Wave and Particle Properties — Quantum Dual Nature Explained
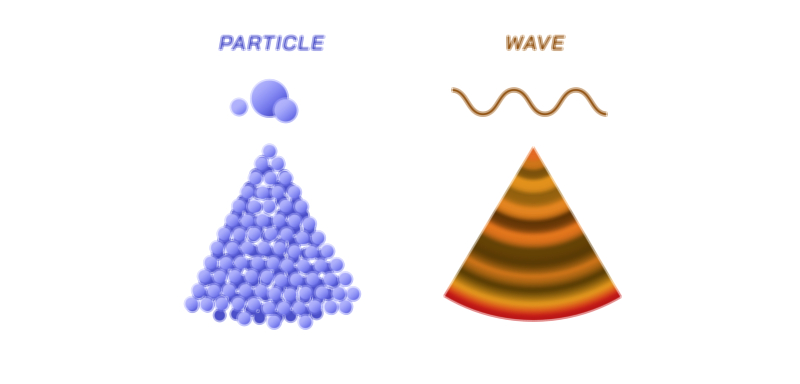
The quantum world challenges the traditional boundaries of physics because microscopic entities such as electrons, photons, neutrons, and even atoms do not behave exclusively as particles or waves, but instead exhibit both properties depending on the situation. This concept, known as wave–particle duality, stands at the foundation of quantum mechanics and reshapes our understanding of matter and energy. While classical physics distinguishes waves and particles as two distinct categories — waves spreading continuously through space and particles existing as localized points — quantum physics reveals that the microscopic world cannot be confined to this either–or viewpoint. A single quantum object can show interference and diffraction like a wave, yet can also appear as a localized object transferring energy in discrete packets called quanta. The dual nature does not reflect two separate identities but rather a single quantum entity described by a mathematical wave function that predicts probability rather than certainty. What we observe — wave behavior or particle behavior — depends on how we measure it.
To better understand the wave-like aspect, consider experiments where particles produce interference patterns, the hallmark of wave physics. In the famous double-slit experiment, a beam of light or a stream of electrons is directed toward two narrow slits closely spaced in a barrier. If electrons behaved like classical particles, they would simply pass through one slit or the other and produce two bands on the screen behind the barrier. Instead, what appears is a series of bright