Wave–Particle Duality — Nature of Light and Quantum Theory
Understanding the Dual Nature of Light in Classical and Modern Physics
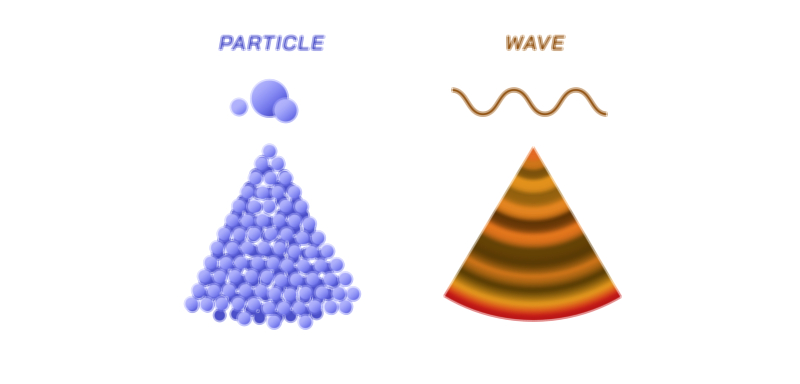
Wave–particle duality stands as one of the most profound concepts in modern physics, revealing that light and matter cannot be fully described by classical categories alone. For centuries, scientists debated whether light behaved like waves or particles, each model explaining certain phenomena while failing miserably at others. Wave theory, supported by interference and diffraction experiments, demonstrated that light spreads, bends, and overlaps in ways typical of waves. Yet particle-like behavior, such as the discrete emission and absorption of light or the sharp shadows cast by objects, suggested the presence of tiny, localized units. The puzzle deepened when experiments showed that neither wave theory nor particle theory alone could account for all observable behavior. The solution arrived with quantum mechanics, which proposed that light simultaneously exhibits characteristics of both waves and particles depending on the context of observation. This duality does not imply that light switches between two identities; rather, it possesses a unified quantum nature that manifests in complementary ways under different conditions. As such, the duality stands as a cornerstone of quantum theory, illustrating that at the smallest scales, nature transcends the limits of classical intuition.
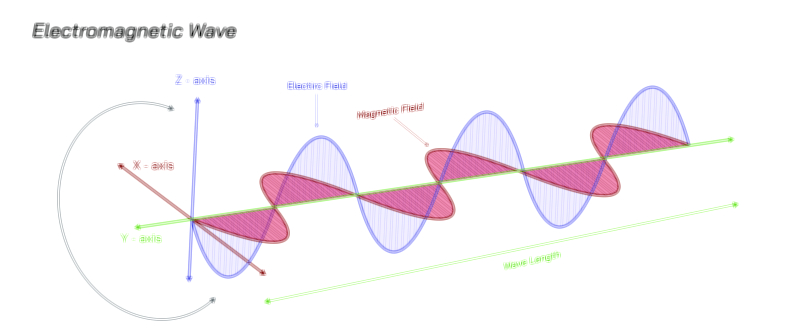
Wave Behavior: Interference, Diffraction, and the Continuity of Light
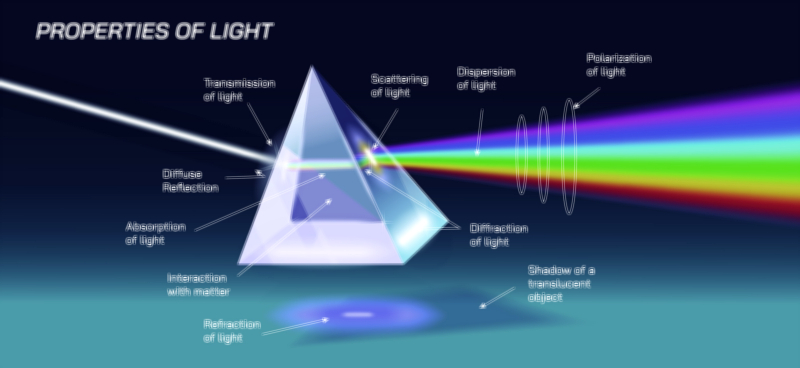
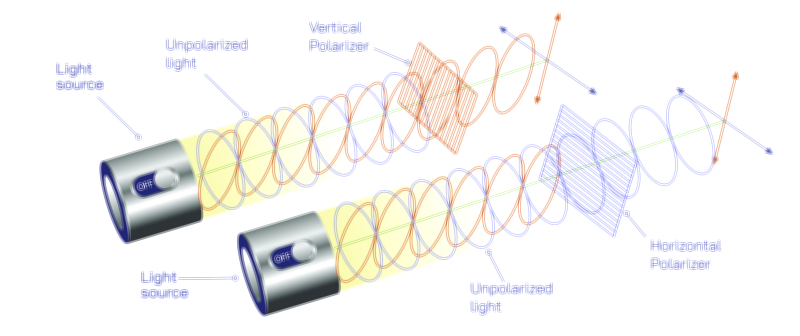
The wave-like behavior of light became evident through early optical experiments demonstrating interference patterns, where overlapping light waves produce alternating bright and dark fringes. This phenomenon cannot arise from classical particles, which would simply accumulate without forming structured patterns. Similarly, diffraction—where light bends around obstacles or spreads when passing through narrow openings—reveals that light propagates as a wave with a definable wavelength. These behaviors firmly established light as an electromagnetic wave, a view later supported by Maxwell’s equations, which showed that oscillating electric and magnetic fields naturally generate propagating waves. Within this framework, light travels through space with continuous energy flow, carrying momentum and information across vast distances. The wave model successfully describes reflection, refraction, polarization, and the formation of rainbows, making it indispensable for classical optics. However, wave theory alone falters when addressing the quantized nature of energy absorption or emission, compelling physicists to confront its limitations and reconsider the fundamental nature of light.
