The Tunnel Effect — Quantum Tunneling Phenomenon Explained
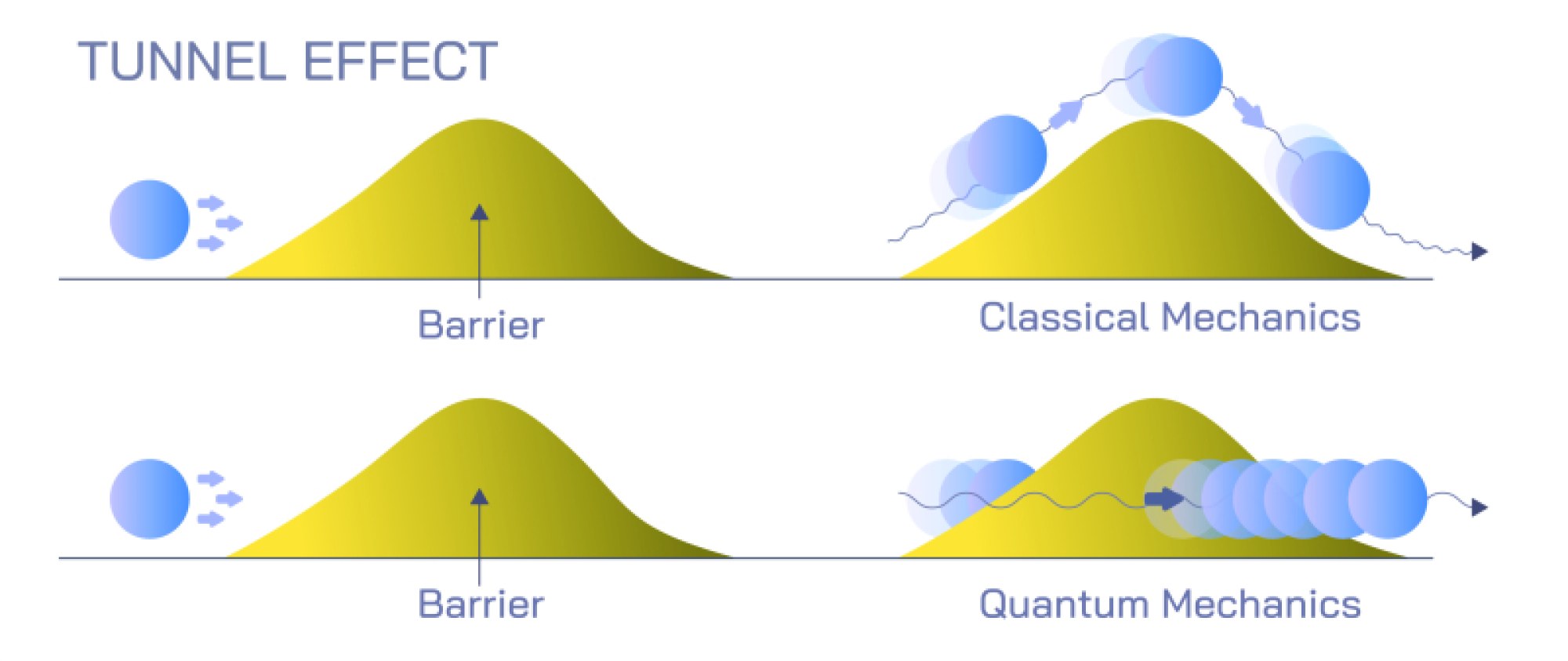
Quantum tunneling, also known as the tunnel effect, is one of the most astonishing and defining behaviors in quantum mechanics because it contradicts the logic of classical physics while shaping many of the processes that power the universe, from nuclear fusion in stars to the functioning of semiconductor devices in modern electronics. In classical physics, a particle such as a ball rolling toward a hill can cross to the other side only if it has enough energy to climb over the hill. If the energy is insufficient, the particle bounces back, unable to pass through the barrier. Quantum mechanics, however, describes a reality where subatomic particles do not behave like miniature solid balls but instead exist as probability waves that spread out in space. Because particles have wave-like nature, their location is not fixed until measured, and they can be found in places that classical rules would forbid. Quantum tunneling is the direct consequence of this wave–probability description. When a particle encounters an energy barrier higher than its kinetic energy, instead of stopping, there is a finite probability that part of its wave function continues through the barrier and emerges on the other side. In that sense, the particle appears to have crossed through a barrier it should not overcome, as though it has “tunneled” through rather than going over it, hence the name.
To visualize tunneling more intuitively, one can imagine an electron approaching a potential energy barrier—perhaps created by an electric field or a gap between two atoms. In classical physics, if the electron’s energy is lower than the height of the barrier, it must reflect because it lacks the required energy to pass. But quantum mechanics describes the electron not just as a point but as a wave packet, a spread of probability. When this probability wave reaches the barrier, it does not terminate abruptly; instead, the wave amplitude decays gradually within the barrier and, if the barrier is thin enough or low enough, part of the wave survives to the other side. This surviving portion means that there is a probability of detecting the electron beyond the barrier. The electron does not “break” the laws of conservation; it does not magically gain energy to leap over the barrier. Instead, because its wave function never fully vanished inside the barrier, there is a nonzero chance of finding the electron after the barrier. Quantum tunneling therefore does not violate physics — it reveals that the microscopic world operates under rules completely different from everyday intuition.