and dark fringes — an interference pattern identical to the pattern formed by waves such as ripples on water. Even more astonishing, if electrons are sent through the slits one at a time, the interference still gradually forms over time. This means individual electrons do not hit the screen randomly; instead, each electron behaves like a probability wave passing through both slits simultaneously and interfering with itself. This experiment demonstrates that quantum objects extend in space like waves and cannot be confined to a single, deterministic path until a measurement is made.
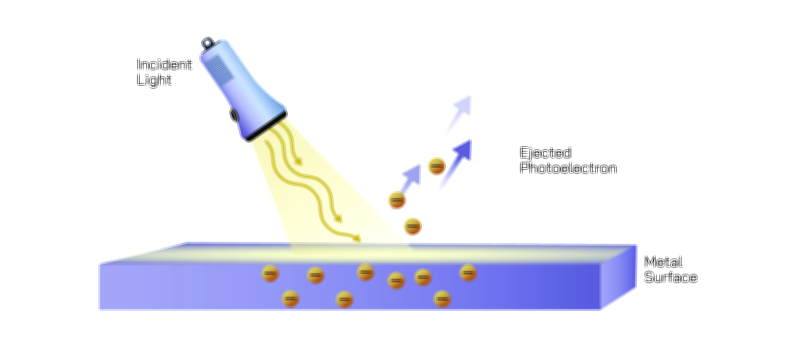
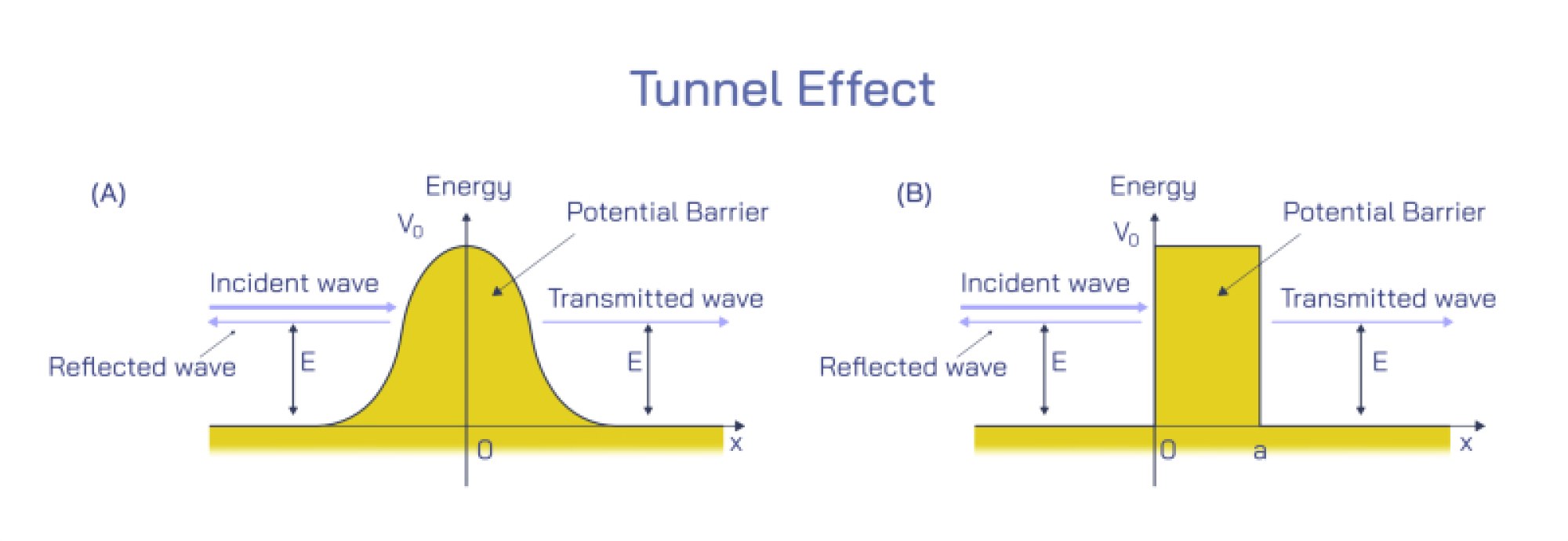
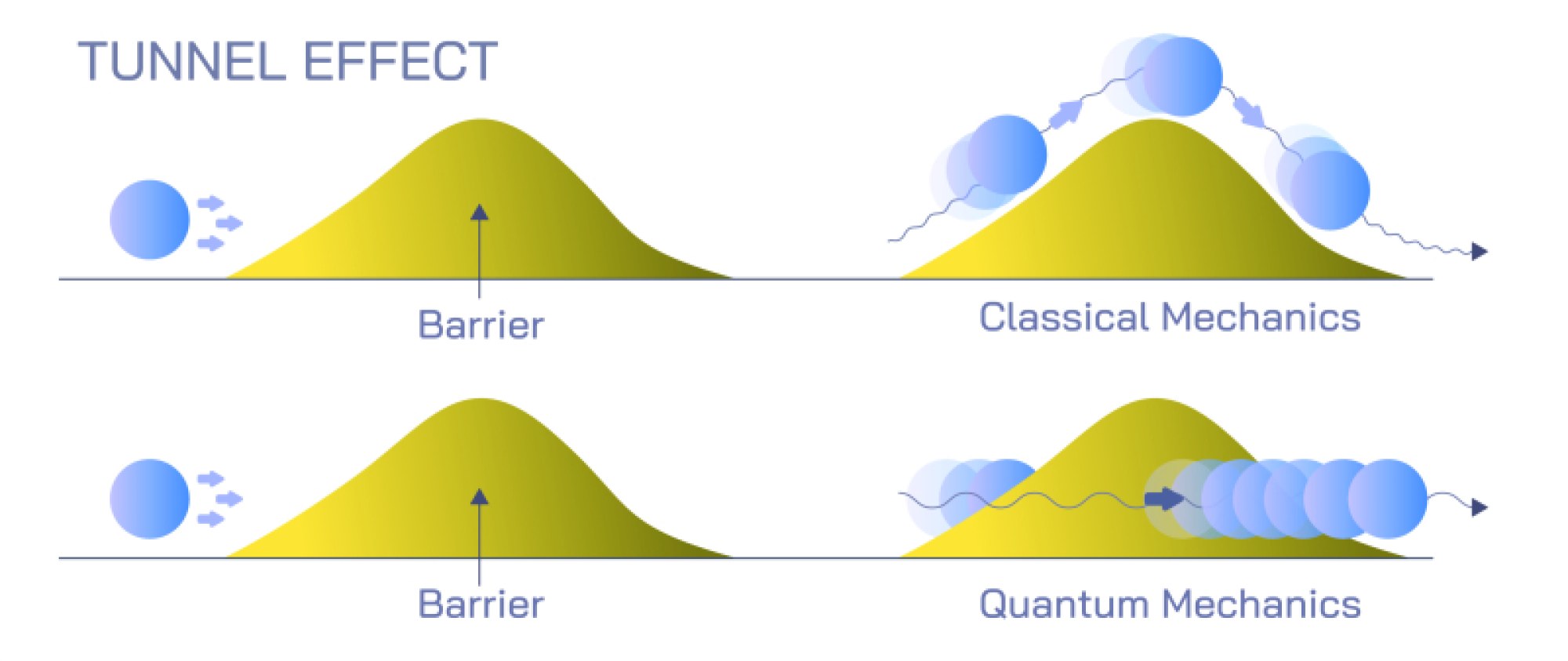
At the same time, quantum objects show particle behavior in ways that waves cannot. When light interacts with matter, it transfers energy in discrete packets called photons, not as a smooth and continuous wave. This was revealed in the photoelectric effect, where shining light on a metal surface ejects electrons only if the light possesses a specific minimum frequency. The intensity of the light does not matter unless the frequency threshold is met, proving that light interacts in quantized bursts rather than continuous spread. Likewise, electrons striking a detection screen produce individual localized impacts, not smeared-out energy distributions. When we observe a quantum entity to determine where it is — such as measuring the position of an electron — the wave-like probability distribution collapses into a definite detected point, reflecting particle identity. Thus, quantum objects behave as localized particles when measured, but as spatially distributed waves when unobserved.
