resistance, and provide excellent adhesion for paints and coatings. In dentistry, zinc phosphate cement presents a somewhat different physical profile: it forms a fine crystalline matrix after mixing powder with phosphoric acid solution, generating a strong, rigid, and durable material ideal for long-lasting restorations. Across all forms, zinc phosphate distinguishes itself through its mechanical strength, low solubility, chemical stability, and capacity for forming protective or structural layers.
Reactivity, Formation Mechanisms, and Chemical Interactions
Zinc phosphate is usually produced through controlled reactions between zinc salts (such as zinc oxide or zinc nitrate) and phosphate salts (such as sodium phosphate or phosphoric acid). In aqueous environments, the reaction leads to the formation of precipitates whose size, hydration level, and crystallinity depend on pH, temperature, and ion concentrations. When used in metal treatment processes, zinc phosphate coatings form through a multistep mechanism involving metal dissolution, phosphate deposition, and crystal growth on the substrate. This chemical conversion results in a solid matrix that adheres strongly to the metal surface, creating a barrier that prevents oxidation and improves paint bonding. In dental cement chemistry, zinc phosphate forms through acid–base neutralization between zinc oxide powder and phosphoric acid liquid, leading to the precipitation of zinc phosphate crystals that grow into a hardened restorative material. The reactivity of zinc phosphate with acids in the mouth or industrial environments is slow, enabling long-term stability. These chemical behaviors illustrate how zinc phosphate transitions from ionic species in solution to complex, solid-state structures with impressive mechanical and protective qualities.
Industrial, Medical, and Protective Applications of Zinc Phosphate
Zinc phosphate’s most prominent industrial application is its role as a corrosion-resistant coating for metal surfaces. Phosphate coatings are widely used in the automotive, construction, and machinery industries, where they serve as a base layer that enhances paint adhesion and prevents rust formation. These coatings also reduce friction between moving parts, extend service life, and provide a uniform surface texture for subsequent finishing steps. In the realm of protective materials, zinc phosphate is valued for its environmental safety compared to older anti-corrosion agents such as chromates, which pose significant health and ecological risks.
In dentistry, zinc phosphate cement has long been used for securing crowns, bridges, orthodontic bands, and other restorations. Its rigidity, strength, and biocompatibility make it a reliable choice even in the era of modern adhesive materials. It continues to be used because of its predictable setting behavior and mechanical robustness.
In pigment and paint manufacturing, zinc phosphate functions as an anti-corrosive pigment, replacing toxic alternatives while providing excellent performance in protective coatings. Its low solubility and stability ensure that it remains intact even under challenging environmental conditions. In chemical synthesis and catalysis, zinc phosphate appears in certain heterogeneous catalysts, while in biomedical fields, modified forms of zinc phosphate are being investigated for drug delivery, bone repair, and tissue engineering due to their biocompatibility and mineral-like character.
Illustrating the Structure and Functional Behavior of Zinc Phosphate
Scientific illustrations of zinc phosphate typically highlight the tetrahedral phosphate ion bonding to zinc centers, forming extended three-dimensional networks. Diagrams often depict the crystalline lattice, showing how Zn²⁺ ions coordinate with oxygen atoms to create stable ionic frameworks. For metal surface treatments, illustrations reveal the microscopic crystal layers that develop during the phosphating process, emphasizing how these layers adhere to and protect steel or other metals. In dentistry, visual diagrams show the chemical reaction between zinc oxide and phosphoric acid, the growth of zinc phosphate crystals, and the formation of the hardened cement matrix. In pigment chemistry, illustrations may portray how zinc phosphate particles integrate into protective paint films, improving corrosion resistance by blocking moisture and oxygen penetration. Through these visual representations, the structural logic and functional versatility of zinc phosphate become readily apparent, demonstrating why this compound maintains such a prominent role across industries, science, and medicine.