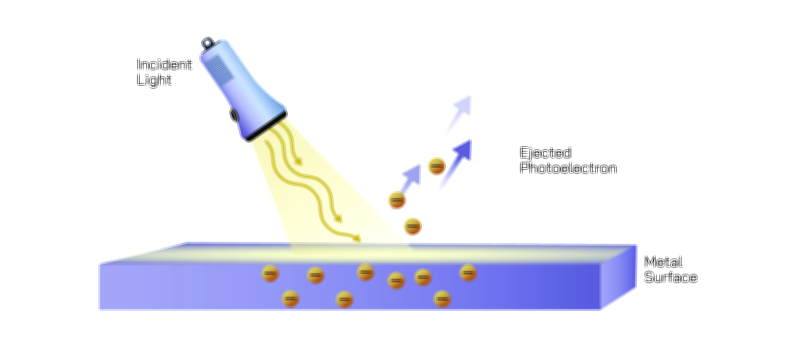
Particle Behavior: Photons, Quantization, and Einstein’s Revolutionary Insight
The particle nature of light emerged from observations that could not be reconciled through wave theory alone, most famously the photoelectric effect. In this process, light shining on a metal surface ejects electrons, but only if the light has a frequency above a certain threshold, regardless of its intensity. Classical wave theory predicted that intense light—regardless of frequency—should accumulate enough energy to eject electrons, yet this never occurred. Einstein provided the breakthrough by proposing that light consists of discrete packets of energy called photons. Each photon carries energy proportional to its frequency, meaning only sufficiently energetic photons can liberate electrons. This explanation not only resolved the photoelectric puzzle but also demonstrated that energy exchange between light and matter occurs in quantized steps, not continuous waves. Subsequent discoveries in Compton scattering and blackbody radiation further confirmed the particle-like behavior of light. Although photons possess no mass, they carry momentum and energy, interacting with matter in localized collisions that reveal a particle aspect underpinning much of quantum physics.
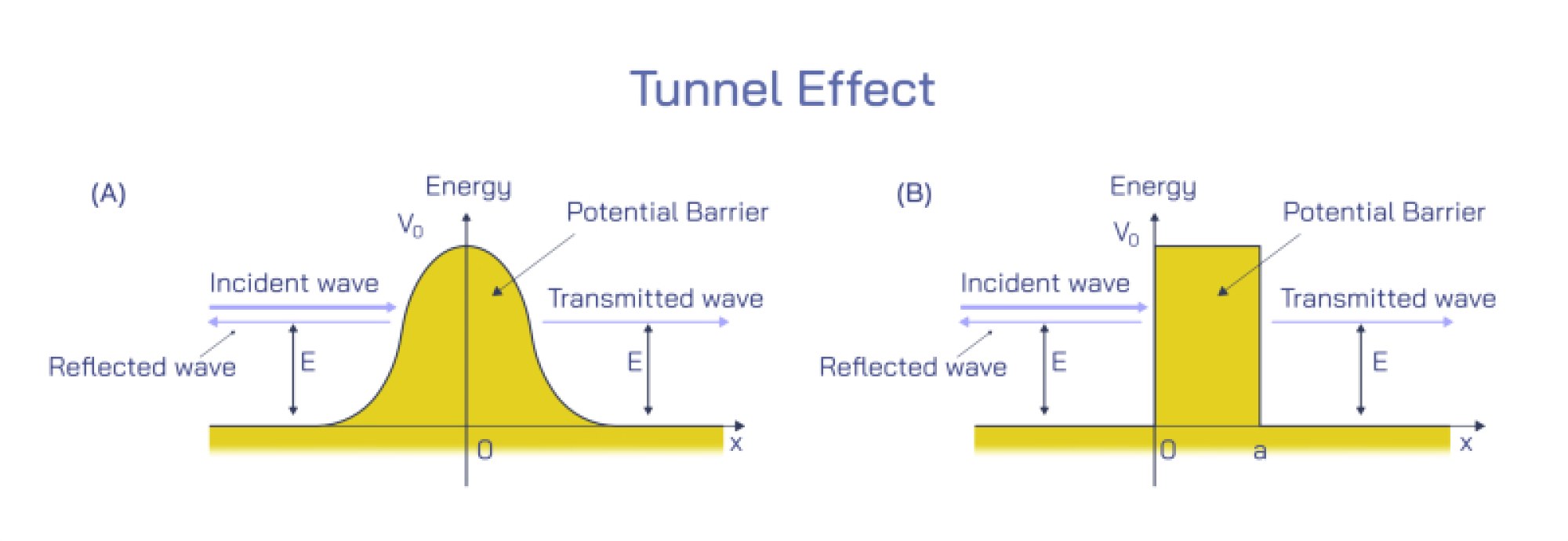
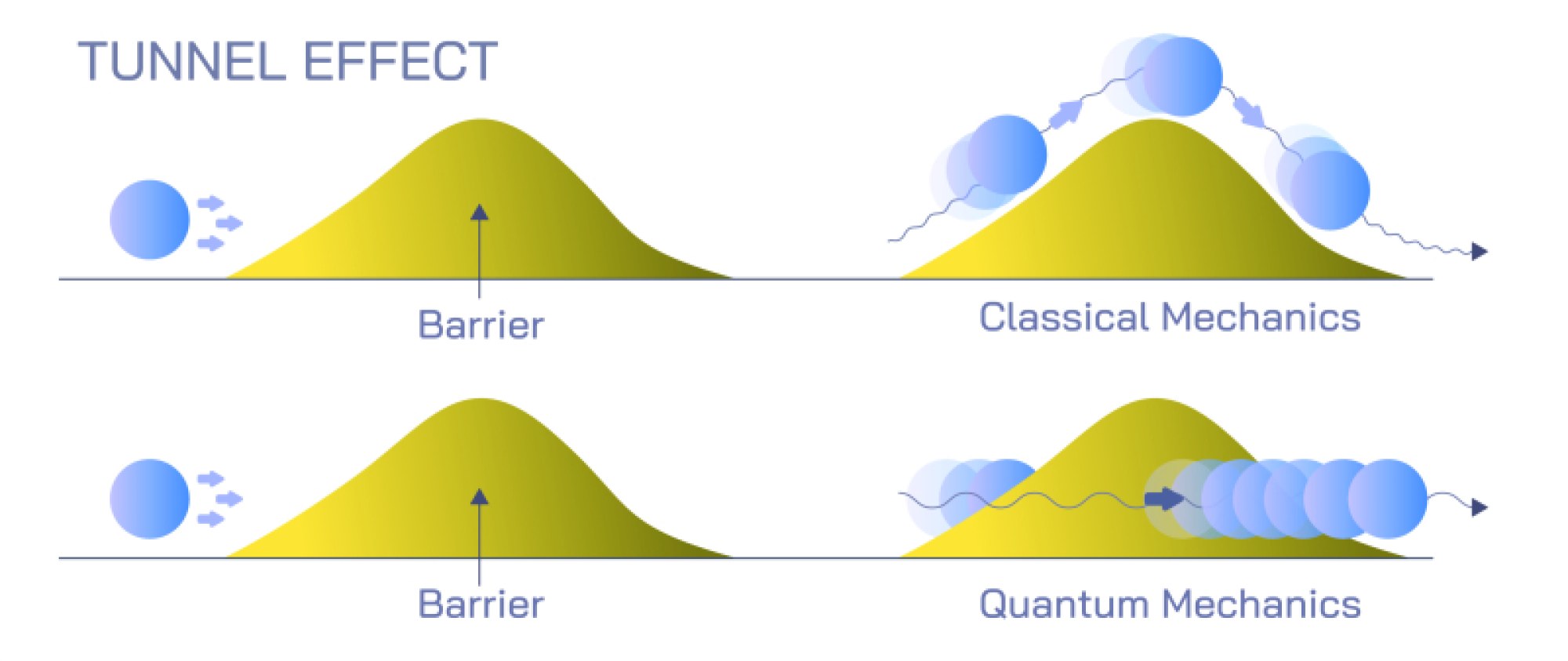
The Quantum Resolution: Complementarity and the Limits of Classical Intuition
Wave–particle duality does not imply that light alternates between wave and particle states; instead, it reflects the quantum principle of complementarity. According to this principle, wave-like and particle-like descriptions provide different perspectives on the same underlying quantum entity. What nature reveals depends on the experimental conditions. When physicists measure interference, they observe the wave aspect; when they detect individual photon impacts, they observe the particle aspect. These observations are not contradictory but instead complementary, each revealing a limited but necessary view of the full quantum picture. Ultimately, the true nature of light lies in the wavefunction—a mathematical object describing probability distributions rather than physical waves or particles in the classical sense. The wavefunction collapses into observable outcomes when measured, producing either wave-like or particle-like patterns depending on the experiment. This quantum interpretation transformed physics by showing that reality at microscopic scales does not align with classical expectations. The idea that particles can interfere with themselves, or that waves can arrive as discrete units, illuminates a deeper layer of existence governed by probabilistic laws rather than deterministic ones.