The transition between wave and particle behavior hinges on measurement and interaction rather than identity change. A quantum object does not switch from being a wave to being a particle; instead, the wave function always exists, encoding the probabilities of different outcomes. When a detection occurs — when the quantum object interacts with a physical measuring device — the wave function narrows to a specific result, giving the appearance of particle-like behavior. The Heisenberg uncertainty principle reinforces this idea: certain properties (such as position and momentum) cannot both be known with arbitrary precision, because confining the wave function to determine a precise position inevitably spreads the momentum distribution. This is not a limitation of measurement tools but a fundamental property of nature: quantum systems are inherently probabilistic before measurement.
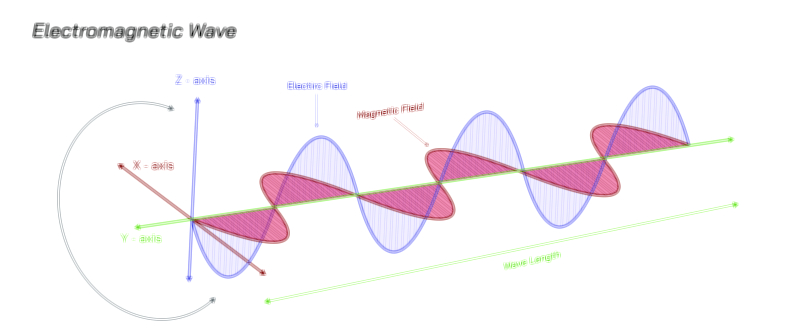
Wave–particle duality also reveals why microscopic and macroscopic worlds appear so different. In quantum phenomena, the wavelength associated with a particle depends on its momentum, as described by the de Broglie relation. Large everyday objects have extremely small wavelengths — too small to detect — which is why cars, people, and baseballs behave exclusively as classical particles. In contrast, lightweight particles such as electrons or photons have wavelengths large enough to create measurable interference and diffraction. The dual nature remains universal, but observable wave properties diminish rapidly as size and momentum increase. In other words, classical physics is not wrong — it is a limiting case of quantum physics that emerges for large or high-energy systems.
The wave–particle duality influences countless scientific and technological advancements. Electron microscopes rely on the wave nature of electrons to achieve magnification far beyond the limits of visible light. Lasers depend on the particle-like properties of photons interacting in quantized energy levels. Semiconductor devices, optical fibers, quantum computers, and superconducting circuits all exploit quantum duality as part of their function. Chemical bonding and electron orbitals emerge from electron wave behavior within atoms, shaping the periodic table and molecular structure. Meanwhile, photon quantization explains why atoms emit light at specific wavelengths, enabling atomic clocks and spectroscopy. The dual nature of matter and energy is not an anomaly — it is the source of patterns that govern both physical structure and technological innovation.
Wave–particle duality also has deep philosophical implications. It forces us to rethink whether physical objects possess definite properties before measurement or whether those properties arise only when measured. The dual behavior emphasizes that the act of observation is not passive — measurement interacts with the quantum system and changes it. Instead of the classical worldview where nature exists fully determined regardless of our presence, quantum mechanics reveals that reality at the microscopic scale evolves in terms of probabilities and interactions rather than deterministic outcomes. Quantum objects do not carry predetermined particle or wave characteristics; they behave according to a framework where the experiment itself determines which aspect becomes visible.
Ultimately, the quantum dual nature expressed through wave and particle properties shows that the microscopic world is governed by principles that cannot be captured by everyday intuition. An electron is not a particle that sometimes behaves like a wave, nor a wave that sometimes behaves like a particle — it is a fundamentally quantum entity described by a probability wave, which manifests as a localized particle only when measured. This unified interpretation resolves the duality not by choosing one behavior over the other, but by recognizing that both arise from the same underlying quantum description. Wave–particle duality remains one of the most elegant demonstrations of how deeply quantum physics reshapes our perception of reality — revealing that matter and energy at the smallest scale exist not as rigid objects but as dynamic systems of probability, interaction, and possibility.