This phenomenon arises from the central equations of quantum theory, particularly the Schrödinger equation, which governs how the wave function evolves. When the wave function enters a region where classical physics would forbid the particle — such as inside an energy barrier — its amplitude decreases but does not instantly drop to zero. The rate of decrease depends on the barrier’s width, height, and the particle’s energy. If the barrier is extremely thick or high, the wave function decays too fully for tunneling to occur. If the barrier is relatively narrow or only moderately high, the probability remains significant enough that particles routinely tunnel across. In this way, quantum tunneling is not a rare event restricted to extreme conditions, but a natural process occurring constantly within atoms and materials. The mathematics quantifies this probability precisely and shows that tunneling efficiency increases for lightweight particles, high-energy particles, narrow barriers, and low barrier heights — which explains why tunneling is particularly prominent for electrons and subatomic particles rather than large macroscopic objects.
Quantum tunneling has profound effects on atomic structure and chemical reactions. In stars, including the Sun, the temperature in the core is not high enough for hydrogen nuclei to overcome their natural electrostatic repulsion by classical means. Without tunneling, protons would never collide close enough to initiate fusion, and stars would not burn. Quantum tunneling allows these nuclei to penetrate repulsive barriers and fuse, releasing energy that powers sunlight and sustains life on Earth. This same principle influences radioactive decay processes. In alpha decay, the alpha particle (two protons and two neutrons) is trapped within the nucleus by a strong nuclear potential barrier. Classical physics predicts that it should stay confined indefinitely if it lacks sufficient energy to escape. Yet quantum theory reveals that the alpha particle’s wave function extends into the barrier and beyond it, giving it a small but persistent probability of appearing outside the nucleus. Eventually it tunnels out, and the atom undergoes radioactive decay. The measured half-lives of radioactive isotopes correlate directly with tunneling probabilities, demonstrating how quantum mechanics governs nuclear stability.
Quantum tunneling is equally essential to the modern electronic world. Semiconductor devices — transistors, diodes, and integrated circuits — rely not only on the flow of electrons across materials but also on the precise design of barriers to control tunneling probability. In a tunnel diode, for example, electrons move through barriers deliberately engineered to enable rapid tunneling, producing extremely fast switching behavior. Scanning tunneling microscopes (STM), which revolutionized nanotechnology, operate by positioning a fine tip extremely close to a conducting surface. Electrons tunnel between the tip and the sample depending on their separation distance, generating a measurable current that varies with surface topography. This allows researchers to image individual atoms and manipulate molecules directly. Solid-state memory technologies such as flash drives store data by trapping electrons behind insulating barriers whose tunneling rates determine whether the cells represent logical “1” or “0.” Quantum tunneling defines how reliably the charge remains trapped and how long memory can be retained.
Biology exhibits possible influences of tunneling as well. Some enzymes accelerate biochemical reactions not only by lowering energy barriers but by positioning molecules so that protons or electrons can tunnel through hydrogen bonds. Quantum tunneling has been proposed to explain the efficiency of certain metabolic processes and even the ability of birds to navigate using magnetic fields, though research in quantum biology is still evolving. The ubiquity of tunneling in both cosmic and molecular scales illustrates how quantum laws shape natural processes at every level.
Despite its counterintuitive nature, quantum tunneling does not imply that macroscopic objects—such as a human or a car—could realistically tunnel through a wall. While the governing principles are universal, the probability depends on mass and barrier width, and the probability of tunneling for objects composed of billions of trillions of particles is effectively zero. Thus, the quantum world does not contradict the everyday world but underlies it at a scale where probability governs instead of strict determinism. Tunneling demonstrates that matter at the smallest scale behaves in terms of likelihood rather than certainty, revealing a universe governed by possibilities instead of rigid mechanical rules.
Quantum tunneling also highlights a deeper philosophical insight: the world we perceive with our senses is only one version of physical reality, shaped by macroscopic averages where improbabilities wash out. At the microscopic level, the universe thrives on probability waves, uncertainty, and nonclassical motion. Barriers that appear impenetrable under classical logic become semi-permeable under quantum rules. Particles move not because they overcome obstacles through added force but because their existence is not confined to a single precise position. Quantum tunneling therefore stands as one of the clearest demonstrations that quantum theory is not just a mathematical abstraction — it is a physical truth that governs the structure of matter, the light of stars, the flow of electricity, and the technology that powers every modern device.
Ultimately, the tunnel effect is a window into the profound divide between classical and quantum physics, showing how particles behave not like miniature versions of everyday objects but like waves governed by probabilities that extend beyond barriers. This phenomenon, once seen as paradoxical, is now recognized as essential to both the structure of the universe and the technology of modern civilization. Tunneling allows fusion to ignite stars, enables radioactive processes that shape planetary chemistry, powers quantum sensors and microscopes, and drives semiconductor components that form the foundation of digital electronics. By revealing that space is not rigid and barriers are not absolute, the quantum tunneling effect transforms the way science understands motion, energy, and the structure of the physical world.