Wave–Particle Duality Beyond Light: Electrons and Matter Waves
Although wave–particle duality was first discovered for light, it extends to all matter, including electrons, atoms, and even large molecules. Experiments showed that electrons passing through two slits produce interference patterns, behaving as waves despite being detected as individual particles on a screen. This behavior is described by Louis de Broglie’s hypothesis, which states that all particles possess a wavelength inversely proportional to their momentum. De Broglie’s insight revealed that wave–particle duality is not unique to light but is a universal feature of quantum systems. This duality enabled the development of electron microscopy, diffraction-based material analysis, and quantum technologies that rely on controlling the wave-like behavior of matter. Even large molecular clusters, far heavier than electrons, have demonstrated interference patterns, proving that quantum behavior persists far beyond the atomic scale. As a result, wave–particle duality has become a unifying principle that links photonic behavior to the motion of particles, bridging electromagnetic theory with the physics of matter and solidifying the quantum framework that supports modern science.
Scientific and Technological Applications of Wave–Particle Duality
Wave–particle duality has shaped modern technology in profound ways. Lasers, for example, depend on the particle-like emission of photons at precise energy levels, while fiber-optic communication relies on the wave-like propagation of light through transparent media. In quantum computing, duality underpins phenomena such as superposition and interference, enabling qubits to perform computations far beyond classical capabilities. Electron microscopes exploit the wave nature of electrons to achieve atomic-scale imaging resolution, surpassing the limits of optical microscopy. Photodetectors, solar cells, and spectroscopy techniques rely on the quantized interactions between photons and electrons. In astrophysics, duality helps interpret radiation from distant galaxies, pulsars, and cosmic background radiation, revealing details about the birth and evolution of the universe. Across chemistry, materials science, and information technology, wave–particle duality remains a foundational concept enabling both theoretical breakthroughs and practical innovations.
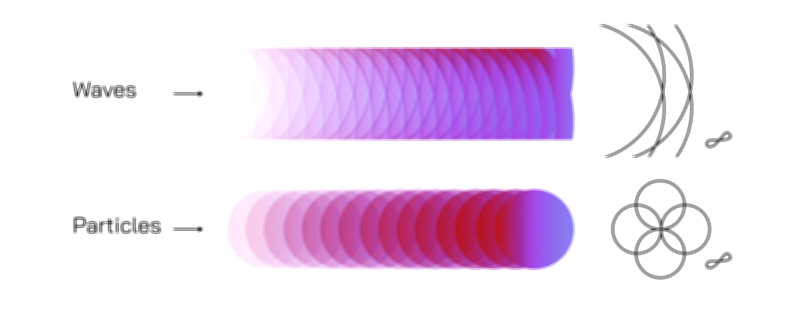
Illustrating Wave–Particle Duality and Quantum Behavior
Illustrations of wave–particle duality often show light or electrons represented as wavy lines merging into localized particle impacts on a detection screen. The iconic double-slit experiment is frequently depicted to show how waves create interference patterns while particles appear as individual dots that gradually form those same patterns over time. Other illustrations highlight photons as energy packets, wavefunctions as probability clouds, and the transition between wave-like propagation and particle-like detection. These diagrams help visualize the abstract concept that quantum entities do not possess fixed identities but instead express different aspects of their nature depending on measurement. Through such visualizations, the mysterious but elegant principle of wave–particle duality becomes clearer, revealing the quantum foundation underlying modern science.